Abstract
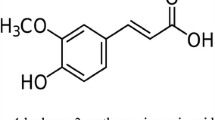
This study was designed to investigate the therapeutic potential of hydroalcoholic extracts of Acorus calamus (AC) in tibial and sural nerve transection (TST)-induced neuropathic pain in rats. The hot plate, paw heat allodynia, acetone drop, and pinprick tests were performed to assess the degree of heat hyperalgesia, heat and cold allodynia, and mechanical hyperalgesia, respectively, at different time intervals, i.e., day 0, 1, 3, 6, 9, 12, 15, 18, and 21. The tissue superoxide anion and total calcium were measured as markers of oxidative stress. Tissue myeloperoxidase activity was measured as a specific marker of inflammation. Histopathological evaluation was also performed in the nervous tissue to assess the axonal degeneration. Pregabalin served as positive control in this study. TST in rats significantly induced thermal hyperalgesia and allodynia, mechanical hyperalgesia, and increased the levels of superoxide anion, total calcium, and myeloperoxidase (MPO) activity. Moreover significant histological changes were also observed. Oral administration of AC hydroalcoholic extract (100 and 200 mg/kg for 14 days) attenuated TST-induced behavioral, biochemical, and histological changes. Acorus calamus has ameliorative potential in TST-induced painful neuropathy, and this effect may be attributed to its multiple actions including anti-inflammatory, antioxidant, and neuroprotective actions.





Similar content being viewed by others
References
Namaka M, Gramlich CR, Ruhlen D, Melanson M, Sutton I, Major J (2004) A treatment algorithm for neuropathic pain. Clin Ther 26:951–979
Alston RP, Pechon P (2005) Dysaesthesia associated with sternotomy for heart surgery. Br J Anaesth 95:153–158
Tavakoli M, Quattrini C, Abbott C, Kallinikos P, Marshall A, Finnigan J, Morgan P, Efron N, Boulton AJ, Malik RA (2010) Corneal confocal microscopy: a novel non-invasive test to diagnose and stratify the severity of human diabetic neuropathy. Diabetes Care. doi:10.1007/s00125-003-1086-8
Pramod K (2006) Chronic pain syndromes (non terminal illness). Indian J Anaesth 50:391–396
Kalita J, Vaipayee A, Misra UK (2006) Comparison of predinisolone with piroxicam in complex regional pain syndrome following stroke: a randomised controlled trial. QJM 99:89–95
Teasell RW, Mehta S, Aubut JA, Foulon B, Wolfe DL, Hsieh JT, Townson AF, Short C (2010) Spinal Cord Injury Rehabilitation Evidence Research Team. A systematic review of pharmacologic treatments of pain after spinal cord injury. Arch Phys Med Rehabil 91:816–831
Zakrzewska JM (2010) Medical management of trigeminal neuropathic pains. Exp Opin Pharmacother 11:1239–1254
Woolf CJ (2004) Pain: moving from symptom control toward mechanism-specific pharmacologic management. Ann Intern Med 140:441–451
Attal N, Cruccu G, Haanpaa M, Hansson P, Jensen TS, Nurmikko T, Sampaio C, Sindrup S, Wiffen P, Task Force EFNS (2006) EFNS guidelines on pharmacological treatment of neuropathic pain. Eur J Neurol 13:1153–1169
Comelli F, Giagnoni G, Bettoni I, Colleoni M, Costa B (2008) Antihyperalgesic effect of a Cannabis sativa extract in a rat model of neuropathic pain: mechanisms involved. Phytother Res 8:1017–1024
Kanter M (2008) Effects of Nigella sativa and its major constituent, thymoquinone on sciatic nerves in experimental diabetic neuropathy. Neurochem Res 1:87–96
Muthuraman A, Diwan V, Jaggi AS, Singh N, Singh D (2008) Ameliorative effects of Ocimum sanctum in sciatic nerve transection-induced neuropathy in rats. J Ethnopharmacol 120:56–62
Duke JA (1985) Handbook of medicinal herbs. CRC, Boca Raton
Mittal N, Ginwal HS, Varshney VK (2009) Pharmaceutical and biotechnological potential of Acorus calamus Linn: an indigenous highly valued medicinal plant species. Pharmacog Rev 3:83–93
Martis G, Rao A, Karanth KS (1991) Neuropharmacological activity of Acorus calamus. Fitoterapia 4:331–337
