Abstract
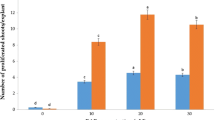
A method for in vitro regeneration of Searsia dentata from nodal and shoot tip explants derived from mature trees is outlined. Nodal explants produced multiple shoots from the axis when cultured on Murashige and Skoog (MS) medium containing 3% sucrose supplemented with 0, 5, 7.5, 10, or 12.5 μM N 6-benzyladenine (BA). An average of 5.3 shoots was obtained from nodal explants on 10 μM BA. For shoot tip explants, however, supplementation of α-naphthaleneacetic acid (NAA) with BA favored a caulogenic response. A maximum of 6.1 shoots were produced per shoot tip explant on MS containing 7.5 μM BA plus 5.0 μM NAA. The in vitro-regenerated shoots produced roots when transferred to full-strength MS medium containing 3% sucrose and 10 μM indole-3-butyric acid (IBA). The developed plantlets were transferred initially to a mist house. After an initial acclimatization period of 3–4 mo, plantlets were shifted to the greenhouse where they thrived for 9 mo. The standardized protocol for mass propagation of S. dentata should eliminate the dependence on natural stands of plants for traditional medicinal purposes, and will also serve as a means of conservation as the species is heavily overexploited.

Similar content being viewed by others
Abbreviations
- BA:
-
N 6-benzyladenine
- IAA:
-
indole-3-acetic acid
- IBA:
-
indole-3-butyric acid
- Kn:
-
kinetin
- MS:
-
Murashige and Skoog (1962)
References
Boggetti B.; Jasik J.; Mantell S. In vitro multiplication of cashew (Anacardium occidentale L.) using shoot node explants of glass house-raised plants. Plant Cell Rep 18: 456–461; 1999.
Bouza L.; Jacques M.; Miginac E. In vitro propagation of Paeonia suffruticosa Andr. Cv. “Mem de Varty”: developmental effects of exogenous hormones during the multiplication phases. Sci. Hortic 57: 241–255; 1994.
Collin H. A. Secondary product formation in plant tissue cultures. Plant Growth Regul 34: 119–134; 2001.
Ďurković J. Regeneration of Acer caudatifolium Hayata plantlets from juvenile explants. Plant Cell Rep 21: 1060–1064; 2003.
Kumar R.; Sharma K.; Agrawal V. In vitro clonal propagation of Holarrhena antidysenterica (L.) Wall. through nodal explants from mature trees. In Vitro Cell Dev. Biol. Plant 41: 137–144; 2005.
Lu M. C. Micropropagation of Vitis thunbergii Sieb. Et Zucc., a medicinal herb, through high frequency shoot tip culture. Sci. Hortic. 107: 64–69; 2005.
Mackay W. A.; Tipton J. L.; Thompson G. A. Micropropagation of Mexican redbud, Cercis canadensis var. mexicana. Plant Cell Tissue Organ Cult 43: 295–299; 1995.
Malik S. K.; Chaudhury R.; Kalia R. K. Rapid in vitro multiplication and conservation of Garcinia indica: A tropical medicinal tree species. Sci. Hortic 106: 539–553; 2005.
Massot B.; Milesi S.; Gontier E.; Bourgaud F.; Guckert A. Optimised culture conditions for the production of furanocoumarins by micropropagated shoots of Ruta graveolens. Plant Cell Tissue Organ Cult 62: 11–19; 2000.
Minitab Inc., PA 16801–3008, USA. 2003.
Murashige T.; Skoog F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant 15: 473–497; 1962.
Patra A.; Rai B.; Rout G. R.; Das P. Successful plant regeneration from callus cultures of Centella asiatica (Linn.) Urban. Plant Growth Regul 24: 13–16; 1998.
Purohit S. D.; Tak K.; Joshi P.; Samar D. Micropropagation of some woody tree species of Aravallis in Rajasthan. In: Srivastava P. S. (ed) Plant Tissue Culture and Molecular Biology—Application and Prospects. Narosa Publishing House, Delhi, pp 84–125; 1998.
Risa J.; Risa A.; Adsersen A.; Gauguin B.; Stafford G. I.; Van Staden J.; Jäger A. K. Screening of plants used in southern Africa for epilepsy and convulsions in GABAA-benzodiazepine receptor assay. J. Ethnopharmacol 93: 177–182; 2004.
Rout G. R. Micropropagation of Clitoria ternatea Linn. (Fabaceae)—an important medicinal plant. In Vitro Cell. Dev. Biol. Plant 41: 516–519; 2005.
Sansberro P.; Rey H.; Mroginski L.; Luna C. In vitro plantlet regeneration of Schinopsis balansae (Anacardiaceae). Trees: Structure and Function 17: 542–546; 2003.
Svenningsen A. B.; Madsen K. D.; Liljefors T.; Stafford G. I.; Van Staden J.; Jäger A. K. Bioflavones from Rhus species with affinity for the GABAA/benzodiazepine receptor. J. Ethnopharmacol 103: 276–280; 2006.
Tang D. Q.; Ishii K.; Ohba K. In vitro regeneration of Alnus cremastogyne Burk. from epicotyl explants. Plant Cell Rep 15: 658–661; 1996.
Vanisree M.; Lee C.-Y.; Lo S.-F.; Nalawade S. M.; Lin C. Y.; Tsay H.-S. Studies on the production of some important secondary metabolites from medicinal plants by plant tissue cultures. Bot. Bull. Acad. Sin 45: 1–22; 2004.
Acknowledgment
Financial assistance was provided by the National Research Foundation (NRF), South Africa.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Johannes van Staden
Rights and permissions
About this article
Cite this article
Prakash, S., Van Staden, J. Micropropagation of Searsia dentata . In Vitro Cell.Dev.Biol.-Plant 44, 338–341 (2008). https://doi.org/10.1007/s11627-008-9129-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-008-9129-y