Abstract
The liquid phase thermodynamics of mixing of the copper-aluminum binary system are investigated as a function of temperature and composition using the electrochemical potential difference method. A copper-selective beta″ alumina is used as a solid electrolyte, synthesized through ion exchange, sintering from base oxide powders, and the floating zone method of crystal growth. Measured thermodynamics of mixing data were used to inform short-range ordering in copper-aluminum melts through Darken’s factor for excess stability and Bhatia–Thornton structure factors, revealing a strong departure from ideality and pronounced ordering. Mixing properties were used to predict viscosity and self-diffusion coefficients. Features observed in calculated electronic entropy of mixing for copper-aluminum were compared with trends in viscosity, demonstrating the utility of electronic properties of mixing in the description of structure–properties in this liquid binary system.




















Similar content being viewed by others
References
M.T. Di Giovanni, E. Cerri, T. Saito, S. Akhtar, P. Åsholt, Y. Li, and M. Di Sabatino: Metall. Ital., 2016, vol. 108, pp. 43–7.
R.A. Gonçalves and M.B. da Silva: Procedia Manuf., 2015, vol. 1, pp. 683–95.
B. Upton: Corrosion, 1963, vol. 19, p. 204t–209t.
Y. Li, T.L. Ngai, and W. Xia: Wear, 1996, vol. 197, pp. 130–6.
M. Založnik, B. Sarler, and D. Gobin: Mater. Tehnol., 2004, vol. 38, pp. 249–55.
G. Gaustad, E. Olivetti, and R. Kirchain: Resour. Conserv. Recycl., 2012, vol. 58, pp. 79–87.
S.R. Wagstaff: J. Sib. Fed. Univ. Eng. Technol., 2018, vol. 11, pp. 409–18.
N. Saunders: in COST 507 Definition of Thermochemical and Thermophysical Properties to Provide a Database for the Development of New Light Alloys, I. Ansara, A.T. Dinsdale, and M.H. Rand, eds., vol. 2, 1998, pp. 28–33.
A.I. Zaitsev, R.Y. Shimko, N.A. Arutyunyan, and S.F. Dunaev: Dokl. Phys. Chem., 2007, vol. 414, pp. 115–9.
H. Oyamada, T. Nagasaka, and M. Hino: Mater. Trans. JIM, 1998, vol. 39, pp. 1225–9.
D.S. Kanibolotsky, O.A. Bieloborodova, N. V Kotova, and V. V Lisnyak: J. Therm. Anal. Calorim., 2002, vol. 70, pp. 975–83.
U.K. Stolz, I. Arpshofen, F. Sommer, and B. Predel: J. Phase Equilibria, 1993, vol. 14, pp. 473–8.
M. Schick, J. Brillo, I. Egry, and B. Hallstedt: J. Mater. Sci., 2012, vol. 47, pp. 8145–52.
V.T. Witusiewicz, U. Hecht, S.G. Fries, and S. Rex: J. Alloys Compd., 2004, vol. 385, pp. 133–43.
A.S. Roik, V.P. Kazimirov, and V.E. Sokolskii: Russ. Metall., 2014, vol. 2014, pp. 23–32.
J. Brillo, A. Bytchkov, I. Egry, L. Hennet, G. Mathiak, I. Pozdnyakova, D.L. Price, D. Thiaudiere, and D. Zanghi: J. Non. Cryst. Solids, 2006, vol. 352, pp. 4008–12.
J. Dziedzic, S. Winczewski, and J. Rybicki: Comput. Mater. Sci., 2016, vol. 114, pp. 219–32.
N. Jakse and A. Pasturel: Phys. Rev. B, 2016, vol. 94, pp. 1–13.
P. Dinsdale and P.N. Quested: J. Mater. Sci., 2004, vol. 9, pp. 7221–8.
J. Schmitz, J. Brillo, I. Egry, and R. Schmid-Fetzer: Int. J. Mat. Res., 2009, vol. 100, pp. 1529–35.
A.R. Kurochkin, P.S. Popel, D.A. Yagodin, A. V Borisenko, and A. V Okhapkin: High Temp., 2013, vol. 51, pp. 224–32.
N.Y. Konstantinova, A.R. Kurochkin, A. V. Borisenko, V. V. Filippov, and P.S. Popel: Russ. Metall., 2016, vol. 2016, pp. 144–9.
A.M. Vora: Phys. Chem. Liq., 2008, vol. 46, pp. 213–22.
R.M. Khusnutdinoff, A.V. Mokshin, S.G. Menshikova, A.L. Beltyukov, and V.I. Ladyanov: J. Exp. Theor. Phys., 2016, vol. 122, pp. 859–68.