Parap RS, Mengi SA (2003) Evaluation of hypolipidemic activity of Acorus calamus Linn in rats. Indian Drugs 40:25–29
Young W (1992) Role of calcium in central nervous system injuries. J Neurotrauma 9:9–25
Shukla PK, Khanna VK, Ali MM, Maurya R, Khan MY, Srimal RC (2006) Neuroprotective effect of Acorus calamus against middle cerebral artery occlusion-induced ischaemia in rat. Hum Exp Toxicol 25:187–194
Hazra R, Ray K, Guha D (2007) Inhibitory role of Acorus calamus in ferric chloride-induced epileptogenesis in rat. Hum Exp Toxicol 26:947–953
Litchfield JTJR, Wilcoxon F (1949) A simplified method of evaluating dose–effect experiments. J Pharmacol Exp Ther 96:99–113
Lipnick RL, Cotruvo JA, Hill RN, Bruce RD, Stitzel KA, Walker AP, Chu I, Goddard M, Segal L, Springer JA, Myers RC (1995) Comparison of the up-and-down, conventional LD50, and fixed-dose acute toxicity procedures. Food Chem Toxicol 33:223–231
Schlede E, Mischke U, Diener W, Kayser D (1995) The international validation study of the acute toxic class method (oral). Arch Toxicol 69:659–670
Combes R, Gaunt I, Balls M (2006) A scientific and animal welfare assessment of the OECD health effects test guidelines for the safety testing of chemicals under the European Union REACH System. Altern Lab Anim 34:77–122
Lee BH, Won R, Baik EJ, Lee SH, Moon CH (2000) An animal model of neuropathic pain employing injury to the sciatic nerve branches. Neuroreport 11:657–661
Eddy NB, Touchberry CF, Lieberman JE (1950) Synthetic analgesics: I. Methadone isomers and derivatives. J Pharmacol Exp Ther 98:121–137
Choi Y, Yoon YW, Na HS, Kim SH, Chung JM (1994) Behavioral signs of ongoing pain and cold allodynia in a rat model of neuropathic pain. Pain 59:369–376
de la Calle L, Mena MA, Gonzalez-Escalada JR, Paıno CL (2002) Intrathecal transplantation of neuroblastoma cells decreases heat hyperalgesia and cold allodynia in a rat model of neuropathic pain. Brain Res Bull 59:205–211
Erichsen HK, Blackburn-Munro G (2002) Pharmacological characterization of the spared nerve injury model of neuropathic pain. Pain 98:151–161
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 193:265–275
Wang HD, Pagano PJ, Du Y, Cayatte AJ, Quinn MT, Brecher P, Cohen RA (1998) Superoxide anion from the adventitia of the rat thoracic aorta inactivates nitric oxide. Circ Res 82:810–818
Severinghaus JW, Ferrebee JW (1950) Calcium determination by flame photometry; methods for serum, urine, and other fluids. J Biol Chem 187:621–630
Grisham MB, Specian RD, Zimmerman TE (1994) Effects of nitric oxide synthase inhibition on the pathophysiology observed in a model of chronic granulomatous colitis. J Pharmacol Exp Ther 271:1114–1121
Patriarca P, Dri P, Snidero M (1977) Interference of myeloperoxidase with the estimation of superoxide dismutase activity. J Lab Clin Med 90:289–294
Yukari S, Sukumar P, Desai AE, Hadererm SS, Peter G, Douglas C, Anthony UDG, Ging KW (2004) Neurologic and histopathologic evaluation after high-volume intrathecal amitriptyline. Reg Anesth Pain Med 29:434–440
Muthuraman A, Jaggi AS, Singh N, Singh D (2008) Ameliorative effects of amiloride and pralidoxime in chronic constriction injury and vincristine-induced painful neuropathy in rats. Eur J Pharmacol 587:104–111
Jain V, Jaggi AS, Singh N (2009) Ameliorative potential of rosiglitazone in tibial and sural nerve transection-induced painful neuropathy in rats. Pharmacol Res 59:385–392
Cui JG, Holmin S, Mathiesen T, Meyerson BA, Linderoth B (2000) Possible role of inflammatory mediators in tactile hypersensitivity in rat models of mononeuropathy. Pain 88:239–248
Kuhad A, Chopra K (2009) Tocotrienol attenuates oxidative-nitrosative stress and inflammatory cascade in experimental model of diabetic neuropathy. Neuropharmacology 57:456–462
Marchand F, Perretti M, McMahon SB (2005) Role of immune system in chronic pain. Nat Rev Neurosci 6:521–532
Woolf CJ, Mannion RJ (1999) Neuropathic pain: aetiology, symptoms, mechanisms, and management. Lancet 353:1959–1964
