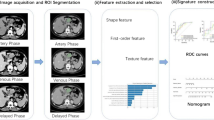
Abstract
Objective
Development and validation of a radiomics nomogram for predicting recurrence and adjuvant therapy benefit populations in high/intermediate-risk gastrointestinal stromal tumors (GISTs) based on computed tomography (CT) radiomic features.
Methods
Retrospectively collected from 2009.07 to 2015.09, 220 patients with pathological diagnosis of intermediate- and high-risk stratified gastrointestinal stromal tumors and received imatinib treatment were randomly divided into (6:4) training cohort and validation cohort. The 2D-tumor region of interest (ROI) was delineated from the portal-phase images on contrast-enhanced (CE) CT, and radiological features were extracted. The most valuable radiological features were obtained using a Lasso-Cox regression model. Integrated construction was conducted of nomograms of radiomics characteristics to predict recurrence-free survival (RFS) in patients receiving adjuvant therapy.
Results
Eight radiomic signatures were finally selected. The area under the curve (AUC) of the radiomics signature model for predicting 3-, 5-, and 7-year RFS in the training and validation cohorts (training cohort AUC = 0.80, 0.84, 0.76; validation cohort AUC = 0.78, 0.80, 0.76). The constructed radiomics nomogram was more accurate than the clinicopathological nomogram for predicting RFS in GIST (C-index: 0.864 95%CI, 0.817–0.911 vs. 0.733 95%CI, 0.675–0.791). Kaplan–Meier survival curve analysis showed a greater benefit from adjuvant therapy in patients with high radiomics scores (training cohort: p < 0.0001; validation cohort: p = 0.017), while there was no significant difference in the low-score group (p > 0.05).
Conclusion
In this study, a nomogram constructed based on preoperative CT radiomics features could be used for RFS prediction in high/intermediate-risk GISTs and assist the clinical decision-making for GIST patients.






Similar content being viewed by others
Data availability
The authors declare that they had full access to all of the data in this study and the authors take complete responsibility for the integrity of the data and the accuracy of the data analysis. Data are available for bona fide researchers who request it from the authors.
Abbreviations
- GIST(s):
-
Gastrointestinal stromal tumor(s)
- CT:
-
Computed tomography
- ROI:
-
Region of interest
- CE-CT:
-
Contrast-enhanced computed tomography
- LASSO-COX:
-
Least absolute shrinkage and selection operator-Cox regression model
- RFS:
-
Recurrence-free survival
- AUC:
-
Area under the curve
- ICC:
-
Intraclass correlation coefficient
- GLCM:
-
Gray-level co-occurrence matrix
- GLRLM:
-
Gray-level run length matrix
- GLSZM:
-
Gray-level size zone matrix
- GLDM:
-
Gray-level dependence matrix
- NGTDM:
-
Neighboring gray tone difference matrix
- Rad-score:
-
Radiomics score
- BMI:
-
Body mass index
- HR:
-
Hazard ratio
- CI:
-
Confidence interval
References
Joensuu H, Hohenberger P, Corless CL (2013) Gastrointestinal stromal tumour. Lancet 382(9896):973–983. https://doi.org/10.1016/S0140-6736(13)60106-3
Anderson WJ, Doyle LA (2021) Updates from the 2020 world health organization classification of soft tissue and bone tumours. Histopathology 78(5):644–657. https://doi.org/10.1111/his.14265
Gronchi A, Miah AB, Dei Tos AP, Abecassis N et al (2021) Soft tissue and visceral sarcomas: ESMO-EURACAN-GENTURIS clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 32(11):1348–1365. https://doi.org/10.1016/j.annonc.2021.07.006
Joensuu H, Eriksson M, Hall KS, Reichardt A, Hermes B, Schütte J, Reichardt P (2020) Survival outcomes associated with 3 years vs 1 year of adjuvant imatinib for patients with high-risk gastrointestinal stromal tumors: an analysis of a randomized clinical trial after 10-year follow-up. JAMA Oncol 6(8):1241–1246. https://doi.org/10.1001/jamaoncol.2020.2091
