Summary
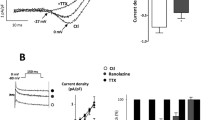
The present study attempted to test a novel hypothesis that Ca2+ sparks play an important role in arterial relaxation induced by tacrolimus. Recorded with confocal laser scanning microscopy, tacrolimus (10 µmol/L) increased the frequency of Ca2+ sparks, which could be reversed by ryanodine (10 µmol/L). Electrophysiological experiments revealed that tacrolimus (10 µmol/L) increased the large-conductance Ca2+-activated K+ currents (BKCa) in rat aortic vascular smooth muscle cells (AVSMCs), which could be blocked by ryanodine (10 µmol/L). Furthermore, tacrolimus (10 and 50 µmol/L) reduced the contractile force induced by norepinephrine (NE) or KCl in aortic vascular smooth muscle in a concentration-dependent manner, which could be also significantly attenuated by iberiotoxin (100 nmol/L) and ryanodine (10 µmol/L) respectively. In conclusion, tacrolimus could indirectly activate BKCa currents by increasing Ca2+ sparks released from ryanodine receptors, which inhibited the NE- or KCl-induced contraction in rat aorta.
Similar content being viewed by others
References
Marks AR. Cellular functions of immunophilins. Physiol Rev, 1996,76(3):631–649
Yasutsune T, Kawakami N, Hirano K, et al. Vasorelaxation and inhibition of the voltage-operated Ca2+ channels by FK506 in the porcine coronary artery. Br J Pharmacol, 1999,126(3):717–729
De Lima JJ, Xue H, Coburn L, et al. Effects of FK506 in rat and human resistance arteries. Kidney Int, 1999,55(4):1518–1527
Rosenbeck LL, Kiel PJ, Kalsekar I, et al. Prophylaxis with sirolimus and tacrolimus +/-antithymocyte globulin reduces the risk of acute graft-versus-host disease without an overall survival benefit following allogeneic stem cell transplantation. Biol Blood Marrow Transplant, 2011, 17(6):916–922
Gollasch M, Wellman GC, Knot HJ, et al. Ontogeny of local sarcoplasmic reticulum Ca2+ signals in cerebral arteries: Ca2+ sparks as elementary physiological events. Circ Res, 1998,83(11):1104–1114
Wellmanand, Nelson MT. Signaling between SR and plasmalemma in smooth muscle: sparks and the activation of Ca2+-sensitive ion channels. Cell Calcium, 2003,34(3):211–229
Song LS, Wang SQ, Xiao RP, et al. beta-Adrenergic stimulation synchronizes intracellular Ca(2+) release during excitation-contraction coupling in cardiac myocytes. Circ Res, 2001,88(8):794–801
Jaggar JH, Porter VA, Lederer WJ, et al. Calcium sparks in smooth muscle. Am J Physiol Cell Physiol, 2000,278(2):C235–256
Nelson MT, Cheng H, Rubart M, et al. Relaxation of arterial smooth muscle by calcium sparks. Science, 1995,270(5236):633–637
Knot HJ, Standen NB, Nelson MT. Ryanodine receptors regulate arterial diameter and wall [Ca2+] in cerebral arteries of rat via Ca2+-dependent K+ channels. J Physiol, 1998,508 (Pt 1):211–221
Wang YX, Zheng YM, Mei QB, et al. FKBP12.6 and cADPR regulation of Ca2+ release in smooth muscle cells. Am J Physiol Cell Physiol, 2004,286(3):C538–546
Dong L, Zheng YM, Van Riper D, et al. Functional and molecular evidence for impairment of calcium-activated potassium channels in type-1 diabetic cerebral artery smooth muscle cells. J Cereb Blood Flow Metab, 2008,28(2):377–386
Picht E, Zima AV, Blatter LA, et al. SparkMaster: automated calcium spark analysis with ImageJ. Am J Physiol Cell Physiol, 2007,293(3):C1073–1081
Novakovic A, Pavlovic M, Stojanovic I, et al. Different K+ channels are involved in relaxation of arterial and venous graft induced by nicorandil. J Cardiovasc Pharmacol, 2011,58(6):602–608
Beech DJ, Cheong A, Rusch NJ. Regulation of arterial tone by KV1 potassium channels. Circ Res, 2005,96(6):e58
Plane F, Johnson R, Kerr P, et al. Heteromultimeric Kv1 channels contribute to myogenic control of arterial diameter. Circ Res, 2005,96(2):216–224
Knotand HJ, Nelson MT. Regulation of arterial diameter and wall [Ca2+] in cerebral arteries of rat by membrane potential and intravascular pressure. J Physiol, 1998,508 (Pt 1):199–209
Benhamand CD, Bolton TB. Spontaneous transient outward currents in single visceral and vascular smooth muscle cells of the rabbit. J Physiol, 1986,381:385–406
Navarro-Antolin J, Levitsky KL, Calderon E, et al. Decreased expression of maxi-K+ channel beta1-subunit and altered vasoregulation in hypoxia. Circulation, 2005, 112(9):1309–1315
Tykocki NR, Thompson JM, Jackson WF, et al. Ryanodine receptors are uncoupled from contraction in rat vena cava. Cell Calcium, 2013,53(2):112–119
Vaithianathan T, Narayanan D, Asuncion-Chin MT, et al. Subtype identification and functional characterization of ryanodine receptors in rat cerebral artery myocytes. Am J Physiol Cell Physiol, 2010,299(2):C264–278
Abrenica B, Pierce GN, Gilchrist JS. Nucleoplasmic calcium regulation in rabbit aortic vascular smooth muscle cells. Can J Physiol Pharmacol, 2003,81(3):301–310
Su Z, Sugishita K, Li F, et al. Effects of FK506 on [Ca2+]i differ in mouse and rabbit ventricular myocytes. J Pharmacol Exp Ther, 2003,304(1):334–341
Ji G, Feldman ME, Greene KS, et al. RYR2 proteins contribute to the formation of Ca2+ sparks in smooth muscle. J Gen Physiol, 2004,123(4):377–386
Jackson-Weaver O, Osmond JM, Riddle MA, et al. Hydrogen sulfide dilates rat mesenteric arteries by activating endothelial large-conductance Ca2+-activated K+ channels and smooth muscle Ca2+ sparks. Am J Physiol Heart Circ Physiol, 2013,304(11):H1446–1454
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by the National Natural Science Foundation of China (No. 81102439).
Both authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, Yf., Wang, C., Zhang, R. et al. Tacrolimus inhibits vasoconstriction by increasing Ca2+ sparks in rat aorta. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 36, 8–13 (2016). https://doi.org/10.1007/s11596-016-1534-6
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-016-1534-6