M. V. Vaghela, A. Y. Vahora, N.K. Bhatt, B.Y. Thakore, and A.R. Jani: AIP Conf. Proc., 2013, vol. 1536, pp. 637–8.
Y. Plevachuk, V. Sklyarchuk, A. Yakymovych, S. Eckert, B. Willers, and K. Eigenfeld: Metall. Mater. Trans. A Phys. Metall. Mater. Sci., 2008, vol. 39, pp. 3040–5.
Z. Wen, J. Cao, Z. Gu, X. Xu, F. Zhang, and Z. Lin: Solid State Ionics, 2008, vol. 179, pp. 1697–1701.
H. Kuwamoto and H. Sato: Solid State Ionics, 1981, vol. 5, pp. 187–8.
N. Weber: Energy Convers., 1974, vol. 14, pp. 1–8.
T.K. Hunt, N. Weber, and T. Cole: Solid State Ionics, 1981, vol. 5, pp. 263–5.
S.M. Jeter: Energy, 1987, vol. 12, pp. 163–70.
D.J. Fray: Met. Trans. B, 1977, vol. 8B, pp. 153–6.
J. Liu and W. Weppner: Solid State Commun., 1990, vol. 76, pp. 311–3.
J. Vangrunderbeek, F. Vandecruys, and R. V. Kumar: Solid State Ionics, 2000, vol. 136–137, pp. 567–71.
G. Dorner, H. Durakpasa, P. Linhardt, and M.W. Breitter: Electrochim. Acta, 1991, vol. 36, pp. 563–8.
R. Kvachkov, A. Yanakiev, C.N. Poulieff, P.D. Yankulov, S. Rashkov, and E. Budevski: Solid State Ionics, 1982, vol. 7, pp. 151–5.
R.J. Baughman and R.A. Lefever: Mater. Res. Bull., 1975, vol. 10, pp. 607–12.
M.S. Whittingham and R.A. Huggins: J. Electrochem. Soc., 1971, vol. 118, pp. 1–6.
J. Briant and G. Farrington: J. Electrochem. Soc., 1981, vol. 128, pp. 1830–4.
G.M. Crosbie and G.J. Tennenhouse: J. Am. Ceram. Soc., 1982, vol. 65, pp. 187–91.
Y. Yu Yao and J.T. Kummer: J. Inorg. Nucl. Chem., 1967, vol. 29, pp. 2453–75.
J.A. Little and D.J. Fray: Electrochim. Acta, 1980, vol. 25, pp. 957–64.
T. Oishi, S. Tagawa, and S. Tanegashima: J. Phys. Chem. Solids, 2005, vol. 66, pp. 251–5.
J.A. Little and D.J. Fray: Trans. Inst. Min. Metall., 1979, vol. 88, pp. C229–33.
J.D. Barrie, B. Dunn, O.M. Stafsudd, and P. Nelson: J. Lumin. 39: 29-33 (1987).
B. Cleaver and A. Davies: Electrochim. Acta, 1973, vol. 18, pp. 733–9.
S.K. Kim: J. Korean Ceram. Soc., 1995, vol. 32, pp. 1040–6.
H. Kim, D.A. Boysen, D.J. Bradwell, B. Chung, K. Jiang, A.A. Tomaszowska, K. Wang, W. Wei, and D.R. Sadoway: Electrochim. Acta, 2012, vol. 60, pp. 154–62.
M.D. Koretsky: Engineering and Chemical Thermodynamics, 2nd edn., Wiley, Hoboken, 2013.
M.-L. Saboungi, W. Geertsma, and D.L. Price: Annu. Rev. Phys. Chem., 1990, vol. 41, pp. 207–44.
M. Saboungi, J. Marr, and M. Blander: J. Chem. Phys., 1978, vol. 68, pp. 1375–84.
C.C. Rinzler and A. Allanore: Philos. Mag., 2016, vol. 96, pp. 3041–53.
H. Eyring: J. Chem. Phys., 1936, vol. 4, pp. 283–91.
T. Tanaka, K. Hack, and S. Hara: MRS Bull., 1999, vol. 24, pp. 45–9.
S. Seetharaman and D. Sichen: Metall. Mater. Trans. B, 1994, vol. 25, pp. 589–95.
J. Brillo: Thermophysical Properties of Multicomponent Liquid Alloys, Walter de Gruyter GmbH & Co KG, Berlin, 2016.
R.N. Singh and F. Sommer: Phys. Chem. Liq., 1998, vol. 36, pp. 17–28.
H.A. Dabkowska, A.B. Dabkowski, R. Hermann, J. Priede, and G. Gerbeth: in Handbook of Crystal Growth: Bulk Crystal Growth: Second Edition, 2014.
J.L. Murray: Int. Met. Rev., 1985, vol. 30, pp. 211–34.
G.H. Teichert, N.S.H. Gunda, S. Rudraraju, A.R. Natarajan, B. Puchala, K. Garikipati, and A. Van der Ven: Comput. Mater. Sci., 2017, vol. 128, pp. 127–39.