van der Graaf WTA, Tielen R, Bonenkamp JJ et al (2018) Nationwide trends in the incidence and outcome of patients with gastrointestinal stromal tumour in the imatinib era. Br J Surg 105(8):1020–1027. https://doi.org/10.1002/bjs.10809
Randall RL, Benjamin RS, Boles S et al (2018) Soft tissue sarcoma, version 1.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 16(5):536–563
Li J, Ye Y, Wang J et al (2017) Chinese consensus guidelines for diagnosis and management of gastrointestinal stromal tumor. Chin J Cancer Res. 29(4):281–293
Hølmebakk T, Wiedswang AM, Meza-Zepeda LA et al (2021) Integrating anatomical, molecular and clinical risk factors in gastrointestinal stromal tumor of the stomach. Ann Surg Oncol 28(11):6837–6845. https://doi.org/10.1245/s10434-021-09605-8
Nishida T, Hølmebakk T, Raut CP, Rutkowski P (2019) Defining tumor rupture in gastrointestinal stromal tumor. Ann Surg Oncol. 26(6):1669–1675. https://doi.org/10.1245/s10434-019-07297-9
Hølmebakk T, Hompland I, Bjerkehagen B et al (2018) Recurrence-free survival after resection of gastric gastrointestinal stromal tumors classified according to a strict definition of tumor rupture: a population-based study. Ann Surg Oncol 25(5):1133–1139. https://doi.org/10.1245/s10434-018-6353-5
Rutkowski P, Ziętek M, Cybulska-Stopa B et al (2021) The analysis of 3-year adjuvant therapy with imatinib in patients with high-risk molecular profiled gastrointestinal stromal tumors (GIST) treated in routine practice. Eur J Surg Oncol 47(5):1191–1195. https://doi.org/10.1016/j.ejso.2020.08.004
Li GZ, Raut CP (2019) Targeted therapy and personalized medicine in gastrointestinal stromal tumors: drug resistance, mechanisms, and treatment strategies. Onco Targets Ther 12:5123–5133. https://doi.org/10.2147/OTT.S180763
von Mehren M, Kane JM, Bui MM et al (2020) NCCN guidelines insights: soft tissue sarcoma, version 1.2021. J Natl Compr Canc Netw 18(12):1604–1612. https://doi.org/10.6004/jnccn.2020.0058
Patel DJ, Lutfi W, Sweigert P, Eguia E, Abood G, Knab L, Baker MS (2020) Adjuvant systemic therapy for intermediate and large gastric gastrointestinal stromal tumors (GISTs): Is there a survival benefit following margin negative surgical resection? Am J Surg 219(3):436–439
Joensuu H, Eriksson M, Hall KS, Hartmann JT, Pink D, Schütte J, Reichardt P (2012) One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA 307(12):1265–1272. https://doi.org/10.1001/jama.2012.347
Raut CP, Espat NJ, Maki RG, Araujo DM, Trent J, Williams TF, DeMatteo RP (2018) Efficacy and tolerability of 5-year adjuvant imatinib treatment for patients with resected intermediate-or high-risk primary gastrointestinal stromal tumor: the PERSIST-5 clinical trial. JAMA Oncol 4(12):e184060–e184060. https://doi.org/10.1001/jamaoncol.2018.4060
Autorino R, Gui B, Panza G et al (2022) Radiomics-based prediction of two-year clinical outcome in locally advanced cervical cancer patients undergoing neoadjuvant chemoradiotherapy. Radiol Med 127(5):498–506. https://doi.org/10.1007/s11547-022-01482-9
Thawani R, McLane M, Beig N et al (2018) Radiomics and radiogenomics in lung cancer: a review for the clinician. Lung Cancer 115:34–41. https://doi.org/10.1016/j.lungcan.2017.10.015
Chetan MR, Gleeson FV (2021) Radiomics in predicting treatment response in non-small-cell lung cancer: current status, challenges and future perspectives. Eur Radiol 31(2):1049–1058. https://doi.org/10.1007/s00330-020-07141-9
Park H, Lim Y, Ko ES et al (2018) Radiomics signature on magnetic resonance imaging: association with disease-free survival in patients with invasive breast cancer. Clin Cancer Res 24(19):4705–4714. https://doi.org/10.1158/1078-0432.CCR-17-3783
Jiang Y, Wang W, Chen C et al (2019) Radiomics signature on computed tomography imaging: association with lymph node metastasis in patients with gastric cancer. Front Oncol 9:340. https://doi.org/10.3389/fonc.2019.00340
