Abstract
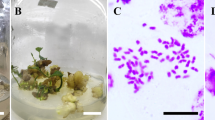
Dioscorea zingiberensis is an important medicinal plant and a source of diosgenin in China. We report research on the induction, characteristics, and chemical assays of polyploid plants of D. zingiberensis. Immersing calli in 0.3% colchicine solution for 16 h prior to culture induced a high number of autotetraploid plants. The induction rate reached as high as 36.7% of treated calli. More than 50 lines of autotetraploid plants were obtained. All tetraploid plants showed typical polyploidy characteristics. Twenty selected tetraploid lines were transferred to the field for determination of morphological characteristics and for chemical assays. Six elite lines have been selected for further selection and breeding into new varieties for commercial production.



Similar content being viewed by others
References
Ackerman, W. L.; Dermen, H. A fertile colchiploid from a sterile interspecific camellia hybrid. J. Hered. 63: 55–59; 1972.
Adanyia, S.; Shirai, D. In vitro induction of tetraploid ginger (Zingiber officinale Roscoe) and its pollen fertility and germinability. Sci. Hort. 88: 277–287; 2001 doi:10.1016/S0304-4238(00)00212-0.
Anderson, J. A.; Mousset-Declas, C.; Williams, E. G.; Taylor, N. L. An in vitro chromosome doubling method for clovers (Trifolium spp.). Genome 34: 1–5; 1991.
Bouvier, L.; Guerif, P.; Djulbic, M.; Durel, C.-E.; Chevreau, E.; Lespinasse, Y. Chromosome doubling of pear haploid plants and homozygosity assessment using isozyme and microsatellite markers. Euphytica 123: 255–262; 2002 doi:10.1023/A:1014998019674.
Chakraborti, S. P.; Vijayan, K.; Roy, B. N.; Qadri, S. M. H. In vitro induction of tetraploidy in mulberry (Morus alba L.). Plant Cell Rep. 17: 799–803; 1998 doi:10.1007/s002990050486.
Chen, Y. Q.; Fan, J. Y.; Yi, F.; Luo, Z. X.; Fu, Y. S. Rapid clonal propagation of Dioscorea zingiberensis. Plant Cell Tissue and Organ Cult. 73: 75–80; 2003 doi:10.1023/A:1022683824635.
Ding, Z.; Zhou, L.; Wang, Y.; Tang, S. Factors influencing diosgenin content of Dioscorea zingiberensis. Chinese Trad. Herbal Drugs 12: 34–35; 1981.
Gao, S. L.; Zhu, D. N.; Cai, Z. H.; Xu, D. R. Autotetraploid plants from colchicine-treated bud culture of Salvia miltiorrhiza Bge. Plant Cell Tiss. Org. Cult. 47: 73–77; 1996.
Griesbach, R. J. Colchicine-induced polyploidy in Eustoma grandiflorum. Hort-Science 25: 1284–1286; 1990.
Hamill, S. D.; Smith, M. K.; Dodd, W. A. In vitro induction of banana autotetraploids by colchicine treatment of micropropagated diploids. Aust. J. Bot. 40: 887–896; 1992 doi:10.1071/BT9920887.
Huang, C. H.; Hang, Y. Y.; Zhou, Y. F.; Guo, K. Y.; Li, Z. Analysis on quality of some main populations of Dioscorea zingiberensis in China. Chemistry and Industry of Forest Products 23: 68–72; 2003.
Jackson, B. P.; Rowson, J. M. Alkaloid biogenesis in tetraploid Stramonium. J. Pharm. Pharmacol. 5: 778–793; 1953.
Li, H.; Wu, F. C.; Zheng, S. X. Preliminary study on polyploid of Dioscorea zingiberensis in vitro. Chinese Agricultural Science Bulletin 20: 33–35; 2004.
Mo, Y.; Lan, L. Q.; Qin, R. W.; Deng, Y.; Zhang, L. J.; Fu, H. L. Study on the germination terms of seeds of Dioscorea zingiberensis and its explant callus culture. Journal of Sichuan University (Natural Science Edition) 41: 837–841; 2004.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissues cultures. J. Plant. Physiol. 15: 473–479; 1962 doi:10.1111/j.1399-3054.1962.tb08052.x.
