Abstract
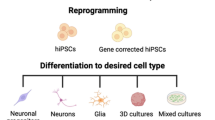
Cellular reprogramming allows for the de novo generation of human neurons and glial cells from patients with neurological and psychiatric disorders. Crucially, this technology preserves the genome of the donor individual and thus provides a unique opportunity for systematic investigation of genetic influences on neuronal pathophysiology. Although direct reprogramming of adult somatic cells to neurons is now possible, the majority of recent studies have used induced pluripotent stem cells (iPSCs) derived from patient fibroblasts to generate neural progenitors that can be differentiated to specific neural cell types. Investigations of monogenic diseases have established proof-of-principle for many aspects of cellular disease modeling, including targeted differentiation of neuronal populations and rescue of phenotypes in patient iPSC lines. Refinement of protocols to allow for efficient generation of iPSC lines from large patient cohorts may reveal common functional pathology and genetic interactions in diseases with a polygenic basis.We review several recent studies that illustrate the utility of iPSC-based cellular models of neurodevelopmental and neurodegenerative disorders to identify novel phenotypes and therapeutic approaches.
Similar content being viewed by others
References
Ambasudhan R, Talantova M, Coleman R, Yuan X, Zhu S, Lipton S A, Ding S (2011). Direct reprogramming of adult human fibroblasts to functional neurons under defined conditions. Cell Stem Cell, 9(2): 113–118
Amir R E, Van den Veyver I B, Wan M, Tran C Q, Francke U, Zoghbi H Y (1999). Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet, 23(2): 185–188
Anderson S L, Qiu J, Rubin B Y (2003a). EGCG corrects aberrant splicing of IKAP mRNA in cells from patients with familial dysautonomia. Biochem Biophys Res Commun, 310(2): 627–633
Anderson S L, Qiu J, Rubin B Y (2003b). Tocotrienols induce IKBKAP expression: a possible therapy for familial dysautonomia. Biochem Biophys Res Commun, 306(1): 303–309
Bock C, Kiskinis E, Verstappen G, Gu H, Boulting G, Smith Z D, Ziller M, Croft G F, Amoroso M W, Oakley D H, Gnirke A, Eggan K, Meissner A (2011). Reference Maps of human ES and iPS cell variation enable high-throughput characterization of pluripotent cell lines. Cell, 144(3): 439–452
Boulting G L, Kiskinis E, Croft G F, Amoroso M W, Oakley D H, Wainger B J, Williams D J, Kahler D J, Yamaki M, Davidow L, Rodolfa C T, Dimos J T, Mikkilineni S, MacDermott A B, Woolf C J, Henderson C E, Wichterle H, Eggan K (2011). A functionally characterized test set of human induced pluripotent stem cells. Nat Biotechnol, 29(3): 279–286
Brennand K J, Simone A, Jou J, Gelboin-Burkhart C, Tran N, Sangar S, Li Y, Mu Y, Chen G, Yu D, McCarthy S, Sebat J, Gage F H (2011). Modelling schizophrenia using human induced pluripotent stem cells. Nature, 473(7346): 221–225
Caiazzo M, Dell’Anno M T, Dvoretskova E, Lazarevic D, Taverna S, Leo D, Sotnikova T D, Menegon A, Roncaglia P, Colciago G, Russo G, Carninci P, Pezzoli G, Gainetdinov R R, Gustincich S, Dityatev A, Broccoli V (2011). Direct generation of functional dopaminergic neurons from mouse and human fibroblasts. Nature, 476(7359): 224–227
Chambers S M, Studer L (2011). Cell fate plug and play: direct reprogramming and induced pluripotency. Cell, 145(6): 827–830
Cheung A Y, Horvath L M, Grafodatskaya D, Pasceri P, Weksberg R, Hotta A, Carrel L, Ellis J (2011). Isolation of MECP2-null Rett Syndrome patient hiPS cells and isogenic controls through Xchromosome inactivation. Hum Mol Genet, 20(11): 2103–2115