C. Wagner: Thermodynamics of Alloys, Addison-Wesley Press, Inc, Cambridge, MA, USA, 1952.
M. Trybuła, N. Jakse, W. Gąsior, and A. Pasturel: Arch. Metall. Mater., 2015, vol. 60, pp. 649–55.
J.-L. Bretonnet, J. Auchet, and J.G. Gasser: J. Non. Cryst. Solids, 1990, vol. 117–118, pp. 395–8.
H.U. Kunzi and H. Guntherodtf: in The Hall Effect and its Applications, C.L. Chien and C.R. Westgate, eds., Springer, New York, NY, 1980, pp. 215–52.
S. Takeuchi and K. Murakami: Sci. Reports Res. Institutes, Tohoku Univ. Ser. A, Physics, Chem. Metall., 1974, vol. 25, pp. 73–86.
S. Glasstone, K.J. Laider, and H. Eyring: The Theory of Rate Processes; The Kinetics of Chemical Reactions, Viscosity, Diffusion, and Electrochemical Phenomena, McGraw-Hill, 1941.
R.E. Aune, M. Hayashi, and S. Sridhar: Ironmak. Steelmak., 2005, vol. 32, pp. 141–50.
O. Kubaschewski and C.B. Alcock: Metallurgical Thermochemistry, 5th edn., Pergamon Press, Oxford, 1979.
T. Ejima and T. Yamamura: J. Phys. Colloq., 1980, vol. 41, pp. 345-348.
H.L. Lukas, S.G. Fries, and B. Sundman: Computational Thermodynamics: The Calphad Method, Cambridge University Press, New York, NY, 2007.
Acknowledgments
The authors wish to thank the MIT UROP Office and the Sanders, Lord, and DeFlorez UROP Funds for their financial support to this project. The authors wish to thank Mr. Andrew Caldwell, Ms. Jaclyn Leigh Cann, Mr. Brian Chmielowiec, Dr. Katsuhiro Nose, and Dr. Youyang Zhao for their insight and assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted April 8, 2018.
Appendices
Appendix A: Example of Raw OCP Data
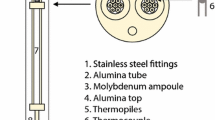
Depicted here is an example of open-circuit potential (OCP) data collected for Cu0.4Al0.6 vs Cu utilizing the copper-selective beta″ alumina (Cuβ″Al2O3) solid electrolyte described in this study. At a given temperature and composition, steady state in OCP readings was achieved within 1800 seconds. Typical standard deviations (SD) in OCP across a measurement period ranged from 0.02 to 2 mV, increasing with higher temperatures (Figure A1).
Appendix B
See Table BI.
Appendix C: List of Symbols
- \( a \) :
-
Activity
- \( c \) :
-
Molar concentration
- \( D \) :
-
Self-diffusion coefficient
- \( E \) :
-
Cell potential difference
- \( E^{A} \) :
-
Activation energy for viscous flow
- \( ES \) :
-
Excess stability
- \( F \) :
-
Faraday constant
- \( \bar{H} \) :
-
Partial molar enthalpy
- \( k_{B} \) :
-
Boltzmann’s constant
- \( M \) :
-
Hall mobility
- \( r \) :
-
Radius of diffusing particle
- \( R \) :
-
Ideal gas constant
- \( R_{H} \) :
-
Hall effect coefficient
- \( \bar{S} \) :
-
Partial molar entropy
- \( S_{CC} \left( 0 \right) \) :
-
Bhatia–Thornton structure factor
- \( S^{e} \) :
-
Electronic state entropy
- \( T \) :
-
Absolute temperature
- \( x \) :
-
Mole fraction
- \( \bar{Y} \) :
-
Partial molar quantity
- \( z \) :
-
Number of electrons per copper(I) ion that travels across the Cuβ″Al2O3 membrane
- \( \alpha \) :
-
Seebeck coefficient
- \( \Delta G^{M} \) :
-
Gibbs energy of mixing
- \( \Delta H^{M} \) :
-
Enthalpy of mixing
- \( \Delta S^{M} \) :
-
Entropy of mixing
- \( \eta \) :
-
Dynamic viscosity
- \( \eta^{\infty } \) :
-
Pre-exponential factor for viscous flow
- \( \mu \) :
-
Partial molar Gibbs energy
- \( \sigma \) :
-
Electrical conductivity
Rights and permissions
About this article
Cite this article
Stinn, C., Allanore, A. Thermodynamic and Structural Study of the Copper-Aluminum System by the Electrochemical Method Using a Copper-Selective Beta″ Alumina Membrane. Metall Mater Trans B 49, 3367–3380 (2018). https://doi.org/10.1007/s11663-018-1400-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-018-1400-y