Summary
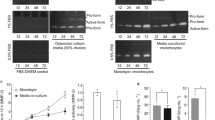
The objective of this study was to determine if an immortalized mammalian chondrocyte cell line had a profile of matrix metalloproteinase (MMP) expression that was consistent with what has been reported for primary chondrocytes in vitro and in vivo. A combination of zymography, Western, and Northern analysis was used to examine the expression of MMPs that are relevant to cartilage degradation. Both interleukin-1β and tumor necrosis factor α induced a 4- to 9-fold increase in the level of MMP-9 expression in conditioned media, and a 17- to 24-fold increase in MMP-3 mRNA. Other compounds such as basic fibroblast growth factor and staurosporine each increased MMP-9 expression individually and potentiated the effects of the two cytokines. Transforming growth factor β had no positive or inhibitory effects. N-methyl arginine blocked the increase in nitric oxide observed following treatment with the cytokines but did not prevent the increased expression of MMPs. The pattern of metalloproteinase expression observed in IRC cells and the response to cytokines is very similar to what has been reported during the pathogenesis of osteoarthritis. The IRC cells should be useful as a model system to study basic mechanisms controlling chondrocyte MMP expression and to identify pharmacological modulators of this process.
Similar content being viewed by others
References
Azzam, H. S.; Arand, G.; Lippman, E. W. Association of MMP-2 activation potential and metastatic progression in human breast cancer cell lines independent of MMP-2 production. J. Natl. Cancer Inst. 85:1758–1764; 1993.
Benya, P. D.; Padilla, S. R.; Nimni, M. E. Independent regulation of collagen types by chondrocytes during the loss of differentiated function in culture. Cell 15:1313; 1978.
Blanco, F. J.; Ochs, R. L.; Schwarz, H., et al. Chondrocyte apoptosis induced by nitric oxide. Am. J. Pathol. 146:75–85; 1995.
Chandrasekhar, S.; Harvey, A. K. Differential regulation of metalloprotease steady-state mRNA levels by Il-1 and FGF in rabbit articular chondrocytes. FEBS Letters 296:195–200; 1992.
Chandrasekhar, S.; Harvey, A. K.; Higginbotham, J. D., et al. Interleukin-1 induced suppression of type II collagen gene transcription involves DNA regulatory elements. Exp. Cell Res. 191:105–114; 1990.
Chatles, I. G.; Palmer, R. M.; Hickery, M. S., et al. Cloning characterization and expression of a cDNA encoding an inducible nitric oxide synthase from human chondrocytes. Proc. Natl. Acad. Sci. USA 90:11419–11423; 1993.
Chomczynski, P.; Sacchi, N. Single step isolation by acid guanidium thiocyanate-phenol chloroform extraction. Anal. Biochem. 162:156–159; 1987.
Crabbe, T.; O’Connell, J. P.; Smith, B. J., et al. Reciprocated matrix metalloproteinase activation; a process performed by interstitial collagenase and progelatinase A. Biochemistry 33:14419–14425; 1994.
Dean, D. D. Proteinase-mediated cartilage degradation in osteoarthritis. Semin. Arthritis Rheum. 20 (Suppl.):2–11; 1991.
DiBattista, J. A.; Martel-Pelletier, J.; Fujimoto, N., et al. Prostaglandins E2 and E1 inhibit cytokine-induced metalloprotease expression in human synovial fibroblasts. Lab. Invest. 71:270–278; 1994.
Fillmore, H. L.; Mainardi, C. L.; Hasty, K. A. Differentiation of PC12 cells with nerve growth factor is associated with induction of transin synthesis and release. J. Neurosci. Res. 31:662–669; 1992.
Fridman, R.; Bird, R. E.; Hoythya, M., et al. Expression of human recombinant 72 kDa gelatinase and TIMP-2: characterization of complex and free enzyme. Biochem. J. 289:411–416; 1993.
Ganu, V. S.; Hu, S. I.; Melton, R., et al. Biochemical and molecular characterization of stromelysin synthesized by human osteoarthritic chondrocytes stimulated with recombinant human interleukin-1. Clin. Exp. Rheumatol. 12:489–496; 1994.
Goldring, M. B.; Birkhead, J. R.; Suen, L.-F., et al. Interleukin-1 beta modulated gene expression in immortalized human chondrocytes. J. Clin. Invest. 94:2307–2416; 1994.
