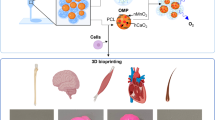
Abstract
A lack of biological activity hinders the application of synthetic hydrogels in tissue engineering and regenerative medicine. However, the use of glycopolypeptides in hydrogel synthesis may provide the materials with the desired biological activities. Herein, we prepared three in situ-forming hydrogels from various phenol-functionalized glycopolypeptides. The gelation time, mechanical properties, degradation properties, and biocompatibility of the hydrogels were assessed. Gelation time ranged from 11 to 380 s, depending on the concentration of horseradish peroxidase. The galactose-modified polypeptide hydrogel showed the highest storage modulus with an obvious stress relaxation phenomenon. The prepared hydrogels exhibited good degradation properties and compatibility to cells and tissues. Furthermore, the rate of immune cell accumulation around the mannose-modified polypeptide hydrogel was the fastest among the hydrogels.
Similar content being viewed by others
References
Pina S, Oliveira J M, Reis R L. Natural-based nanocomposites for bone tissue engineering and regenerative medicine: A review. Adv Mater, 2015, 27: 1143–1169
Griffith L G, Naughton G. Tissue engineering-current challenges and expanding opportunities. Science, 2002, 295: 1009–1014
Langer R, Vacanti J P. Tissue engineering. Science, 1993, 260: 920–926
Li Y, Rodrigues J, Tomas H. Injectable and biodegradable hydrogels: Gelation, biodegradation and biomedical applications. Chem Soc Rev, 2012, 41: 2193–2221
Annabi N, Tamayol A, Uquillas J A, et al. 25th anniversary article: Rational design and applications of hydrogels in regenerative medicine. Adv Mater, 2014, 26: 85–124
Hussey G S, Dziki J L, Badylak S F. Extracellular matrix-based materials for regenerative medicine. Nat Rev Mater, 2018, 3: 159–173
Pérez R A, Won J E, Knowles J C, et al. Naturally and synthetic smart composite biomaterials for tissue regeneration. Adv Drug Deliver Rev, 2013, 65: 471–496
Place E S, Evans N D, Stevens M M. Complexity in biomaterials for tissue engineering. Nat Mater, 2009, 8: 457–470
Lutolf M P, Hubbell J A. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nat Biotech, 2005, 23: 47–55
Dvir T, Timko B P, Kohane D S, et al. Nanotechnological strategies for engineering complex tissues. Nat Nanotech, 2011, 6: 13–22
Krannig K S, Schlaad H. Emerging bioinspired polymers: Glycopolypeptides. Soft Matter, 2014, 10: 4228–4235
Tsang K Y, Cheung M C H, Chan D, et al. The developmental roles of the extracellular matrix: Beyond structure to regulation. Cell Tissue Res, 2010, 339: 93–110
Rozario T, DeSimone D W. The extracellular matrix in development and morphogenesis: A dynamic view. Dev Biol, 2010, 341: 126–140
Godula K, Bertozzi C R. Synthesis of glycopolymers for microarray applications via ligation of reducing sugars to a poly(acryloyl hydrazide) scaffold. J Am Chem Soc, 2010, 132: 9963–9965
Kramer J R, Deming T J. Glycopolypeptides via living polymerization of glycosylated-l-lysine N-carboxyanhydrides. J Am Chem Soc, 2010, 132: 15068–15071
Mecham R P. The Extracellular Matrix: An Overview. Berlin: Springer, 2011
Rawat M, Gama C I, Matson J B, et al. Neuroactive chondroitin sulfate glycomimetics. J Am Chem Soc, 2008, 130: 2959–2961
Oh Y I, Sheng G J, Chang S K, et al. Tailored glycopolymers as anticoagulant heparin mimetics. Angew Chem Int Ed, 2013, 52: 11796–11799