Chiang C H, Su Y, Wen Z, Yoritomo N, Ross C A, Margolis R L, Song H, Ming G L (2011). Integration-free induced pluripotent stem cells derived from schizophrenia patients with a DISC1 mutation. Mol Psychiatry, 16(4): 358–360
Duan X, Chang J H, Ge S, Faulkner R L, Kim J Y, Kitabatake Y, Liu X B, Yang C H, Jordan J D, Ma D K, Liu C Y, Ganesan S, Cheng H J, Ming G L, Lu B, Song H (2007). Disrupted-In-Schizophrenia 1 regulates integration of newly generated neurons in the adult brain. Cell, 130(6): 1146–1158
Falk A, Koch P, Kesavan J, Takashima Y, Ladewig J, Alexander M, Wiskow O, Tailor J, Trotter M, Pollard S, Smith A, Brüstle O (2012). Capture of neuroepithelial-like stem cells from pluripotent stem cells provides a versatile system for in vitro production of human neurons. PLoS ONE, 7(1): e29597
Faulkner R L, Jang M H, Liu X B, Duan X, Sailor K A, Kim J Y, Ge S, Jones E G, Ming G L, Song H, Cheng H J (2008). Development of hippocampal mossy fiber synaptic outputs by new neurons in the adult brain. Proc Natl Acad Sci USA, 105(37): 14157–14162
Gore A, Li Z, Fung H L, Young J E, Agarwal S, Antosiewicz-Bourget J, Canto I, Giorgetti A, Israel M A, Kiskinis E, Lee J H, Loh Y H, Manos P D, Montserrat N, Panopoulos A D, Ruiz S, Wilbert M L, Yu J, Kirkness E F, Izpisua Belmonte J C, Rossi D J, Thomson J A, Eggan K, Daley G Q, Goldstein L S, Zhang K (2011). Somatic coding mutations in human induced pluripotent stem cells. Nature, 471(7336): 63–67
Hansen D V, Rubenstein J L, Kriegstein A R (2011). Deriving excitatory neurons of the neocortex from pluripotent stem cells. Neuron, 70(4): 645–660
Harrison P J, Weinberger D R (2005). Schizophrenia genes, gene expression, and neuropathology: on the matter of their convergence. Mol Psychiatry, 10:40–68
Herbert M R (2010). Contributions of the environment and environmentally vulnerable physiology to autism spectrum disorders. Curr Opin Neurol, 23(2): 103–110
Hussein SM, Batada N N, Vuoristo S, Ching RW, Autio R, Närvä E, Ng S, Sourour M, Hämäläinen R, Olsson C, Lundin K, Mikkola M, Trokovic R, Peitz M, Brüstle O, Bazett-Jones D P, Alitalo K, Lahesmaa R, Nagy A, Otonkoski T (2011). Copy number variation and selection during reprogramming to pluripotency. Nature, 471(7336): 58–62
Israel M A, Yuan S H, Bardy C, Reyna S M, Mu Y, Herrera C, Hefferan M P, Van Gorp S, Nazor K L, Boscolo F S, Carson C T, Laurent L C, Marsala M, Gage F H, Remes A M, Koo E H, Goldstein L S (2012). Probing sporadic and familial Alzheimer’s disease using induced pluripotent stem cells. Nature, 482(7384): 216–220
Jiang H, Ren Y, Yuen E Y, Zhong P, Ghaedi M, Hu Z, Azabdaftari G, Nakaso K, Yan Z, Feng J (2012). Parkin controls dopamine utilization in human midbrain dopaminergic neurons derived from induced pluripotent stem cells. Nat Commun, 3: 668
Juopperi T A, Song H, Ming G L (2011). Modeling neurological diseases using patient-derived induced pluripotent stem cells. Future Neurol, 6(3): 363–373
Keller F, Persico A M (2003). The neurobiological context of autism. Mol Neurobiol 28(1): 1–22
Kim J Y, Duan X, Liu C Y, Jang M H, Guo J U, Pow-anpongkul N, Kang E, Song H, Ming G L (2009). DISC1 regulates new neuron development in the adult brain via modulation of AKT-mTOR signaling through KIAA1212. Neuron, 63(6): 761–773
Kim K Y, Hysolli E, Park I H (2011). Neuronal maturation defect in induced pluripotent stem cells from patients with Rett syndrome. Proc Natl Acad Sci USA, 108(34): 14169–14174
Koch P, Opitz T, Steinbeck J A, Ladewig J, Brüstle O (2009). A rosettetype, self-renewing human ES cell-derived neural stem cell with potential for in vitro instruction and synaptic integration. Proc Natl Acad Sci USA, 106(9): 3225–3230
