Abstract
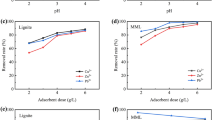
In this study, lignite-loaded nano-FeS (nFeS@Lignite) was successfully prepared by ultrasonic precipitation, and its potential for treating acid Cr(VI)-containing wastewater was explored. The results showed that the 40-–80-nm rod-shaped nFeS was successfully loaded onto lignite particles, and the maximum adsorption capacity of Cr(VI) by nFeS@Lignite reached 33.08 mg∙g−1 (reaction time = 120 min, pH = 4, temperature = 298.15 K). The adsorption process of Cr(VI) by nFeS@Lignite fitted the pseudo-second-order model and the Langmuir isotherm model, and thermodynamic results showed that the adsorption process was an endothermic process with an adsorption enthalpy of 28.0958 kJ·mol−1. The inhibition intensity of coexisting anions on Cr(VI) removal was in the order of PO43− > NO3− > SO42− > Cl−, and the increase of ionic strength resulted in more pronounced inhibition. Electrostatic adsorption, reduction, and precipitation were synergistically engaged in the adsorption of Cr(VI) by nFeS@Lignite, among which reduction played a major role. The characterization results showed that Fe2+, S2−, and Cr(VI) were converted to FeOOH, S8, SO42−, Fe2O3, Cr2O3, and Fe(III)-Cr(III) complexes. This research demonstrates that nFeS@Lignite is a good adsorbent with promising potential for application in the remediation of heavy metal-contaminated wastewater.












Similar content being viewed by others
Data availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abebe B, Taddesse AM, Kebede T et al (2017) Fe-Al-Mn ternary oxide nanosorbent: synthesis, characterization and phosphate sorption property. J Environ Chem Eng 5:1330–1340. https://doi.org/10.1016/j.jece.2017.02.026
Abilio TE, Soares BC, Jose JC et al (2021) Hexavalent chromium removal from water: adsorption properties of in natura and magnetic nanomodified sugarcane bagasse. Environ Sci Pollut Res 28:24816–24829. https://doi.org/10.1007/s11356-020-11726-8
Agah A, Falahati N (2021) Studying effect of modifying nano-mineral adsorbents on efficiency of dye removal from industrial effluents. J Min Environ 12:219–233. https://doi.org/10.22044/jme.2020.10122.1950
Bao S, Di J, Yang J et al (2022) Experimental study on adsorption characteristics of Cu2+ and Zn2+ by datong lignite. Environ Eng Res 27:210037. https://doi.org/10.4491/eer.2021.037
Barrera-Diaz CE, Lugo-Lugo V, Bilyeu B (2012) A review of chemical, electrochemical and biological methods for aqueous Cr(VI) reduction. J Hazard Mater 223–224:1–12. https://doi.org/10.1016/j.jhazmat.2012.04.054
Bayramoglu G, Arica MY (2011) Synthesis of Cr(VI)-imprinted poly(4-vinyl pyridine-co-hydroxyethyl methacrylate) particles: Its adsorption propensity to Cr(VI). J Hazard Mater 187:213–221. https://doi.org/10.1016/j.jhazmat.2011.01.022
Bayramoglu G, Akbulut A, Arica MY (2016) Aminopyridine modified Spirulina platensis biomass for chromium(VI) adsorption in aqueous solution. Water Sci Technol 74:914–926. https://doi.org/10.2166/wst.2016.281
Bayramoglu G, BurcuAngi S, Acikgoz-Erkaya I et al (2022) Preparation of effective green sorbents using O. Princeps alga biomass with different composition of amine groups: comparison to adsorption performances for removal of a model acid dye. J Mol Liq 347:118375. https://doi.org/10.1016/j.molliq.2021.118375
Chen Y, Liang W, Li Y et al (2019) Modification, application and reaction mechanisms of nano-sized iron sulfide particles for pollutant removal from soil and water: a review. Chem Eng J 362:144–159. https://doi.org/10.1016/j.cej.2018.12.175
Dang H, Zhang Y, Du P (2014) Enhanced removal of soluble Cr(VI) by using zero-valent iron composite supported by surfactant-modified zeolites. Water Sci Technol 70:1398–1404. https://doi.org/10.2166/wst.2014.392
Dong Y, Zeng W, Lin H et al (2020) Preparation of a novel water-soluble organosilane coating and its performance for inhibition of pyrite oxidation to control acid mine drainage at the source. Appl Surf Sci 531:147328. https://doi.org/10.1016/j.apsusc.2020.147328
