Abstract
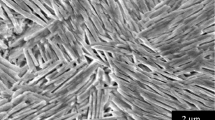
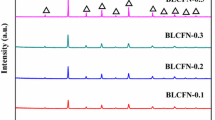
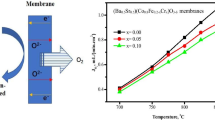
A stable and high oxygen permeable membrane system has been developed via partial replacement of iron with zirconium in SrCo0.8Fe0.2O3−δ compound. The initial powder series of composition SrCo0.8Fe0.2−yZryO3−δ (0 ≤ y ≤ 0.1), synthesized by an oxalate-based sol-gel route, is shown to exhibit a single perovskite-type cubic phase at 27 °C, transform to a brownmillerite (Ca2AlFeO5)-type orthorhombic structure, and finally assume a stable open cubic phase with disordered oxygen vacancies at temperatures determined by zirconium content, transitions being at 200 and 800 °C, respectively for the system y = 0.05. The highest JO2 of 2.54 × 10−6 mol cm−2 s−1 is realized in optimum composition of y = 0.05 at 1000 °C. The formation of cubic phase in Sr–Co–Fe–O system with abundance of disordered anion vacancies is crucial for both the high oxygen permeability and extended operational stability at elevated temperatures.














Similar content being viewed by others
References
Hashim SM, Mohamed AR, Bhatia S (2010) Current status of ceramic-based membranes for oxygen separation from air. Adv Colloid Interf Sci 160:88–100
Teraoka Y, Honbe Y, Ishii J, Furukawa H, Moriguchi I (2002) Catalytic effects in oxygen permeation through mixed-conductive LSCF perovskite membranes. Solid State Ionics 152–153:681–687
Bouwmeester HJM, Burggraaf AJ (1996) Fundamentals of inorganic membrane science and technology. Elsevier, Amsterdam Chapter 10
Hong WK, Choi GM (2010) Oxygen permeation of BSCF membrane with varying thickness and surface coating. J Membr Sci 346:353–360
Shao Z, Xiong G, Cong Y, Yang W (2000) Synthesis and oxygen permeation study of novel perovskite-type BaBixCo0.2Fe0.8−xO3−δceramic membranes. J Membr Sci 164:167–176
Vashook VV, Daroukh MA, Ullmann H (2001) Oxygen ion diffusion in perovskite-type oxides determined by permeation and by relaxation measurements. Ionics. 7:59–66
Zeng P, Shao Z, Liu S, Xu ZP (2009) Influence of M cations on structural, thermal and electrical properties of new oxygen selective membranes based on SrCo0.95M0.05O3−δperovskite. Sep Purif Technol 67:304–311
Li K (2007) Ceramic membrane for separation and reaction. John Wiley, England Chapter 6
Sunarso J, Baumann S, Serra JM et al (2008) Mixed ionic–electronic conducting (MIEC) ceramic-based membranes for oxygen separation. J Membr Sci 320:13–41
Teraoka Y, Nobunaga T, Yamazoe N (1988) Effect of cation substitution on the oxygen semipermeability of perovskite-type oxides. Chem Lett 3:503–506
Teraoka Y, Zhang HM, Furukawa S, Yamazoe N (1985) Oxygen permeation through perovskite-type oxides. Chem Lett:1743–1746
Xinfa D, Heng Z, Weiming L (2008) Preparation and characterization of a perovskite-type mixed conducting SrFe0.6Cu0.3Ti0.1O3−δmembrane for partial oxidation of methane to syngas. Chin J Chem Eng 16(3):411–415
Li Y, Zhao H, Xu N et al (2010) Systematic investigation on structure stability and oxygen permeability of Sr-doped BaCo0.7Fe0.2Nb0.1O3−δceramic membranes. J Membr Sci 362:460–470
Wang W, Cong Y, Yang W (2002) Oxygen permeation study in a tubular Ba0.5Sr0.5Co0.8Fe0.2O3-δ oxygen permeable membrane. J Membr Sci 210:259–271