Krencik R, Weick J P, Liu Y, Zhang Z J, Zhang S C (2011). Specification of transplantable astroglial subtypes from human pluripotent stem cells. Nat Biotechnol, 29(6): 528–534
Lee G, Papapetrou E P, Kim H, Chambers S M, Tomishima M J, Fasano C A, Ganat YM, Menon J, Shimizu F, Viale A, Tabar V, Sadelain M, Studer L (2009). Modelling pathogenesis and treatment of familial dysautonomia using patient-specific iPSCs. Nature, 461(7262): 402–406
Lister R, Pelizzola M, Kida Y S, Hawkins R D, Nery J R, Hon G, Antosiewicz-Bourget J, O’Malley R, Castanon R, Klugman S, Downes M, Yu R, Stewart R, Ren B, Thomson J A, Evans R M, Ecker J R (2011). Hotspots of aberrant epigenomic reprogramming in human induced pluripotent stem cells. Nature, 471(7336): 68–73
Mao Y, Ge X, Frank C L, Madison J M, Koehler A N, Doud M K, Tassa C, Berry E M, Soda T, Singh K K, Biechele T, Petryshen T L, Moon R T, Haggarty S J, Tsai L H (2009). Disrupted in schizophrenia 1 regulates neuronal progenitor proliferation via modulation of GSK3beta/beta-catenin signaling. Cell, 136(6): 1017–1031
Martin I, Dawson V L, Dawson T M (2011). Recent advances in the genetics of Parkinson’s disease. Annu Rev Genomics Hum Genet, 12(1): 301–325
Marchetto M C, Carromeu C, Acab A, Yu D, Yeo G W, Mu Y, Chen G, Gage F H, Muotri A R (2010). A model for neural development and treatment of Rett syndrome using human induced pluripotent stem cells. Cell, 143(4): 527–39
Millar J K, Wilson-Annan J C, Anderson S, Christie S, Taylor M S, Semple C A, Devon R S, St Clair D M, Muir W J, Blackwood D H, Porteous D J (2000). Disruption of two novel genes by a translocation co-segregating with schizophrenia. Hum Mol Genet, 9(9): 1415–1423
Nguyen H N, Byers B, Cord B, Shcheglovitov A, Byrne J, Gujar P, Kee K, Schüle B, Dolmetsch R E, Langston W, Palmer T D, Pera R R (2011). LRRK2 mutant iPSC-derived DA neurons demonstrate increased susceptibility to oxidative stress. Cell Stem Cell, 8(3): 267–280
Pang Z P, Yang N, Vierbuchen T, Ostermeier A, Fuentes D R, Yang T Q, Citri A, Sebastiano V, Marro S, Südhof T C, Wernig M (2011). Induction of human neuronal cells by defined transcription factors. Nature, 476(7359): 220–223
Park I H, Zhao R, West J A, Yabuuchi A, Huo H, Ince T A, Lerou P H, Lensch M W, Daley G Q (2008). Reprogramming of human somatic cells to pluripotency with defined factors. Nature, 451(7175): 141–146
Paşca S P, Portmann T, Voineagu I, Yazawa M, Shcheglovitov A, Paşca A M, Cord B, Palmer T D, Chikahisa S, Nishino S, Bernstein J A, Hallmayer J, Geschwind D H, Dolmetsch R E (2011). Using iPSC-derived neurons to uncover cellular phenotypes associated with Timothy syndrome. Nat Med, 17(12): 1657–1662
Pfisterer U, Kirkeby A, Torper O, Wood J, Nelander J, Dufour A, Björklund A, Lindvall O, Jakobsson J, Parmar M (2011). Direct conversion of human fibroblasts to dopaminergic neurons. Proc Natl Acad Sci USA, 108(25): 10343–10348
Pomp O, Dreesen O, Leong D F, Meller-Pomp O, Tan T T, Zhou F, Colman A (2011). Unexpected X chromosome skewing during culture and reprogramming of human somatic cells can be alleviated by exogenous telomerase. Cell Stem Cell, 9(2): 156–165
Qiang L, Fujita R, Yamashita T, Angulo S, Rhinn H, Rhee D, Doege C, Chau L, Aubry L, Vanti W B, Moreno H, Abeliovich A (2011). Directed conversion of Alzheimer’s disease patient skin fibroblasts into functional neurons. Cell, 146(3): 359–371
Ross C A, Margolis R L, Reading S A, Pletnikov M, Coyle J T (2006). Neurobiology of schizophrenia. Neuron, 52(1): 139–153
Sachs N A, Sawa A, Holmes S E, Ross C A, DeLisi L E, Margolis R L (2005). A frameshift mutation in Disrupted in Schizophrenia 1 in an American family with schizophrenia and schizoaffective disorder. Mol Psychiatry, 10(8): 758–764