Green, L.; Wagner, D.; Glogowski, J., et al. Analysis of nitrate, nitrite and [15N] nitrate in biological fluids. Anal. Biochem. 126:131–138; 1982.
Harvey, A. K.; Stack, S. T.; Chandrasekhar, S. Differential modulation of degradative and repair responses of interleukin-1 treated chondrocytes by platelet derived growth factor. Biochem. J. 292:129–136; 1993.
Hasty, K. A.; Reife, R. A.; Kang, A. H., et al. The role of stromelysin in the cartilage destruction that accompanies inflammatory arthritis. Arthritis Rheum. 33:388–397; 1990.
Herron, G. S.; Banda, J. J.; Clark, E. J., et al. Secretion of metalloproteinases by stimulated capillary endothelial cells. II. Expression of collagenases and stromelysin activities is regulated by endogenous inhibitor. J. Biol. Chem. 261:2814–2818; 1986.
Horton, W. E., Jr.; Cleveland, J.; Rupp, V., et al. An established rat cell line expressing chondrocyte properties. Exp. Cell Res. 178:457–468; 1988.
Hulkower, K. I.; Georgescu, H. I.; Evans, C. H. Interleukin-1 and basic fibroblast growth factor are not mediated by protein kinase C. Biochem. J. 276:157–162; 1991.
Jones, B. E.; Moshyedi, P.; Gallo, S., et al. Characterization and novel activation of 72 kDa metalloproteinases of retinal interphotoreceptor matrix and Y-79 in culture medium. Exp. Eye Res. 59:257–269; 1994.
Kerr, L. D.; Miller, D. B.; Matrisian, L. M. TGF-beta-1 inhibition of transin/stromelysin gene expression is mediated through a FOS binding sequence. Cell 61:267–278; 1990.
Kohn, E. C.; Jacobs, W.; Kim, Y. S., et al. Calcium influx modulates expression of matrix metalloproteinase-2 (72 kDa type IV collagenase, gelatinase A). J. Biol. Chem. 269:21505–21511; 1994.
Kondo, S.; Ishiguro, N.; Iwata, H., et al. The effects of nitric oxide on chondrocytes and lymphocytes. Biochem. Biophys. Res. Commun. 197:1421–1437; 1993.
Koolwijk, P.; Miltenburg, A. M.; Van Erck, M. G., et al. Activated gelatinase B (MMP-9) and urokinase-type plasminogen activator in synovial fluids of patients with arthritis. Correlation with clinical and experimental variables of inflammation. J. Rheumatol. 22:385–393; 1995.
Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685; 1970.
Lees, M.; Taylor, D. J.; Woolley, D. E. Mast cell proteinases activate precursor forms of collagenase and stromelysin, but not of gelantinases A and B. Eur. J. Biochem. 223:171–177; 1994.
Mallein-Gerin, F.; Olsen, B. R. Expression of simian virus 40 large T (tumor) oncogene in mouse chondrocytes induces cell proliferation without loss of the differentiated phenotype. Proc. Natl. Acad. Sci. USA 90:3289–3293; 1993.
Manicourt, D. H.; Fujimoto, N.; Obata, K., et al. Serum levels of collagenase, stromelysin-1 and TIMP-1. Age and sex related differences in normal subjects and relationship to the extent of joint involvement. Arthritis Rheum. 37:1774–1783; 1994.
Martel-Pelletier, J.; McCollum, R.; Fujimoto, N., et al. Excess of metalloproteases over tissue inhibitor of metalloproteases may contribute to cartilage degradation in osteoarthritis and rheumatoid arthritis. Lab. Invest. 70:807–815; 1994.
Martel-Pelletier, J.; Pelletier, J. P.; Cloutier, J. M., et al. Neutral proteases capable of proteoglycan digesting activity in osteoarthritic and normal human articular cartilage. Arthritis Rheum. 27:305–312; 1984.
Matrisian, L. M. Metalloproteinases and their inhibitors in matrix remodeling. Trends Genet. 6:121–125; 1990.
Matrisian, L. M. The matrix degrading metalloproteinases. Bioassays 14:455–463; 1992.
Mitchell, P. G.; Cheung, H. S. Tumor necrosis factor alpha and epidermal growth factor regulation of collagenase and stromelysin in adult porcine articular chondrocytes. J. Cell. Physiol. 149:132–140; 1991.