Bouwmeester HJM, Kruidhof H, Burggraaf AJ (1994) Importance of the surface exchange kinetics as rate limiting step in oxygen permeation through mixed-conducting oxides. Solid State Ionics 72:185–194
McIntosh S, Vente JF, Haije WG, Blank DHA, Bouwmeester HJM (2006) Phase stability and oxygen non-stoichiometry of SrCo0.8Fe0.2O3−δ measured by in situ neutron diffraction. Solid State Ionics 177:833–842
Kruidhof H, Bouwmeester HJM, Doorn RHE, Burggraaf AJ (1993) Influence of order-disorder transitions on oxygen permeability through selected nonstoichiometric perovskite-type oxides. Solid State Ionics 63–65:816–822
Qiu L, Lee TH, Liu LM, Yang YL, Jacobson AJ (1995) Oxygen permeation studies of SrCo0.8Fe0.2O3-δ. Solid State Ionics 76:321–329
Fan CG, Deng ZQ, Zuo YB, Liu W, Chen CS (2004) Preparation and characterization of SrCo0.8Fe0.2O3–δ–SrSnO3 oxygen-permeable composite membrane. Solid State Ionics 166:339–342
Lu H, Cong Y, Yang W (2007) Oxygen permeability and improved stability of a permeable Zr-substituted perovskite membrane for air separation. Mater Sci Eng B 141:55–60
Liu H, Zhu Z, Cong Y, Zhang T, Yang W (2012) Remarkable dependence of electrochemical performance of SrCo0.8Fe0.2O3-δ on A-site on stoichiometry. Phys Chem Chem Phys 14:7234–7239
Yang L, Tan L, Gu X, Jin W, Zhang L, Xu N (2003) A new series of Sr(Co,Fe,Zr)O3−δperovskite-type membrane materials for oxygen permeation. Ind Eng Chem Res 42:2299–2305
Yang L, Gu X, Tan L, Zhang L, Wang C, Xu N (2003) Role of ZrO2 addition on oxygen transport and stability of ZrO2-promoted SrCo0.4Fe0.6O3–δ. Sep Purif Technol 32:301–306
Fan CG, Zuob YB, Li JT, Lu JQ, Chen CS, Bae DS (2007) Highly permeable La0.2Ba0.8Co0.8Fe0.2−xZrxO3−δmembranes for oxygen separation. Sep Purif Technol 55:35–39
Yao W, Cheng H, Zhao H et al (2016) Synthesis, oxygen permeability, and structural stability of BaCo0.7Fe0.3−xZrxO3−δ ceramic membranes. J Membr Sci 504:251–262
Li S, Jin W, Huang P et al (1999) Perovskite-related ZrO2 -doped SrCo0.4Fe0.6O3–δmembrane for oxygen permeation. AICHE J 45(2):276–284
Chen W, Zuo YB, Chen CS, Winnubst AJA (2010) Effect of Zr4+ doping on the oxygen stoichiometry and phase stability of SrCo0.8Fe0.2O3−δ oxygen separation membrane. Solid State Ionics 181:971–975
Lu H, Deng ZQ, Tong J, Yang W (2005) Oxygen permeability and structural stability of Zr-doped oxygen-permeable Ba0.5Sr0.5Co0.8Fe0.2O3–δ membrane. Mater Lett 59:2285–2288
Qi X, Lin YS, Swartz SL (2000) Electric transport and oxygen permeation properties of lanthanum cobaltite membranes synthesized by different methods. Ind Eng Chem Res 39:646–653
Martynczuk J, Arnold M, Feldhoff A (2008) Influence of grain size on the oxygen permeation performance of perovskite-type (Ba0.5Sr0.5)(Fe0.8Zn0.2)O3−δmembranes. J Membr Sci 322:375–382
Jaiswal SK, Kashyap VK, Kumar J (2012) On the sol–gel synthesis and characterization of strontium ferrite ceramic material. Mater Res Bull 47:692–699
Kashyap VK, Jaiswal SK, Kumar J (2016) On the structural stability and oxygen permeation behaviorof inorganic SrCo0.8Fe0.2O3−δ membranes. Ionics. 22:2471–2485
Kumar A, Kumar J (2008) On the synthesis and optical absorption studies of nano-size magnesium oxide powder. J Phys Chem Solids 69:2764–2772