Sun R, Limkin EJ, Vakalopoulou M et al (2018) A radiomics approach to assess tumour-infiltrating CD8 cells and response to anti-PD-1 or anti-PD-L1 immunotherapy: an imaging biomarker, retrospective multicohort study. Lancet Oncol 19(9):1180–1191. https://doi.org/10.1016/S1470-2045(18)30413-3
Dongsheng Gu, Yabin Hu, Ding H et al (2019) CT radiomics may predict the grade of pancreatic neuroendocrine tumors: a multicenter study. Eur Radiol 29(12):6880–6890. https://doi.org/10.1007/s00330-019-06176-x
Wang C, Li H, Jiaerken Y et al (2019) Building CT radiomics-based models for preoperatively predicting malignant potential and mitotic count of gastrointestinal stromal tumors. Transl Oncol 12(9):1229–1236. https://doi.org/10.1016/j.tranon.2019.06.005
Zhao Y, Feng M, Wang M et al (2021) CT Radiomics for the Preoperative Prediction of Ki67 Index in gastrointestinal stromal tumors: a multi-center study. Front Oncol 11:689136. https://doi.org/10.3389/fonc.2021.689136
Starmans MPA, Timbergen MJM, Vos M et al (2022) Differential diagnosis and molecular stratification of gastrointestinal stromal tumors on CT images using a radiomics approach. J Digit Imaging 35(2):127–136. https://doi.org/10.1007/s10278-022-00590-2
Zhang L, Kang L, Li G et al (2020) Computed tomography-based radiomics model for discriminating the risk stratification of gastrointestinal stromal tumors. Radiol Med 125(5):465–473. https://doi.org/10.1007/s11547-020-01138-6
Palatresi D, Fedeli F, Danti G et al (2022) Correlation of CT radiomic features for GISTs with pathological classification and molecular subtypes: preliminary and monocentric experience. Radiol Med 127(2):117–128. https://doi.org/10.1007/s11547-021-01446-5
Zhang QW, Gao YJ, Zhang RY, Zhou XX, Chen SL, Zhang Y, Ge ZZ (2020) Personalized CT-based radiomics nomogram preoperative predicting Ki-67 expression in gastrointestinal stromal tumors: a multicenter development and validation cohort. Clin Trans Med 9(1):1–11. https://doi.org/10.1186/s40169-020-0263-4
Xue C, Yuan J, Lo GG, Chang AT, Poon DM, Wong OL, Chu WC (2021) Radiomics feature reliability assessed by intraclass correlation coefficient: a systematic review. Quant Imaging Med Surg 11(10):4431
Zwanenburg A, Vallières M, Abdalah MA et al (2020) The image biomarker standardization initiative: standardized quantitative radiomics for high-throughput image-based phenotyping. Radiology 295(2):328–338. https://doi.org/10.1148/radiol.2020191145
van Griethuysen JJM, Fedorov A, Parmar C et al (2017) Computational radiomics system to decode the radiographic phenotype. Cancer Res 77(21):e104–e107. https://doi.org/10.1158/0008-5472
Lubner MG, Smith AD, Sandrasegaran K, Sahani DV, Pickhardt PJ (2017) CT texture analysis: definitions, applications, biologic correlates, and challenges. Radiographics 37(5):1483–1503. https://doi.org/10.1148/rg.2017170056
Mullah MAS, Hanley JA, Benedetti A (2021) LASSO type penalized spline regression for binary data. BMC Med Res Methodol 21(1):1–14. https://doi.org/10.1186/s12874-021-01234-9
Cheung LC, Pan Q, Hyun N, Katki HA (2019) Prioritized concordance index for hierarchical survival outcomes. Statist Med 38(15):2868–2882. https://doi.org/10.1002/sim.8157
Deniffel D, Abraham N, Namdar K et al (2020) Using decision curve analysis to benchmark performance of a magnetic resonance imaging-based deep learning model for prostate cancer risk assessment. Eur Radiol 30(12):6867–6876. https://doi.org/10.1007/s00330-020-07030-1
Nishida T, Sato S, Ozaka M et al (2022) Long-term adjuvant therapy for high-risk gastrointestinal stromal tumors in the real world. Gastric Cancer. https://doi.org/10.1007/s10120-022-01310-z
Kang S, Ryu M-H, Bang YH et al (2021) Adjuvant imatinib treatment for 5-years vs 3-years in patients with ruptured localized gastrointestinal stromal tumor: a retrospective analysis. Cancer Res Treat. https://doi.org/10.4143/crt.2021.1040
Li J, Ye Y, Wang J et al (2017) Chinese consensus guidelines for diagnosis and management of gastrointestinal stromal tumor. Chin J Cancer Res 29:281–293