Gao J, Yang L, Liu Y et al (2018) Scavenging of Cr(VI) from aqueous solutions by sulfide-modified nanoscale zero-valent iron supported by biochar. J Taiwan Inst Chem Eng 91:449–456. https://doi.org/10.1016/j.jtice.2018.06.033
Ge W, Zhao T, Chen S et al (2017) The effect of adsorbed chromium on the pyrolysis behavior of brown coal and the recovery of chromium. J Therm Anal Calorim 128:513–522. https://doi.org/10.1007/s10973-016-5890-z
Golovina VV, Eremina AO, Chesnokov NV et al (2018) Thermally activated brown and black coals as the sorbents of chromium(VI) from aqueous solutions. Solid Fuel Chem 52:240–246. https://doi.org/10.3103/s0361521918040043
Guo X, Fu S, Di J et al (2020) Preparation of nano FeS loaded on lignite by ultrasonic precipitation. Coal Sci Technol 9:152–159 (in Chinese)
He H, Wang J, Fei X et al (2022) Sequestration of free and chelated Ni(II) by structural Fe(II): Performance and mechanisms. Environ Pollut 292:118374. https://doi.org/10.1016/j.envpol.2021.118374
Hong Q, Liu C, Wang Z et al (2021) Electron transfer enhancing Fe(II)/Fe(III) cycle by sulfur and biochar in magnetic FeS@biochar to active peroxymonosulfate for 2,4-dichlorophenoxyacetic acid degradation. Chem Eng J 417:129238. https://doi.org/10.1016/j.cej.2021.129238
Jellali S, Azzaz A, Jeguirim M et al (2021) Use of lignite as a low-cost material for cadmium and copper removal from aqueous solutions: assessment of adsorption characteristics and exploration of involved mechanisms. Water 13:164. https://doi.org/10.3390/w13020164
Li Q, Zhang Y, Liao Y et al (2020) Removal of hexavalent chromium using biogenic mackinawite (FeS)-deposited kaolinite. J Colloid Interface Sci 572:236–245. https://doi.org/10.1016/j.jcis.2020.03.077
Lin Z, Hu Y, Yuan Y et al (2021) Comparative analysis of kinetics and mechanisms for Pb(II) sorption onto three kinds of microplastics. Ecotoxicol Environ Saf 208:111451. https://doi.org/10.1016/j.ecoenv.2020.111451
Liu W, Jin L, Xu J et al (2019) Insight into pH dependent Cr(VI) removal with magnetic Fe3S4. Chem Eng J 359:564–571. https://doi.org/10.1016/j.cej.2018.11.192
Lu J, Fan R, Wu H et al (2022) Simultaneous removal of Cr(VI) and Cu(II) from acid wastewater by electrocoagulation using sacrificial metal anodes. J Mol Liq 359:119276. https://doi.org/10.1016/j.molliq.2022.119276
Lv D, Zhou J, Cao Z et al (2019) Mechanism and influence factors of chromium(VI) removal by sulfide-modified nanoscale zerovalent iron. Chemosphere 224:306–315. https://doi.org/10.1016/j.chemosphere.2019.02.109
Lyu H, Tang J, Huang Y et al (2017) Removal of hexavalent chromium from aqueous solutions by a novel biochar supported nanoscale iron sulfide composite. Chem Eng J 322:516–524. https://doi.org/10.1016/j.cej.2017.04.058
Mu Y, Ai Z, Zhang L et al (2015) Insight into core-shell dependent anoxic Cr(VI) removal with Fe@Fe2O3 nanowires: indispensable role of surface bound Fe(II). ACS Appl Mater Interfaces 7:1997–2005. https://doi.org/10.1021/am507815t
Musat V, Stanica N, Anghel EM et al (2022) Magnetic core-shell iron oxides-based nanophotocatalysts and nanoadsorbents for multifunctional thin films. Membranes 12:466. https://doi.org/10.3390/membranes12050466
Nistico R (2021) A synthetic guide toward the tailored production of magnetic iron oxide nanoparticles. Bol Soc Esp Ceram Vidr 60:29–40. https://doi.org/10.1016/j.bsecv.2020.01.011
Park M, Park J, Kang J et al (2018) Removal of hexavalent chromium using mackinawite (FeS)-coated sand. J Hazard Mater 360:17–23. https://doi.org/10.1016/j.jhazmat.2018.07.086
Parnis M, García FE, Toledo MV et al (2022) Zerovalent iron nanoparticles-alginate nanocomposites for Cr(VI) removal in water-Influence of temperature, pH, dissolved oxygen, matrix, and nZVI surface composition. Water 14:484. https://doi.org/10.3390/w14030484
Qin L, He L, Yang W et al (2020) Preparation of a novel iron-based biochar composite for removal of hexavalent chromium in water. Environ Sci Pollut Res 27:9214–9226. https://doi.org/10.1007/s11356-019-06954-6
Rashid Khan Hu, Awan U, Zaman K et al (2021) Assessing hybrid solar-wind potential for industrial decarbonization strategies: global shift to green development. Energies 14:7620. https://doi.org/10.3390/en14227620