Nagai T, Ito W, Sakon T (2007) Relationship between cation substitution and stability of perovskite structure in SrCoO3–δ-based mixed conductors. Solid State Ionics 177:3433–3444
Wu Z, Dong X, Jin W, Fan Y, Xu N (2007) A dense oxygen separation membrane deriving from nanosized mixed conducting oxide. J Membr Sci 291:172–179
Deng ZQ, Yang WS, Liu W, Chen CS (2006) Relationship between transport properties and phase transformations in mixed-conducting oxides. J Solid State Chem 179:362–369
He Y, Zhu X, Guo Z, Yang W (2010) Phase transitions in Sr1+xCo0.8Fe0.2O3−δ oxides. Mater Lett 64:1618–1621
Shin MJ, Yu JH (2012) Oxygen transport of A-site deficient Sr1−xFe0.5Co0.5O3−δ(x = 0–0.3) membranes. J Membr Sci 401–402:40–47
McIntosh S, Vente JF, Haije WG, Blank DHA, Bouwmeester HJM (2006) Structure and oxygen stoichiometry of SrCo0.8Fe0.2O3−δ andBa0.5Sr0.5Co0.8Fe0.2O3−δ. Solid State Ionics 177:1737–1742
Jaiswal SK, Kumar J (2011) On the sol–gel synthesis and structure, optical, magnetic and impedance behaviour of strontium cobaltite powder. J Alloys Compd 509:3859–3865
Didier C, Claridge J, Rosseinsky M (2014) Crystal structure of brownmillerite Ba2InGaO5. J Solid State Chem 218:38–43
Goodenough JB, Ruiz-Diaz JE, Zhen YS (1990) Oxide-ion conduction in Ba2In2O5 and Ba3In2MO8 (M=Ce, Hf, or Zr). Solid State Ionics 44:21–31
Young V, Otagawa T (1985) XPS studies on strontium compounds. Appl Surf Sci 20:228–248
Zhang HL, Wang DZ, Yang B, Huang NK (1997) XPS measurement for the elements in the interface between oxygen ion irradiatedZrO2 – Y2O3 films and iron substrate. Phys Status Solidi 160:145–150
Jiratova K, Mikulova J, Klempa J, Grygar T, Bastl Z, Kovanda F (2009) Modification of Co–Mn–Al mixed oxide with potassium and its effect on deep oxidation of VOC. Appl Catal A 361:106–116
Bu Y, Zhong Q, Xu D, Tan W (2013) Redox stability and sulfur resistance of Sm0.9Sr0.1CrxFe1–xO3–δ perovskite materials. J Alloys Compd 578:60–66
Dupin JC, Gonbeau D, Benqlilou-Moudden H, Vinatier P, Levasseur A (2001) XPS analysis of new lithium cobalt oxide thin-films before and after lithium deintercalation. Thin Solid Films 384:23–32
Norman C, Leach C (2011) In situ high temperature X-ray photoelectron spectroscopy study of barium strontium iron cobalt oxide. J Membr Sci 382:158–165
Grosvenor AP, Kobe BA, Biesinger MC, McIntyre NS (2004) Investigation of multiplet splitting of Fe 2p XPS spectra and bonding in iron compounds. Surf Interface Anal 36:1564–1574
Machocki A, Ioannides T, Stasinska B et al (2004) Manganese–lanthanum oxides modified with silverfor the catalytic combustionof methane. J Catal 227:282–296
Alaee MA, Movahednia MM, Mohammadi T (2009) Effect of Ba content on oxygen permeation performance of BaxSr1-xCo0.8Fe0.2O3-δ(x =0.2, 0.5, and 0.8) perovskite-type membrane. J Chem Eng Data 54:3082–3091
Hench LL, West JK (1990) The sol-gel process. Chem Rev 90:33–72
Steele BCH (1992) Oxygen ion conductors and their technological applications. Mater Sci Eng B13:79–87
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kashyap, V.K., Jaiswal, S.K. & Kumar, J. Effect of Zr4+on the phase stability and oxygen permeation characteristics of SrCo0.8Fe0.2−yZryO3−δ (y ≤ 0.1) membranes. Ionics 26, 1895–1911 (2020). https://doi.org/10.1007/s11581-019-03393-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-019-03393-8