Abstract
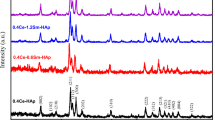
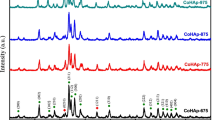
The high-temperature phase stability of Ca10−x Cs x (PO4)6(OH)2, (x = 0–3) compositions synthesized by various wet chemical methods was investigated. The thermal expansion property of Ca10(PO4)6(OH)2 (abbreviated as CaHAp) and Cs-substituted CaHAp was measured by high-temperature XRD and dilatometry. The average crystallite size of the powders synthesized by wet chemical methods was found to be 10–50 nm range as shown by XRD and TEM. Up to 30 mol% Cs loading was observed to show only the apatite phase by XRD when the apatite powder was nanocrystalline in nature. However, high-temperature stability of the Cs-substituted system is limited to ≤5 mol%. Cs3(PO4) is observed to be separated out on heating the material above 773 K for compositions substituted with more than 5 mol% of Cs in the Ca-sublattice. The coefficient of thermal expansion measured by HTXRD is αa = 12.42 × 10−6 K−1, αc = 14.98 × 10−6 K−1; and αa = 12.62 × 10−6 K−1, αc = 12.57 × 10−6 K−1 for CaHAp and Ca9.78Cs0.2(PO4)6(OH)1.96, respectively, in the temperature range of 298-1083 K. Bulk thermal expansion measurements are seen to be in agreement with the lattice expansion results.










Similar content being viewed by others
References
M. Mathew and S. Tagaki, Structures of Biological Materials in Dental Research, J. Res. Natl Inst. Stand. Technol., 2001, 106(6), p 1035–1044
M. Wei, J.H. Evans, T. Bostrom, and L. Grøndhal, Synthesis and Characterization of Hydroxyapatite, Fluoride-Substituted Hydroxyapatite and Fluorapatite, J. Mater. Sci. Mater. Med., 2003, 14, p 311–320
M.E. Fleet and Y. Pan, Site Preference of Nd in Fluorapatite [Ca10 (PO4)6F2], J. Solid State Chem., 1994, 112, p 78–81
P. Trocellier, Immobilization of Radionuclides in Single-Phase Crystalline Waste Forms: A Review on Their Intrinsic Properties and Long Term Behaviour, Ann. Chim. Sci. Mat., 2000, 25, p 321–337
J.O. Nriagu, Lead Orthophosphates. IV. Formation and Stability in the Environment, Geochim. Cosmochim. Acta, 1974, 38, p 887–898
J.O. Nriagu, Lead Orthophosphates—II. Stability of Cholopyromophite at 25 °C, Geochim. Cosmochim. Acta, 1973, 37, p 367–377
J.O. Nriagu, Lead Orthophosphates—III. Stabilities of Fluoropyromorphite and Bromopyromorphite at 25 °C, Geochim. Cosmochim. Acta, 1973, 37, p 1735–1743
L.S. Keto and S.B. Jacobsen, Nd and Sr Isotopic Variations of Early Paleozoic Oceans, Earth and Planet, Sci. Lett., 1987, 84, p 27–41
J.L. Conca and J. Wright, An Apatite II, Permeable Reactive Barrier to Remediate Groundwater Containing Zn, Pb and Cd, Appl. Geochem., 2006, 21, p 1288–1300
X. Chen, J.V. Wright, J.L. Conca, and L.M. Peurrung, Effects of pH on Heavy Metal Sorption on Mineral Apatite, Environ. Sci. Technol., 1997, 31(3), p 624–631
M.I. Kay, R.A. Young, and A.S. Posner, Crystal Structure of Hydroxyapatite, Nature, 1964, 204, p 1050–1052
A. Chartier, C. Meis, and J.D. Gale, Computational Study of Cs Immobilization in the Apatites Ca10(PO4)6F2, Ca4La6(SiO4)6F2 and Ca2La8(SiO4)6O2, Phy. Rev. B, 2001, 64, p 9–085110
H. Jena, R. Asuvathraman, and K.V.G. Kutty, Phase Stability and Thermal Expansion Studies on Cs-Substituted Nano-Crystalline Ca-Hydroxyapatites Synthesized by Novel Wet Chemical Methods, Proceedings of 16th National Symposium on Thermal Analysis (THERMANS-2008), S. Varma et al., Ed., 4-6 Feb, 2008, IGCAR, Kalpakkam, India, p 202–204
B.D. Cullity and S.R. Stock, Elements of X-Ray Diffraction, 3rd ed., Prentice-Hall Inc, Upper Saddle River, 2001, p 167–171
R. Jenkins and R.L. Snyder, Introduction to X-ray Powder Diffractometry, Wiley & Sons Inc, New York, 1996, p 89–91
K.V. Govindan Kutty, R. Asuvathraman, M.V. Krishnaiah, V. Ganesan, R. Parthasarathy, D. Sai Subalakshmi, B. Suhasini, K.C. Srinivas, K.A Gopal, and P.V. Kumar, Design, Fabrication and Commissioning of a Push Rod Dilatometer for Thermal Expansion Studies on Solids, IGC-283, Indira Gandhi Centre for Atomic Research, Kalpakkam, India, 2006
H. Jena, Ch.V. Rao, F.P. Eddy, J. Dooley, and B. Rambabu, Structural and Proton Transport Studies on Nano-Crystalline [Ca10(PO4)6(OH)2] (HAp), HAp-Nafion® Composite, and Natural Human Bbone, Phys. Status Solidi A, 2009, 206(11), p 2536–2541
H. Fernandez-Morán and A. Engström, Electron Microscopy and X-ray Diffraction of Bone, Biochim. Biophys. Acta, 1957, 23, p 260–264
R.D. Shannon, Revised Effective Ionic Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides, Acta Cryst., 1976, A32, p 751–767
S. Koutsopoulos, Synthesis and Characterization of Hydroxyapatite Crystals: A Review Study on the Analytical Methods, J. Biomed. Mater. Res., 2002, 62, p 600–612
H. Yu, H. Zhang, X. Wang, Z. Gu, X. Li, and F. Deng, Local Structure of Hydroxy Peroxy Apatite: A Combined XRD, FT-IR, Raman, SEM, and Solid State NMR Study, J. Phys. Chem. Solid, 2007, 68(10), p 1863–1871
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jena, H., Asuvathraman, R. & Govindan Kutty, K.V. Thermal Expansion and Phase Stability Investigations on Cs-Substituted Nanocrystalline Calcium Hydroxyapatites. J. of Materi Eng and Perform 20, 108–113 (2011). https://doi.org/10.1007/s11665-010-9651-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11665-010-9651-z