Abstract
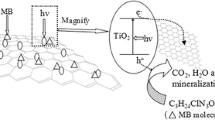
TiO2-Graphene Oxide intercalated composite (TiO2-Graphene Oxide) has been successfully prepared at low temperature (80°C) with graphite oxide (GO) and titanium sulfate (Ti(SO4)2) as initial reactants. GO was firstly exfoliated by NaOH and formed single and multi-layered graphite oxide mixture which can be defined as graphene oxide, [TiO]2+ induced by the hydrolysis of Ti(SO4)2 diffused into graphene oxide interlayer by electrostatic attraction. The nucleation and growth of TiO2 crystallites took place at low temperature and TiO2-Graphene Oxide composite was successfully synthesized. Furthermore, the photocatalytic properties of TiO2-Graphene Oxide under the irradiation of UV light were also studied. The results show that the degradation rate of methyl orange is 1.16 mg min−1 g−1(refer to the efficiency of the initial 15 min). Compared with P25 powder, this kind of intercalation composite owns much better efficiency. On the other hand, the reusable properties and stable properties of TiO2-Graphene Oxide intercalated composite are also discussed in this paper. At last, crystalline structure, interface status, thermal properties and microscopic structure of TiO2-Graphene Oxide were characterized by X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), thermogravimetric analysis (TGA), field emission scanning electron microscopy (FESEM) and high-resolution Transmission Electron Microscopy (HRTEM). Also, we have analyzed major influencing factors and mechanism of the composite structures which evidently improve the photocatalytic properties.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hoffmann M R, Martin S T, Choi W, et al. Environmental applications of semiconductor photocatalysis. Chem Rev, 1995, 95: 69–96
Li X Z, Liu H, Cheng L F, et al. Photocatalytic oxidation using a new catalyst-TiO2 microsphere-for water and wastewater treatment. Environ Sci Technol, 2003, 37: 3989–3994
Adachi M, Murata Y, Takao J, et al. Highly efficient dyesensitized solar cells with a titania thin-film electrode composed of a network structure of single-crystal-like TiO2 nanowires made by the “oriented attachment” mechanism. J Am Chem Soc, 2004, 126: 14943–14949
Zakrzewska K. Mixed oxides as gas sensors. Thin Solid Films, 2001, 391: 229–238
Kalyanasundaram K, Grätzel M. Applications of functionalized transition metal complexes in photonic and optoelectronic devices. Coord Chem Rev, 1998, 177: 347–414
Yin S, Hasegawa H, Maeda D, et al. Synthesis of visible-light-active nanosize rutile titania photocatalyst by low temperature dissolution-reprecipitation process. J Photochem Photobiol A Chem, 2004, 163: 1–8
Li Y T, Sun X G, Li H W, et al. Preparation of anatase TiO2 nanoparticles with high thermal stability and specific surface area by alcohothermal method. Powder Tech, 2009, 194: 149–152
Minero C, Vione D. A quantitative evalution of the photocatalytic performance of TiO2 slurries. Appl Catal B: Environ, 2006, 67: 257–269
Gao Y, Liu H T, Ma M J. Preparation and photocatalytic behavior of TiO2-carbon nanotube hybrid catalyst for acridine dye decomposition. React Kinet Catal Lett, 2007, 90: 11–18
Gao B, Chen G Z, Li Puma G. Carbon nanotubes/titanum dioxide (CNTs/TiO2) nanocomposites prepared by conventional and novel surfactant wrapping sol-gel methods exhibiting enhanced photocatalytic activity. Appl Catal B: Environ, 2009, 89: 503–509