Sakthivel A, Thangagiri B, Jeyasubramanian K et al (2021) Switching the hydrophobic Neyveli lignite into hydrophilic type by surface modification and its subsequent use for removing Cr(VI)/F− from artificial pollutant. Fuel 298:120787. https://doi.org/10.1016/j.fuel.2021.120787
Samaraweera H, Edwards J, Reid C et al (2021) Pyrolyzed Ca-impregnated lignite for aqueous phosphate removal: batch and column studies. J Environ Chem Eng 9:106077. https://doi.org/10.1016/j.jece.2021.106077
Setshedi KZ, Bhaumik M, Songwane S et al (2013) Exfoliated polypyrrole-organically modified montmorillonite clay nanocomposite as a potential adsorbent for Cr(VI) removal. Chem Eng J 222:186–197. https://doi.org/10.1016/j.cej.2013.02.061
Shu Y, Ji B, Cui B et al (2020) Almond shell-derived, biochar-supported, nano-zero-valent iron composite for aqueous hexavalent chromium removal: performance and mechanisms. Nanomaterials 10:198. https://doi.org/10.3390/nano10020198
Shu Y, Ji B, Li Y et al (2021) Natural pyrite improved steel slag towards environmentally sustainable chromium reclamation from hexavalent chromium-containing wastewater. Chemosphere 282:130974. https://doi.org/10.1016/j.chemosphere.2021.130974
Singh S, Kumar V, Gupta P et al (2021) The synergy of mercury biosorption through Brevundimonas sp. IITISM22: kinetics, isotherm, and thermodynamic modeling. J Hazard Mater 415:125653. https://doi.org/10.1016/j.jhazmat.2021.125653
Sun L, Wang H, Li X et al (2021) Ultrasonic-assisted preparation of α-Tocopherol/casein nanoparticles and application in grape seed oil emulsion. Ultrason Sonochem 80:105810. https://doi.org/10.1016/j.ultsonch.2021.105810
Sun W, Chen S, Xu M et al (2019) A model for transient diffusion in bidisperse pore structures. Pet Sci 16:1455–1470. https://doi.org/10.1007/s12182-019-0338-2
Sun Y, Lv D, Zhou J et al (2017) Adsorption of mercury (II) from aqueous solutions using FeS and pyrite: A comparative study. Chemosphere 185:452–461. https://doi.org/10.1016/j.chemosphere.2017.07.047
Vilela PB, Dalalibera A, Duminelli EC et al (2019) Adsorption and removal of chromium (VI) contained in aqueous solutions using a chitosan-based hydrogel. Environ Sci Pollut Res 26:28481–28489. https://doi.org/10.1007/s11356-018-3208-3
Wang K, Sun Y, Tang J et al (2020a) Aqueous Cr(VI) removal by a novel ball milled Fe0-biochar composite: role of biochar electron transfer capacity under high pyrolysis temperature. Chemosphere 241:125044. https://doi.org/10.1016/j.chemosphere.2019.125044
Wang L, Shi C, Wang L et al (2020b) Rational design, synthesis, adsorption principles and applications of metal oxide adsorbents: a review. Nanoscale 12:4790–4815. https://doi.org/10.1039/c9nr09274a
Wang T, Liu Y, Wang J et al (2019) In-situ remediation of hexavalent chromium contaminated groundwater and saturated soil using stabilized iron sulfide nanoparticles. J Environ Manage 231:679–686. https://doi.org/10.1016/j.jenvman.2018.10.085
Wang X, Wang Y, Wang X et al (2011) Microwave-assisted preparation of bamboo charcoal-based iron-containing adsorbents for Cr(VI) removal. Chem Eng J 174:326–332. https://doi.org/10.1016/j.cej.2011.09.044
Wei Z, Zhang S, Wang X et al (2021) A high Cr (VI) absorption efficiency and easy recovery adsorbent: electrospun polyethersulfone/polydopamine nanofibers. J Appl Polym Sci 138:50642. https://doi.org/10.1002/app.50642
Wu J, Wang XB, Zeng RJ (2017) Reactivity enhancement of iron sulfide nanoparticles stabilized by sodium alginate: taking Cr (VI) removal as an example. J Hazard Mater 333:275–284. https://doi.org/10.1016/j.jhazmat.2017.03.023
Xi Y, Xie T, Liu Y et al (2022) Carboxymethyl cellulose stabilized ferrous sulfide@extracellular polymeric substance for Cr(VI) removal: characterization, performance, and mechanism. J Hazard Mater 425:127837. https://doi.org/10.1016/j.jhazmat.2021.127837
Xiong Z, Zheng H, Hu Y et al (2021) Selective adsorption of Congo red and Cu(II) from complex wastewater by core-shell structured magnetic carbon@zeolitic imidazolate frameworks-8 nanocomposites. Sep Purif Technol 277:119053. https://doi.org/10.1016/j.seppur.2021.119053
