Abstract
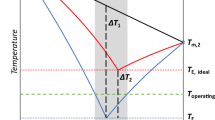
In order to study the theoretical cycle characteristic of [mmim]DMP (1-methyl-3-methylimidazolium dimethylphosphate) /methanol absorption refrigeration, the modified UNIFAC group contribution model and the Wilson model are established through correlating the experimental vapor pressure data of [mmim]DMP/methanol at T=280∼370 K and methanol mole fraction x= 0.529∼0.965. Thermodynamic performances of absorption refrigeration utilizing [mmim]DMP/methanol, LiBr/H2O and H2O/NH3 are investigated and compared with each other under the same operating conditions. From the results, some conclusions are obtained as follows: 1) the circulation ratio of the [mmim]DMP /methanol absorption refrigeration is higher than that of the LiBr/H2O absorption refrigeration, but still can be acceptable and tolerable. 2) The COP of the [mmim]DMP/methanol absorption refrigeration is smaller than that of the LiBr/H2O absorption refrigeration, while it is higher than that of the H2O/NH3 absorption refrigeration under most operating conditions. 3) The [mmim]DMP/methanol absorption refrigeration are still available with high COP when the heat source temperature is too high to drive LiBr/H2O absorption refrigeration.
Similar content being viewed by others
References
Pedro F. A., Adrián R. P. et al., “Modeling of high-pressure vapor-liquid equilibrium in ionic liquids + gas systems using the PRSV equation of state”, Fluid Phase Equilibria, Vol. 295, No. 1 (2010), pp. 9–16.
Mara G. F., Luís M. S. et al., “Evaluation of COSMO-RS for the prediction of LLE and VLE of alcohols + ionic liquids”, Fluid Phase Equilibria, Vol. 255, No. 2 (2007), pp. 167–178.
Liang S. Q., Chen W. et al., “The latent application of ionic liquids in absorption refrigeration”, Application of ionic liquids in science and technology, InTechOpen, Croatia (2011), pp. 467–494.
Mark B. S., Akimichi Y., “Absorption cycle using ionic liquids as working fluids”, USA (2006), Patent No. US2006/0197053 A1.
Akimichi Y., Mark B. S., “Vapor-liquid equilibria of ammonia + ionic liquid mixtures”, Applied Energy, Vol. 84, No. 12 (2007), pp. 1258–1273.
Cai W. H., Sen M., Paolucci S., “Dynamic modeling of an absorption refrigeration”, Proceedings of IMECE 2007, Seattle, Washington, USA: ASME, Vol. 6 (2007), pp. 227–236.
Ángel M., María D. B., “Thermodynamic analysis of absorption refrigeration cycles using ionic liquid + supercritical CO2 pairs”, Supercritical Fluids, Vol. 55, No. 2 (2010), pp. 852–859.
Kim K. S., Shin B. K., Lee H. et al., “Refractive index and heat capacity of 1-Butyl-3-Methylimidazolium bromide and 1-Butyl-3-Methylimidazolium tetrafluoroborate and vapor pressure of binary systems for 1-Butyl-3-Methylimidzolium bromide + trifluoroethanol and 1-Butyl-3-Methylimidazolium tetrafluoroborate + trifluoroethanol”, Fluid Phase Equilibria, Vol. 218, No. 2 (2004), pp. 215–220.
Zhang X. D., Hu. D. P., “Performance simulation of the absorption chiller using water and ionic liquid 1-ethyl-3-methylimidazolium dimethylphosphate as the working pair”, Applied Thermal Engineering, Vol. 31, No. 16 (2010), pp. 3316–3321.
Zhao J., Liang S. Q. et al., “Vapor liquid equilibrium for high concentration solution of 1, 3-dimethylimidazolium dimethylphosphate/methanol”, Chemical Engineering, Vol. 38, No. 3 (2010), pp. 53–56.
Liang S. Q., Zhao J. et al., “Absorption refrigeration cycle utilizing a new working pair of ionic liquid type”, Journal of Engineering Thermophysics, Vol. 31, No. 10 (2010), pp. 1627–1630.
Zhao J., Jiang X. C., Li C. X. et al., “Vapor pressure measurement for binary and ternary systems containing a phosphoric ionic liquid”, Fluid Phase Equilibria, Vol. 247, No. 1–2 (2006), pp. 190–198.
Fredenslund A., Gmehling J., Rasmussen P., “Vapor-liquid equilibria using UNIFAC: a group contribution method”, Elsevier Scientific Publishing Co., New York (1997).
Wang D., Xuan A. G. et al., “Study on gas-liquid equilibria with the UNIFAC model for the systems of synthesizing dimethyl carbonate”, Fluid Phase Equilibria, Vol. 302, No. 1–2 (2011), pp. 269–273.
Silke N., Jürgen G., “Prediction of phase equilibria and excess properties for systems with ionic liquids using modified UNIFAC: typical results and present status of the modified UNIFAC matrix for ionic liquids”, AIChE Journal, Vol. 49, No. 2 (2003), pp. 530–537.
Wang J. F., Sun W. et al., “Correlation of infinite dilution activity coefficient of solute in ionic liquid using UNIFAC model”, Fluid Phase Equilibria, Vol. 264, No. 1–2 (2008), pp. 235–241.
Juan A. G., Isaiés G. F. et al., “Thermodynamics of mixtures with strongly negative deviations from Raoult’s Law Part 4. Application of the DISQUAC model to mixtures of 1-alkanols with primary or secondary linear amines. Comparison with Dortmund UNIFAC and ERAS results”, Fluid Phase Equilibria, Vol. 168, No. 1 (2000), pp. 31–58.
Li S. Y., “The determination and correlation of the excess enthalpies for the binary system of alcohols+ (ketones, or ionic liquids) and the application of COSMO-type model”, Zhejiang University, Hangzhou (2008).
Chen N. H., “Generalized correlation for latent heat of vaporization”, Journal of Chemical and Engineering Data, Vol. 10, No. 2 (1962), pp. 207–210.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chen, W., Liang, S., Guo, Y. et al. Thermodynamic performances of [mmim]DMP/Methanol absorption refrigeration. J. Therm. Sci. 21, 557–563 (2012). https://doi.org/10.1007/s11630-012-0581-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11630-012-0581-y