Cecile H, Perrine I, Mohammed A, Denis G, Jean V, Frederique S (2005) Calcium dependence of axotomized sensory neurons excitability. Neurosci Lett 380:330–334
Siau C, Bennett GJ (2006) Dysregulation of neuronal calcium homeostasis in chemotherapy-evoked painful peripheral neuropathy. Anesth Analg 102:1485–1490
Glass JD, Culver DG, Levey AI, Nash NR (2002) Very early activation of m-calpain in peripheral nerve during Wallerian degeneration. J Neurol Sci 196:9–20
Zeinab K, Bereha K (2001) A role for free radicals and nitric oxide in delayed recovery in aged rats with chronic constriction nerve injury. Free Radic Biol Med 31:430–439
Siniscalco D, Fuccio C, Giordano C, Ferraraccio F, Palazzo E, Luongo L, Rossi F, Maione S, de Novellis V (2007) Role of reactive oxygen species and spinal cord apoptotic genes in the development of neuropathic pain. Pharmacol Res 55:158–166
Carriedo SG, Sensi SL, Yin HZ, Weiss JH (2000) AMPA exposures induce mitochondrial Ca(2+) overload and ROS generation in spinal motor neurons in vitro. J Neurosci 20:240–250
Rafatullah S, Tariq M, Mossa JS, Al-Yahya MA, Al-Said MS, Ageel AM (1994) Anti-secretagogue, anti-ulcer and cytoprotective properties of Acorus calamus in rats. Fitotherpia 65:19–23
Mehrotra S, Mishra KP, Maurya R, Srimal RC, Yadav VS, Pandey R, Singh VK (2003) Anticellular and immunosuppressive properties of ethanolic extract of Acorus calamus rhizome. Int Immunopharmacol 3:53–61
Gilani AU, Shah AJ, Ahmad M, Shaheen F (2006) Antispasmodic effect of Acorus calamus Linn. is mediated through calcium channel blockade. Phytother Res 20:1080–1084
Shukla PK, Khanna VK, Ali MM, Maurya RR, Handa SS, Srimal RC (2002) Protective effect of Acorus calamus against acrylamide induced neurotoxicity. Phytother Res 16:256–260
Manikandan S, Srikumar R, Jeya Parthasarathy N, Sheela Devi R (2005) Protective effect of Acorus calamus Linn on free radical scavengers and lipid peroxidation in discrete regions of brain against noise stress exposed rat. Biol Pharm Bull 28:2327–2330
Tippani R, Porika M, Rao AV, Abbagani S, Yellu NR, Tammidala C (2008) Analgesic activity of root extract of Acorus calamus Linn. Pharmacol Online 3:240–243
Yang L, Li S, Huang Y, Liang J, Wang Y (2008) Effects of Acorus gramineus and its component, alpha-asarone, on apoptosis of hippocampal neurons after seizure in immature rats. Neural Reg Res 3:19–24
Kumar K, Taylor RS, Jacques L, Eldabe S, Meglio M, Molet J, Thomson S, O’Callaghan J, Eisenberg E, Milbouw G, Buchser E, Fortini G, Richardson J, North RB (2007) Spinal cord stimulation versus conventional medical management for neuropathic pain: a multicentre randomised controlled trial in patients with failed back surgery syndrome. Pain 132:179–188
Bender G, Florian JA Jr, Bramwell S, Field MJ, Tan KK, Marshall S, DeJongh J, Bies RR, Danhof M (2010) Pharmacokinetic–pharmacodynamic analysis of the static allodynia response to pregabalin and sildenafil in a rat model of neuropathic pain. J Pharmacol Exp Ther 334:599–608
Park HJ, Joo HS, Chang HW, Lee JY, Hong SH, Lee Y, Moon DE (2010) Attenuation of neuropathy-induced allodynia following intraplantar injection of pregabalin. Can J Anaesth 57:664–671
Acknowledgments
The authors are grateful to the Department of Pharmaceutical Sciences and Drug Research, Punjabi University, Patiala, India, for supporting this study and providing technical facilities for the work. The authors also acknowledge the financial support from Council of Scientific and Industrial Research (CSIR) through senior research fellowship (SRF).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muthuraman, A., Singh, N. & Jaggi, A.S. Effect of hydroalcoholic extract of Acorus calamus on tibial and sural nerve transection-induced painful neuropathy in rats. J Nat Med 65, 282–292 (2011). https://doi.org/10.1007/s11418-010-0486-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-010-0486-6