Xin Wu, Li J, Wentong Xu et al (2018) Postoperative imatinib in patients with intermediate risk gastrointestinal stromal tumor. Future Oncol 14(17):1721–1729. https://doi.org/10.2217/fon-2017-0691
Lin Y, Wang M, Jia J et al (2020) Development and validation of a prognostic nomogram to predict recurrence in high-risk gastrointestinal stromal tumour: A retrospective analysis of two independent cohorts. EbioMedicine 60:103016
Jiang Y, Chen C, Xie J, Wang W, Zha X, Lv W, Li G (2018) Radiomics signature of computed tomography imaging for prediction of survival and chemotherapeutic benefits in gastric cancer. EbioMedicine 36:171–182. https://doi.org/10.1016/j.ebiom.2018.09.007
Choe J, Lee SM, Do KH et al (2020) Outcome prediction in resectable lung adenocarcinoma patients: value of CT radiomics. Eur Radiol 30(9):4952–4963. https://doi.org/10.1007/s00330-020-06872-z
Fan S, Cui X, Liu C et al (2021) CT-based radiomics signature: a potential biomarker for predicting postoperative recurrence risk in stage ii colorectal cancer. Front Oncol 19(11):644933. https://doi.org/10.3389/fonc.2021.644933
Park S, Sham JG, Kawamoto S et al (2021) CT radiomics-based preoperative survival prediction in patients with pancreatic ductal adenocarcinoma. AJR Am J Roentgenol 217(5):1104–1112. https://doi.org/10.2214/AJR.20.23490
Tang S, Jing Ou, Liu J et al (2021) Application of contrast-enhanced CT radiomics in prediction of early recurrence of locally advanced oesophageal squamous cell carcinoma after trimodal therapy. Cancer Imaging 21(1):38. https://doi.org/10.1186/s40644-021-00407-5
Chen T, Lili Xu, Ye L et al (2019) A new nomogram for recurrence-free survival prediction of gastrointestinal stromal tumors: comparison with current risk classification methods. Eur J Surg Oncol 45(6):1109–1114. https://doi.org/10.1016/j.ejso.2018.12.014
Lin Y, Wang M, Jia J et al (2020) Development and validation of a prognostic nomogram to predict recurrence in high-risk gastrointestinal stromal tumour: a retrospective analysis of two independent cohorts. EbioMedicine 60:103016. https://doi.org/10.1016/j.ebiom.2020.103016
Cao X, Cui J, Yu T, Li Z, Zhao G (2020) Fibrinogen/albumin ratio index is an independent prognosis predictor of recurrence-free survival in patients after surgical resection of gastrointestinal stromal tumors. Front Oncol 10:1459. https://doi.org/10.3389/fonc.2020.01459
Acknowledgements
We thank all patients, their families, and all investigators involved in the present study. At the same time, I would like to thank my lover Ling Dan.
Funding
This research was funded by Construction Project of Fujian Province Minimally Invasive Medical Center (No. [2021] 662).
Author information
Authors and Affiliations
Contributions
J.-W.X. contributed to conceptualization; H.-L.Z. was involved in data curation; F.-H.W. contributed to formal analysis; C.-M.H. was involved in funding acquisition; J.-T.L. contributed to investigation; F.-H.W. and J.-W.X. were involved in methodology; P.L. contributed to project administration; P.L. and C.-M.H. were involved in resources; F.-H.W. and J.-T.L. provided software; H.-L.Z. and Q.-Y.C. were involved in validation; C.-H.Z. contributed to visualization; F.-H.W. and H.-L.Z. were involved in writing—original draft; and Q.-Y.C., C.-M.H., and J.-W.X contributed to writing—review and editing.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Informed consent statement
This study is a retrospective study, and patients’ informed consent was waived with the approval of the Institutional Review Board.
Institutional review board statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, FH., Zheng, HL., Li, JT. et al. Prediction of recurrence-free survival and adjuvant therapy benefit in patients with gastrointestinal stromal tumors based on radiomics features. Radiol med 127, 1085–1097 (2022). https://doi.org/10.1007/s11547-022-01549-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-022-01549-7