Summary
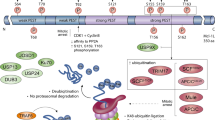
Mcl-1 is one member of the Bcl-2 family that has a very short protein half-life. Since its identification in 1993, a great number of studies have implicated that Mcl-1 plays an important role in various cell survival pathways. However, not until recently did the molecular mechanism by which Mcl-1 antagonizes apoptosis have begun to be elucidated. Mcl-1 is rapidly degraded in response to cell death signals and is immediately re-induced by survival stimuli. These results indicate that Mcl-1 plays an apical role in many cell death and survival regulatory programs.
Article PDF
Similar content being viewed by others
References
Kozopas K.M., Yang T., Buchan H.L., Zhou P. and Craig R.W., (1993). MCL1, a gene expressed in programmed myeloid cell differentiation, has sequence similarity to BCL2. Proc. Natl. Acad. Sci. U.S.A. 90: 3516–3520
Altmeyer A., Simmons R.C., Krajewski S., Reed J.C., Bornkamm G.W. and Chen-Kiang S., (1997). Reversal of EBV immortalization precedes apoptosis in IL-6-induced human B cell terminal differentiation. Immunity 7: 667–677
Chao J.R., Wang J.M., Lee S.F., Peng H.W., Lin Y.H., Chou C.H., Li J.C., Huang H.M., Chou C.K., Kuo M.L., Yen J.J. and Yang-Yen H.F., (1998). mcl-1 is an immediate-early gene activated by the granulocyte–macrophage colony-stimulating factor (GM-CSF) signaling pathway and is one component of the GM-CSF viability response. Mol. Cell. Biol. 18: 4883–4898
Huang H.M., Huang C.J. and Yen J.J., (2000). Mcl-1 is a common target of stem cell factor and interleukin-5 for apoptosis prevention activity via MEK/MAPK and PI-3K/Akt pathways. Blood 96: 1764–1771
Jourdan M., De Vos J., Mechti N. and Klein B., (2000). Regulation of Bcl-2-family proteins in myeloma cells by three myeloma survival factors: interleukin-6, interferon-alpha and insulin-like growth factor 1. Cell. Death. Differ. 7: 1244–1252
Leu C.M., Chang C. and Hu C., (2000). Epidermal growth factor (EGF) suppresses staurosporine-induced apoptosis by inducing mcl-1 via the mitogen-activated protein kinase pathway. Oncogene 19: 1665–1675
Opferman J.T., Letai A., Beard C., Sorcinelli M.D., Ong C.C. and Korsmeyer S.J., (2003). Development and maintenance of B and T lymphocytes requires antiapoptotic MCL-1. Nature 426: 671–676
Yang T., Buchan H.L., Townsend K.J. and Craig R.W., (1996). MCL-1, a member of the BLC-2 family, is induced rapidly in response to signals for cell differentiation or death, but not to signals for cell proliferation. J. Cell. Physiol. 166: 523–536
Wang J.M., Chao J.R., Chen W., Kuo M.L., Yen J.J. and Yang-Yen H.F., (1999). The antiapoptotic gene mcl-1 is up-regulated by the phosphatidylinositol 3-kinase/Akt signaling pathway through a transcription factor complex containing CREB. Mol. Cell. Biol. 19: 6195–6206
Wang J.M., Lai M.Z. and Yang-Yen H.F., (2003). Interleukin-3 stimulation of mcl-1 gene transcription involves activation of the PU.1 transcription factor through a p38 mitogen-activated protein kinase-dependent pathway. Mol. Cell. Biol. 23: 1896–1909
Townsend K.J., Zhou P., Qian L., Bieszczad C.K., Lowrey C.H., Yen A. and Craig R.W., (1999). Regulation of MCL1 through a serum response factor/Elk-1-mediated mechanism links expression of a viability-promoting member of the BCL2 family to the induction of hematopoietic cell differentiation. J. Biol. Chem. 274: 1801–1813
Nijhawan D., Fang M., Traer E., Zhong Q., Gao W., Du F. and Wang X., (2003). Elimination of Mcl-1 is required for the initiation of apoptosis following ultraviolet irradiation. Genes. Dev. 17: 1475–1486
Liu H., Peng H.W., Cheng Y.S., Yuan H.S. and Yang-Yen H.F., (2005). Stabilization and enhancement of the antiapoptotic activity of mcl-1 by TCTP. Mol. Cell. Biol. 25: 3117–3126
Yang T., Kozopas K.M. and Craig R.W., (1995). The intracellular distribution and pattern of expression of Mcl-1 overlap with, but are not identical to, those of Bcl-2. J. Cell. Biol. 128: 1173–1184
Jamil S., Sobouti R., Hojabrpour P., Raj M., Kast J. and Duronio V., (2005). A proteolytic fragment of Mcl-1 exhibits nuclear localization and regulates cell growth by interaction with Cdk1. Biochem. J. 387: 659–667
Cuconati A., Mukherjee C., Perez D. and White E., (2003). DNA damage response and MCL-1 destruction initiate apoptosis in adenovirus-infected cells. Genes. Dev. 17: 2922–2932