Kramer J R, Deming T J. Recent advances in glycopolypeptide synthesis. Polym Chem, 2014, 5: 671–682
Herzner H, Reipen T, Schultz M, et al. Synthesis of glycopeptides containing carbohydrate and peptide recognition motifs. Chem Rev, 2000, 100: 4495–4538
Wang Y, Kiick K L. Monodisperse protein-based glycopolymers via a combined biosynthetic and chemical approach. J Am Chem Soc, 2005, 127: 16392–16393
Miura Y. Design and synthesis of well-defined glycopolymers for the control of biological functionalities. Polym J, 2012, 44: 679–689
Geng J, Mantovani G, Tao L, et al. Site-directed conjugation of “clicked” glycopolymers to form glycoprotein mimics: Binding to mammalian lectin and induction of immunological function. J Am Chem Soc, 2007, 129: 15156–15163
Miura Y, Hoshino Y, Seto H. Glycopolymer nanobiotechnology. Chem Rev, 2016, 116: 1673–1692
Ren K, He C, Xiao C, et al. Injectable glycopolypeptide hydrogels as biomimetic scaffolds for cartilage tissue engineering. Biomaterials, 2015, 51: 238–249
Xiao C, Zhao C, He P, et al. Facile synthesis of glycopolypeptides by combination of ring-opening polymerization of an Alkyne-Substituted N-carboxyanhydride and click “glycosylation”. Macromol Rapid Commun, 2010, 31: 991–997
Huang Y, Zeng Y, Yang J, et al. Facile functionalization of polypeptides by thiol-yne photochemistry for biomimetic materials synthesis. Chem Commun, 2011, 47: 7509–7511
Engler A C, Lee H, Hammond P T. Highly efficient “grafting onto” a polypeptide backbone using click chemistry. Angew Chem Int Ed, 2009, 48: 9334–9338
Lu H, Cheng J. Hexamethyldisilazane-mediated controlled polymerization of α-amino acid N-carboxyanhydrides. J Am Chem Soc, 2007, 129: 14114–14115
Levene P. The pentacetate of α-mannose. J Biol Chem., 1924, 59: 141–144
Ladmiral V, Mantovani G, Clarkson G J, et al. Synthesis of neoglycopolymers by a combination of “click chemistry” and living radical polymerization. J Am Chem Soc, 2006, 128: 4823–4830
Bae J W, Choi J H, Lee Y, et al. Horseradish peroxidase-catalysed in situ-forming hydrogels for tissue-engineering applications. J Tissue Eng Regen Med, 2015, 9: 1225–1232
Yu L, Ding J. Injectable hydrogels as unique biomedical materials. Chem Soc Rev, 2008, 37: 1473–1481
Wang D, Yang X, Liu Q, et al. Enzymatically cross-linked hydrogels based on a linear poly(ethylene glycol) analogue for controlled protein release and 3D cell culture. J Mater Chem B, 2018, 6: 6067–6079
Jin R, Hiemstra C, Zhong Z, et al. Enzyme-mediated fast in situ formation of hydrogels from dextran-tyramine conjugates. Biomaterials, 2007, 28: 2791–2800
Veitch N C. Horseradish peroxidase: A modern view of a classic enzyme. Phytochemistry, 2004, 65: 249–259
Park K M, Shin Y M, Joung Y K, et al. In situ forming hydrogels based on tyramine conjugated 4-arm-PPO-PEO via enzymatic oxidative reaction. Biomacromolecules, 2010, 11: 706–712
Huebsch N, Lippens E, Lee K, et al. Matrix elasticity of void-forming hydrogels controls transplanted-stem-cell-mediated bone formation. Nat Mater, 2015, 14: 1269–1277
Lee H P, Gu L, Mooney D J, et al. Mechanical confinement regulates cartilage matrix formation by chondrocytes. Nat Mater, 2017, 16: 1243–1251
Gilbert P M, Havenstrite K L, Magnusson K E G, et al. Substrate elasticity regulates skeletal muscle stem cell self-renewal in culture. Science, 2010, 329: 1078–1081