Orton, T. J.; Steidl, R. P. Cytogenetic analysis of plants regenerated from colchicine-treated callus cultures of an interspecific Hordeum hybrid. Theor. Appl. Genet. 57: 89–95; 1980 doi:10.1007/BF00745037.
Rowson, J. M. Increased alkaloid contents of induced polyploid of Datura. Nature 154: 81–82; 1949 doi:10.1038/154081b0.
Savikin-Fodulovic, K.; Grubisic, D.; Culafic, L.; Menkovic, N.; Ristic, M. Diosgenin and phytosterols content in five callus lines of Diocorea balcanica. Plant Science 135: 63–67; 1998 doi:10.1016/S0168-9452(98)00036-3.
Sikdar, A. K.; Jolly, M. S. Induced polyploidy in mulberry (Morus spp.): induction of tetraploids. Sericologia 34: 105–116; 1994.
Stanys, V.; Weckman, A.; Staniene, G.; Duchovskis, P. In vitro induction of polyploidy in Japanese quince (Chaenomeles japonica). Plant Cell Tissue and Organ Cult. 84: 263–268; 2006 doi:10.1007/s11240-005-9029-3.
Van Duren, M.; Morpurgo, R.; Dolezel, J.; Afza, R. Induction and verification of autotetraploids in diploid banana (Musa acuminata) by in vitro techniques. Euphytica 88: 25–34; 1996 doi:10.1007/BF00029262.
Viloria, Z.; Grosser, J. W.; Bracho, B. Immature embryo rescue, culture and seedling development of acid citrus fruit derived from interploid hybridization. Plant Cell Tissue and Organ Cult. 82: 159–167; 2005 doi:10.1007/s11240-005-0153-x.
Wang, Y. X.; Liu, H.; Bao, J. G.; Hong, Y.; Yang, Z. H.; Zhang, C. X. The saccharification-membrane retrieval-hydrolysis (SMRH) process: a novel approach for cleaner production of diosgenin derived from Dioscorea zingiberensis. J. Clean. Prod 16: 1133–1137; 2008 doi:10.1016/j.jclepro.2007.05.008.
Wang, Z. A.; Wang, R. Z. Selecting new species of Dioscorea zingiberensis contained high saponin by tissue culture. Chinese Traditional and Herbal Drugs 33: 361–363; 2002.
Xie, C. X.; Gao, S. L.; Zhu, D. N.; Qin, H. Z. Comparison of different methods of diosgenin extraction from Dioscorea zingiberensis. Journal of Plant Resources and Environment 14: 23–25; 2005.
Yan, L. X.; Hu, C. G.; Yao, J. L. Haploid callus and plantlets regenerated from anther culture of Dioscorea zingiberensis (Dioscoreaceae). Acta Botanica Yunnanica 29: 33–37; 2007.
Yan, Y. C.; Lin, H. H.; Dai, Q. L.; Huang, Q. Q. Studies on tissue culture and rapid propagation of Dioscorea zingiberensis. Journal of Sichuan University (Natural Science Edition) 39: 136–140; 2002.
Yuan, S.; Yan, Y.-C.; Lin, H.-H. Plant regeneration through somatic embryogenesis from callus cultures of Dioscorea zingiberensis. Plant Cell Tissue and Organ Cult. 80: 157–161; 2005 doi:10.1007/s11240-004-9543-8.
Zeng, S. H.; Chen, C. W.; Liu, H.; Liu, J. H.; Deng, X. X. In vitro induction, regeneration and analysis of autotetraploids derived from protoplasts and callus treated with colchicine in Citrus. Plant Cell Tissue Organ Cult. 87: 85–93; 2006 doi:10.1007/s11240-006-9142-y.
Zhang, J.; Zhang, M.; Deng, X. Obtaining autotetraploids in vitro at a high frequency in Citrus sinensis. Plant Cell Tissue Organ Cult. 89: 211–216; 2007 doi:10.1007/s11240-007-9240-5.
Acknowledgment
This study was carried out with the financial assistance of Science and Technology Bureau of Jiangsu, P.R. China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: A. Altman
Rights and permissions
About this article
Cite this article
Heping, H., Shanlin, G., Lanlan, C. et al. In vitro induction and identification of autotetraploids of Dioscorea zingiberensis . In Vitro Cell.Dev.Biol.-Plant 44, 448–455 (2008). https://doi.org/10.1007/s11627-008-9177-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-008-9177-3