Warr M.R., Acoca S., Liu Z., Germain M., Watson M., Blanchette M., Wing S.S. and Shore G.C., (2005). BH3-ligand regulates access of MCL-1 to its E3 ligase. FEBS. Lett. 579: 5603–5608
Zhong Q., Gao W., Du F. and Wang X., (2005). Mule/ARF-BP1, a BH3-only E3 ubiquitin ligase, catalyzes the polyubiquitination of Mcl-1 and regulates apoptosis. Cell. 121: 1085–1095
Chen D., Kon N., Li M., Zhang W., Qin J. and Gu W., (2005). ARF-BP1/Mule is a critical mediator of the ARF tumor suppressor. Cell. 121: 1071–1083
Liu Z., Oughtred R. and Wing S.S., (2005). Characterization of E3Histone, a novel testis ubiquitin protein ligase which ubiquitinates histones. Mol. Cell. Biol. 25: 2819–2831
Chen L., Willis S.N., Wei A., Smith B.J., Fletcher J.I., Hinds M.G., Colman P.M., Day C.L., Adams J.M. and Huang D.C., (2005). Differential targeting of prosurvival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol. Cell. 17: 393–403
Wang K., Gross A., Waksman G. and Korsmeyer S.J., (1998). Mutagenesis of the BH3 domain of BAX identifies residues critical for dimerization and killing. Mol. Cell. Biol. 18: 6083–6089
Willis S.N., Chen L., Dewson G., Wei A., Naik E., Fletcher J.I., Adams J.M. and Huang D.C., (2005). Proapoptotic Bak is sequestered by Mcl-1 and Bcl-xL, but not Bcl-2, until displaced by BH3-only proteins. Genes. Dev. 19: 1294–1305
Fujise K., Zhang D., Liu J. and Yeh E.T., (2000). Regulation of apoptosis and cell cycle progression by MCL1. Differential role of proliferating cell nuclear antigen. J. Biol. Chem. 275:39,458–39,465
Zhang D., Li F., Weidner D., Mnjoyan Z.H. and Fujise K., (2002). Physical and functional interaction between myeloid cell leukemia 1 protein (MCL1) and Fortilin. The potential role of MCL1 as a fortilin chaperone. J. Biol. Chem. 277: 37430–37438
Bae J., Donigian J.R. and Hsueh A.J., (2003). Tankyrase 1 interacts with Mcl-1 proteins and inhibits their regulation of apoptosis. J. Biol. Chem. 278: 5195–5204
Thaw P., Baxter N.J., Hounslow A.M., Price C., Waltho J.P. and Craven C.J., (2001). Structure of TCTP reveals unexpected relationship with guanine nucleotide-free chaperones. Nat. Struct. Biol. 8: 701–704
MacDonald S.M., Rafnar T., Langdon J. and Lichtenstein L.M., (1995). Molecular identification of an IgE-dependent histamine-releasing factor. Science 269: 688–690
Li F., Zhang D. and Fujise K., (2001). Characterization of fortilin, a novel antiapoptotic protein. J. Biol. Chem. 276: 47,542–47,549
Gachet Y., Tournier S., Lee M., Lazaris-Karatzas A., Poulton T. and Bommer U.A., (1999). The growth-related, translationally controlled protein P23 has properties of a tubulin binding protein and associates transiently with microtubules during the cell cycle. J. Cell. Sci. 112 (Pt 8): 1257–1271
Tuynder M., Fiucci G., Prieur S., Lespagnol A., Geant A., Beaucourt S., Duflaut D., Besse S., Susini L., Cavarelli J., Moras D., Amson R. and Telerman A., (2004). Translationally controlled tumor protein is a target of tumor reversion. Proc. Natl. Acad. Sci. U.S.A. 101: 15,364–15,369
Yarm F.R., (2002). Plk phosphorylation regulates the microtubule-stabilizing protein TCTP. Mol. Cell. Biol. 22: 6209–6221
Rinkenberger J.L., Horning S., Klocke B., Roth K. and Korsmeyer S.J., (2000). Mcl-1 deficiency results in peri-implantation embryonic lethality. Genes. Dev. 14: 23–27
Opferman J.T., Iwasaki H., Ong C.C., Suh H., Mizuno S., Akashi K. and Korsmeyer S.J., (2005). Obligate role of anti-apoptotic MCL-1 in the survival of hematopoietic stem cells. Science 307: 1101–1104
Kaufmann S.H., Karp J.E., Svingen P.A., Krajewski S., Burke P.J., Gore S.D. and Reed J.C., (1998). Elevated expression of the apoptotic regulator Mcl-1 at the time of leukemic relapse. Blood 91: 991–1000
Zhou P., Levy N.B., Xie H., Qian L., Lee C.Y., Gascoyne R.D. and Craig R.W., (2001). MCL1 transgenic mice exhibit a high incidence of B-cell lymphoma manifested as a spectrum of histologic subtypes. Blood 97: 3902–3909
Acknowledgements
This work was supported in part by an intramural fund from Academia Sinica, and by grants NHRI-EX93-9119BN, NSC 92-3112-B-001-016 from National Health Research Institutes and the National Science Council of Taiwan to H.-F. Yang-Yen.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang-Yen, HF. Mcl-1: a highly regulated cell death and survival controller. J Biomed Sci 13, 201–204 (2006). https://doi.org/10.1007/s11373-005-9064-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11373-005-9064-4