Seibler P, Graziotto J, Jeong H, Simunovic F, Klein C, Krainc D (2011). Mitochondrial Parkin recruitment is impaired in neurons derived from mutant PINK1 induced pluripotent stem cells. J Neurosci, 31(16): 5970–5976
Shi Y, Kirwan P, Smith J, Robinson H P, Livesey F J (2012). Human cerebral cortex development from pluripotent stem cells to functional excitatory synapses. Nat Neurosci, 15(3): 477–486
Slaugenhaupt S A, Blumenfeld A, Gill S P, Leyne M, Mull J, Cuajungco M P, Liebert C B, Chadwick B, Idelson M, Reznik L, Robbins C, Makalowska I, Brownstein M, Krappmann D, Scheidereit C, Maayan C, Axelrod F B, Gusella J F (2001). Tissue-specific expression of a splicing mutation in the IKBKAP gene causes familial dysautonomia. Am J Hum Genet, 68(3): 598–605
Slaugenhaupt S A, Mull J, Leyne M, Cuajungco M P, Gill S P, Hims M M, Quintero F, Axelrod F B, Gusella J F (2003). Rescue of a human mRNA splicing defect by the plant cytokinin kinetin. Hum Mol Genet, 13(4): 429–436
Soldner F, Laganière J, Cheng A W, Hockemeyer D, Gao Q, Alagappan R, Khurana V, Golbe L I, Myers R H, Lindquist S, Zhang L, Guschin D, Fong L K, Vu B J, Meng X, Urnov F D, Rebar E J, Gregory P D, Zhang H S, Jaenisch R (2011). Generation of isogenic pluripotent stem cells differing exclusively at two early onset Parkinson point mutations. Cell, 146(2): 318–331
Spitzer N C (2006). Electrical activity in early neuronal development. Nature, 444(7120): 707–712
St Clair D, Blackwood D, Muir W, Carothers A, Walker M, Spowart G, Gosden C, Evans H J (1990). Association within a family of a balanced autosomal translocation with major mental illness. Lancet, 336(8706): 13–16
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S (2007). Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell, 131(5): 861–872
Takahashi K, Yamanaka S (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126(4): 663–676
Tchieu J, Kuoy E, Chin M H, Trinh H, Patterson M, Sherman S P, Aimiuwu O, Lindgren A, Hakimian S, Zack J A, Clark A T, Pyle A D, Lowry W E, Plath K (2010). Female human iPSCs retain an inactive X chromosome. Cell Stem Cell, 7(3): 329–342
Tropea D, Giacometti E, Wilson N R, Beard C, McCurry C, Fu D D, Flannery R, Jaenisch R, Sur M (2009). Partial reversal of Rett Syndrome-like symptoms in MeCP2 mutant mice. Proc Natl Acad Sci USA, 106(6): 2029–2034
Uhlhaas P J, Singer W (2010). Abnormal neural oscillations and synchrony in schizophrenia. Nat Rev Neurosci, 11(2): 100–113
Vierbuchen T, Ostermeier A, Pang Z P, Kokubu Y, Südhof T C, Wernig M (2010). Direct conversion of fibroblasts to functional neurons by defined factors. Nature, 463(7284): 1035–1041
Weinberger D R (1987). Implications of normal brain development for the pathogenesis of schizophrenia. Arch Gen Psychiatry, 44(7): 660–669
Yoo A S, Sun A X, Li L, Shcheglovitov A, Portmann T, Li Y, Lee-Messer C, Dolmetsch R E, Tsien R W, Crabtree G R (2011). MicroRNA-mediated conversion of human fibroblasts to neurons. Nature, 476(7359): 228–231
Yu J, Vodyanik M A, SmugaOtto K, Antosiewicz-Bourget J, Frane J L, Tian S, Nie J, Jonsdottir G A, Ruotti V, Stewart R, Slukvin I I, Thomson J A (2007). Induced pluripotent stem cell lines derived from human somatic cells. Science, 318(5858): 1917–1920
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Christian, K.M., Song, H. & Ming, Gl. Application of reprogrammed patient cells to investigate the etiology of neurological and psychiatric disorders. Front. Biol. 7, 179–188 (2012). https://doi.org/10.1007/s11515-012-1216-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11515-012-1216-0