Abstract
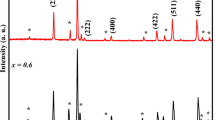
In this investigation, Mn3O4 spinel-type oxide was synthesized at low temperature using the Pechini process. We employed a sol-gel route, in which a solution of Mn(II) in a mixture of citric acid and ethylene glycol was heated to form a polymeric precursor, followed by annealing at lower temperature. The oxide obtained was identified by X-ray diffraction, scanning electron spectroscopy, and Raman spectroscopy. The results revealed that the formation of Mn3O4 hausmannite structure with a minor secondary phase of MnSO4 occurred at or above 280 °C. The sample powder consisted of fine grains with homogeneous morphology and an average size close to 1 μm was obtained. This new preparation procedure yielded an electrode oxide which appears to be a promising cathode material for fuel cells and metal-air batteries.









Similar content being viewed by others
References
Zang W, Yang Z, Liu Y, Tang S, Han X, Chen MJ (2004) Cryst Growth 263:394
Zhou G, Rong F, Xian-Hui C, Yi-Cheng W (2001) Inorg Chem Commun 4:294
Weixin Z, Cheng W, Xiaoming Z, Yi X, Yitai Q (1999) Solid State Ionics 117:331
Cimino S, Colonna S, De Rossi S, Faticanti M, Lisi L, Pettiti I, Porta P (2002) J Catal 205:309
Mao L, Zhang D, Sotomura T, Nakatsu K, Koshiba N, Ohaka T (2003) Electrochim Acta 48:1015
Ohsaka T, Mao L, Arihara K, Sotomura T (2004) Electrochem Commun 6:273
Cao YL, Yang HX, Ai XP, Xiao LF (2003) J Electroanal Chem 557:127
Klapste B, Vondrak J, Velicka J (2002) Electrochim Acta 47:2365
Yang J, Xu JJ (2003) Electrochem Commun 5:306
Keqiang D (2010) Russian Journal of Electrochemistry 46:461
Gorkenko OYA, Grabry IE, Anelichev VA, Bosak AA, Kavel AR, Guttler B, Svetchnikov VL, Zandbergen HW (2002) Solid State Communications 124:15–20
Teng F, Xu T, Liang S, Buergen G, Yao W, Zhu Y (2008) Catalysis Communications 9(6):1119–1124
Ardizzone S, Bianch CL, Tirelli D (1998) Colloids Surf A: Physicochem Eng Aspects 134:305
Pechini MP (1967) US Patent no. 3330676
El Baydi M, Poillerat G, Rehspringer JL, Gautier JL, Koenig JF, Chartier P (1994) J Solid State Chem 109:281
Chu CT, Dunn B (1987) J Am Ceram Soc C-375
Baythoun MSG, Sale FR (1982) J Mater Sci 17:2757
Kahoul A, Hammouche A (2010) Ionics 16:105–109
Olmos AV, Redón R, Gattorno GR, Zamora MEM, Leal F, Osorio Ana LF, Saniger JM (2005) J Coll Interf Sci 291:175–180
Boyero JM, Fernández EL, Gallardo-Amores JM, Ruano RC, Sánchez VE, Pérez EB (2001) Int J Inorg Mater 3:889
Escax V, Impéror-Clerc M, Bazin D, Davidson A (2005) C R Chimie 8:663–677
Yanga LX, Zhua YJ, Tonga H, Wanga WW, Cheng GF (2006) J Solid State Chemistry 179:1225–1229
Bernard MC, Goff AHL, Thi BV (1993) J Electrochem Soc 140:3065
Buciuman F, Patcas F, Craciun R, Zahn DRT (1999) Phys Chem Chem Phys 1:185
Kapteijn F, Van Langeveld AD, Moulijn JA, Andreini A, Vuurman MA, Turek AM, Jehng JM, Wachs IE (1994) J Catal 150
Hu CC, Wu YT, Chang KH (2008) Chem Mater 20(9):2890–2894
Rogulski Z, Siwek H, Paleska I, Czerwinski A (2003) J Electroanal Chem 543:175–185
Wu MQ, Snook GA, Chen GZ, Fray DG (2004) J Electrochem Commun 6:499–504
Messaoudi B, Joiret S, Keddam M, Takenouti H (2001) Electrochimica Acta 46:2487–2498
Hu CC, Liao SC, Chang KH, Yang YL, Lin KM (2010) J Power Sources 195(21):7259–7263
Wei ZD, Huang WZ, Zhang ST, Tan J (2000) J App Electrochem 30:1133–1136
Acknowledgment
The authors acknowledge the support of the Comité Mixte d’Evaluation et de Prospective de la Coopération Inter-universitaire Algéro-Française (CMEP) through Project No. 06 MDU 686.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nâamoune, F., Messaoudi, B., Kahoul, A. et al. A new sol-gel synthesis of Mn3O4 oxide and its electrochemical behavior in alkaline medium. Ionics 18, 365–370 (2012). https://doi.org/10.1007/s11581-011-0621-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-011-0621-8