Oh W C, Jung A R, Ko W B. Characterization and relative photonic efficiencies of a new nanocarbon/TiO2 composite photocatalyst designed for organic dye decomposition and bactericidal activity. Mater Sci Engin C, 2009, 29: 1338–1347
Gao B, Peng C, Chen G Z, et al. Photo-electro-catalysis enhancement on carbon nanotubes/titanium dioxide (CNTs/TiO2) composite prepared by a novel surfactant wrapping sol-gel method. Appl Cata B: Environ, 2008, 85: 17–23
Lin J, Zong R, Zhou M, et al. Photoelectric catalytic degradation of methylene blue by C60-modified TiO2 nanotube array. Appl Catal B: Environ, 2009, 89: 425–431
Dekany I, Kruger-Grasser R, Weiss A. Selective liquid sorption properties of hydrophobized graphite oxide nanostructures. Colloid Polym Sci, 1998, 276: 570–576
Bourlinos A B, Gournis D, Petridis D, et al. Graphite oxide: chemical reduction to graphite and surface modification with primary aliphatic amines and amino acids. Langmuir, 2003, 19: 6050–6055
Lerf A, He H, Forster M, et al. Structure of graphite oxide revisited. J Phys Chem B, 1998, 102: 4477–4482
Kovtyukhova N I, Ollivier P J, Martin B R, et al. Layer-by-layer assembly of ultrathin composite films from micron-sized graphite oxide sheets and polycations. Chem Mater, 1999, 11: 771–778
Bourlinos A B, Giannelis E P, Sanakis Y, et al. A graphite oxide-like carbogenic material derived from a molecular precursor. Carbon, 2006, 44: 1906–1912
He H, Klinowski J, Forster M, et al. A new structural model for graphite oxide. Chem Phys Lett, 1998, 287: 53–56
Hontoria-Lucas C, Lopez-Peinado A J, Lopez-Gonzalez J D, et al. Study of oxygen-containing groups in a series of graphite oxides: Physical and chemical characterization. Carbon, 1995, 33: 1585–1592
Szabo T, Szeri A, Dekany I. Composite graphitic nanolayers prepared by self-assembly between finely dispersed graphite oxide and a cationic polymer. Carbon, 2005, 43: 87–94
Jeong H K, Lee Y P, Lahaye R J W E. Evidence of graphitic AB stacking order of graphite oxides. J Am Chem Soc, 2008, 130: 1362–1366
Liu Z H, Wang Z M, Yang X J, et al. Intercalation of organic ammonium ions into layered graphite oxide. Langmuir, 2002, 18: 4926–4932
Xu J Y, Hu Y, Song L, et al. Preparation and characterization of polyacrylamide intercalated graphite oxide. Mater Res Bull, 2001, 36: 1833–1836
Wang G C, Yang Z Y, Li X W. Synthesis of poly(anilineco-o-anisidine)-intercalated graphite oxide composite by delamination/reassembling method. Carbon, 2005, 43: 2564–2570
Higashika S, Kimura K, Matsuo Y. Synthesis of polyaniline- intercalated graphite oxide. Letters to the Editor/Carbon, 1999, 37: 351–358
Rabin B, Liu P K Y, Scully S F. Intercalation of polypyrrole into graphite oxide. Syn Met, 2006, 156: 1023–1027
Rabin B, Liu P K Y, Wade W. Encapsulation of polyanilines into graphite oxide. Langmuir, 2006, 22: 1729–1734
Zhang Y H, He Y Q. Microstructure of graphite intercalated tin oxide and its influence on SnO2 based gas sensors. Front Mater Sci China, 2007, 1: 297–302
Yu J G, Zhao X J, Zhao Q N. XPS study on TiO2 photocatalytic thin film prepared by the sol-gel method. Chin J Mater Res, 2000, 14: 203–209
Pouilleau J, Devilliers D, Groult H. Surface study of a titanium-based ceramic electrode material by X-ray photoelectron spectroscopy. J Mater Sci, 1997, 32: 5645–5651
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zhang, Q., He, Y., Chen, X. et al. Structure and photocatalytic properties of TiO2-Graphene Oxide intercalated composite. Chin. Sci. Bull. 56, 331–339 (2011). https://doi.org/10.1007/s11434-010-3111-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-010-3111-x