Montai, M.; Smith, R. L.; Schurman, D. J., et al. Expression of a 92-kD type IV collegenase/gelatinase (gelatinase B) in osteoarthritic cartilage and its induction in normal human articular cartilage by interleukin-1. J. Clin. Invest. 92:179–185; 1993.
Murrell, G. A.; Jang, D.; Williams, R. J. Nitric oxide activates metalloproteinase enzymes in articular cartilage. Biochem. Biophys. Res. Commun. 206:15–21; 1995.
Nguyen, Q.; Mort, J. S.; Roughley, P. J. Preferential mRNA expression of prostromelysin relative to precollagenase and in situ localization in human articular cartilage. J. Clin. Invest. 89:1189–1197; 1992.
Ogata, Y.; Itoh, Y.; Nagase, H. Steps involved in activation of the promatrix metalloproteinase 9 (progelatinase B) tissue inhibitor of metalloproteinases-1 complex by 4-aminophenylmercuric acetate and proteinases. J. Biol. Chem. 270:18506–18511; 1995.
Overall, C. M. Repression of tissue inhibitor of matrix metalloproteinase expression by all-trans-retinoic acid in rat bone cell population: comparisons with transforming growth factor beta-1. J. Cell. Physiol. 164:17–25; 1995.
Oxford, J. T.; Doege, K.; Horton, W. E., Jr., et al. Characterization of type II and type XI collagen synthesis by an immortalized rat chondrocyte cell line (IRC) having a low level of type II collagen in RNA synthesis. Exp. Cell Res. 213:28–36; 1994.
Pelletier, J. P.; Martel-Pelletier, J.; Howell, D. S., et al. Collagenase and collagenolytic activity in human osteoarthritic cartilage. Arthritis Rheum. 26:63–68; 1983.
Pelletier, J. P.; Roughly, P.; DiBattista, J. A., et al. Are cytokines involved in osteoarthritic pathophysiology? Semin. Arthritis Rheum. 20 (Suppl.):12–25; 1991.
Pujol, J. P.; Galera, P.; Redini, F., et al. Role of cytokines in osteoarthritis: comparative effects of interleukin-1 and transforming growth factor beta on cultured rabbit chondrocytes. J. Rheumatol. 27 (Suppl.):76–79; 1991.
Sapolsky, A. I.; Keiser, H.; Howell, D. S., et al. Metalloproteases of human articular cartilage that digest cartilage proteoglycan at neutral and acid pH. J. Clin. Invest. 58:1030–1041; 1976.
Stefanovic-Racic, M.; Morales, T. I.; Taskiran, D., et al. The role of nitric oxide in proteoglycan turnover by bovine articular cartilage organ cultures. J. Immunol. 156:1213–1220; 1996.
Taskiran, D.; Stefanovic-Racic, M.; Georgescu, H., et al. Nitric oxide mediates suppression of cartilage proteoglycan synthesis by interleukin-1. Biochem. Biophys. Res. Commun. 200:142–148; 1994.
Tokuraku, M.; Sato, H.; Murakami, S., et al. Activation of the precursor of gelatinase A/72 kDa type IV collagenase/MMP-2 in lung carcinoma correlates with the expression of membrane type matrix metalloproteinase (MT-MMP) and with lymph node metastasis. Int. J. Cancer 64:355–359; 1995.
Vincenti, M. P.; Clark, I. M.; Brinckerhoff, C. E. Using inhibitors of metalloproteinases to treat arthritis. Arthritis Rheum. 37:1115–1126; 1994.
Werb, Z. Textbook of rheumatology. In: Kelly, W. N.; Harris, E., Jr.; Ruddy, S., et al., ed. 3rd ed. Philadelphia: W. B. Saunders Co.; 1989:300–309.
Zafarullah, M.; Su, S.; Martel-Pelletier, J., et al. Tissue inhibitor of metalloproteinase-2 (TIMP-2) mRNA is constitutively expressed in bovine, human normal, and osteoarthritic articular chondrocytes. J. Cell. Biochem. 60(2):211–217; 1996.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Horton, W.E., Udo, I., Precht, P. et al. Cytokine inducible matrix metalloproteinase expression in immortalized rat chondrocytes is independent of nitric oxide stimulation. In Vitro Cell.Dev.Biol.-Animal 34, 378–384 (1998). https://doi.org/10.1007/s11626-998-0019-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11626-998-0019-8