Dashnau J L, Sharp K A, Vanderkooi J M. Carbohydrate intramolecular hydrogen bonding cooperativity and its effect on water structure. J Phys Chem B, 2005, 109: 24152–24159
Murphy C M, Haugh M G, O’Brien F J. The effect of mean pore size on cell attachment, proliferation and migration in collagen-glycosaminoglycan scaffolds for bone tissue engineering. Biomaterials, 2010, 31: 461–466
O’Brien F J, Harley B A, Yannas I V, et al. The effect of pore size on cell adhesion in collagen-GAG scaffolds. Biomaterials, 2005, 26: 433–441
Wu D Q, Sun Y X, Xu X D, et al. Biodegradable and pH-sensitive hydrogels for cell encapsulation and controlled drug release. Biomacromolecules, 2008, 9: 1155–1162
Anderson J M, Rodriguez A, Chang D T. Foreign body reaction to biomaterials. Seminars Immunol, 2008, 20: 86–100
Chernyak A Y, Sharma G V M, Kononov L O, et al. 2-Azidoethyl glycosides: Glycosides potentially useful for the preparation of neoglycoconjugates. Carbohydrate Res, 1992, 223: 303–309
Oswald L, Trinh T T, Chan-Seng D, et al. Debromination of ATRP-made wang soluble polymer supports. Polymer, 2015, 72: 341–347
Chen C, Wang Z, Li Z. Thermoresponsive polypeptides from pegylated poly-l-glutamates. Biomacromolecules, 2011, 12: 2859–2863
Poché D S, Moore M J, Bowles J L. An unconventional method for purifying the N-carboxyanhydride derivatives of γ-alkyl-L-glutamates. Synth Commun, 1999, 29: 843–854
Daly W H, Poché D. The preparation of N-carboxyanhydrides of α-amino acids using bis(trichloromethyl)carbonate. Tetrahedron Lett, 1988, 29: 5859–5862
Jeong B, Bae Y H, Kim S W. Thermoreversible gelation of PEG-PLGA-PEG triblock copolymer aqueous solutions. Macromolecules, 1999, 32: 7064–7069
Park H, Guo X, Temenoff J S, et al. Effect of swelling ratio of injectable hydrogel composites on chondrogenic differentiation of encapsulated rabbit marrow mesenchymal stem cells in vitro. Biomacromolecules, 2009, 10: 541–546
Liu X, Jin X, Ma P X. Nanofibrous hollow microspheres self-assembled from star-shaped polymers as injectable cell carriers for knee repair. Nat Mater, 2011, 10: 398–406
Yoon H I, Yhee J Y, Na J H, et al. Bioorthogonal copper free click chemistry for labeling and tracking of chondrocytes in vivo. Bioconjugate Chem, 2016, 27: 927–936
Monks A, Scudiero D, Skehan P, et al. Feasibility of a high-flux anticancer drug screen using a diverse panel of cultured human tumor cell lines. J Natl Cancer Inst, 1991, 83: 757–766
Kloxin A M, Tibbitt M W, Anseth K S. Synthesis of photodegradable hydrogels as dynamically tunable cell culture platforms. Nat Protoc, 2010, 5: 1867–1887
Yesilyurt V, Webber M J, Appel E A, et al. Injectable self-healing glucose-responsive hydrogels with pH-regulated mechanical properties. Adv Mater, 2016, 28: 86–91
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was supported by the National Natural Science Foundation of China (Grant Nos. 21574127, 51622307, 51520105004, 51833010 and 51773199), and the Youth Innovation Promotion Association, CAS.
Supporting Information
The supporting information is available online at tech.scichina.com and link.springer.com . The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
Rights and permissions
About this article
Cite this article
Shi, S., Yu, S., Li, G. et al. Biocompatible in situ-forming glycopolypeptide hydrogels. Sci. China Technol. Sci. 63, 992–1004 (2020). https://doi.org/10.1007/s11431-019-1466-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11431-019-1466-1