Xu Y, Bao J, Zhang X et al (2019) Functionalized polyethersulfone nanofibrous membranes with ultra-high adsorption capacity for organic dyes by one-step electrospinning. J Colloid Interface Sci 533:526–538. https://doi.org/10.1016/j.jcis.2018.08.072
Yang H, Shen K, Fu P et al (2019) Preparation of a novel carbonaceous material for Cr(VI) removal in aqueous solution using oily sludge of tank bottom as a raw material. J Environ Chem Eng 7:102898. https://doi.org/10.1016/j.jece.2019.102898
Yang Y, Zhang Y, Wang G et al (2021) Adsorption and reduction of Cr(VI) by a novel nanoscale FeS/chitosan/biochar composite from aqueous solution. J Environ Chem Eng 9:105407. https://doi.org/10.1016/j.jece.2021.105407
Yao Y, Mi N, He C et al (2020) A novel colloid composited with polyacrylate and nano ferrous sulfide and its efficiency and mechanism of removal of Cr(VI) from water. J Hazard Mater 399:123082. https://doi.org/10.1016/j.jhazmat.2020.123082
Yuan Y, Wei X, Yin H et al (2022) Synergistic removal of Cr(VI) by S-nZVI and organic acids: the enhanced electron selectivity and pH-dependent promotion mechanisms. J Hazard Mater 423:127240. https://doi.org/10.1016/j.jhazmat.2021.127240
Zeng S, Zhong D, Xu Y et al (2022) A novel sulfide-modified nanoscale zero valent iron supported on porous anion exchange resin composite for Cr(VI) effective removal from waste. Chem Phys Lett 794:139494. https://doi.org/10.1016/j.cplett.2022.139494
Zhang H, Peng L, Chen A et al (2019a) Chitosan-stabilized FeS magnetic composites for chromium removal: characterization, performance, mechanism, and stability. Carbohydr Polym 214:276–285. https://doi.org/10.1016/j.carbpol.2019.03.056
Zhang R, Li D, Sun J et al (2020) In situ synthesis of FeS/Carbon fibers for the effective removal of Cr(VI) in aqueous solution. Front Environ Sci Eng 14:68. https://doi.org/10.1007/s11783-020-1247-8
Zhang S, Lyu H, Tang J et al (2019b) A novel biochar supported CMC stabilized nano zero-valent iron composite for hexavalent chromium removal from water. Chemosphere 217:686–694. https://doi.org/10.1016/j.chemosphere.2018.11.040
Zhao C, Liu L, Yang X et al (2022) Pyrolysis temperature and feedstock affected Cr(VI) removal capacity of sulfidated zerovalent iron: Importance of surface area and electrical conductivity. Chemosphere 296:133927. https://doi.org/10.1016/j.chemosphere.2022.133927
Zhao T, Ge W, Nie Y et al (2016a) Highly efficient detoxification of Cr(VI) by brown coal and kerogen: process and structure studies. Fuel Process Technol 150:71–77. https://doi.org/10.1016/j.fuproc.2016.05.001
Zhao T, Ge W, Yue F et al (2016b) Mechanism study of Cr(III) immobilization in the process of Cr(VI) removal by huolinhe lignite. Fuel Process Technol 152:375–380. https://doi.org/10.1016/j.fuproc.2016.06.037
Zhou L, Dong F, Liu J et al (2017) Coupling effect of Fe3+(aq) and biological, nano-sized FeS-coated limestone on the removal of redox-sensitive contaminants (As, Sb and Cr): implications for in situ passive treatment of acid mine drainage. Appl Geochem 80:102–111. https://doi.org/10.1016/j.apgeochem.2017.03.005
Zou H, Zhao J, He F et al (2021) Ball milling biochar iron oxide composites for the removal of chromium (Cr(VI)) from water: performance and mechanisms. J Hazard Mater 413:125252. https://doi.org/10.1016/j.jhazmat.2021.125252
Funding
The project is funded by the National Natural Science Foundation of China (41672247), Liaoning Province’s “Program for Promoting Liaoning Talents” (XLYC1807159), discipline innovation team of Liaoning Technical University (LNTU20TD-21), Liaoning Provincial Department of Education Project (LJKZ0324).
Author information
Authors and Affiliations
Contributions
Saiou Fu, Junzhen Di and Xuying Guo conceived and designed the experiments. The experiments were performed by Saiou Fu and Yanrong Dong. Data was analyzed by Saiou Fu and Sihang Bao. The manuscript was written by Saiou Fu and Hanzhe Li. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
All authors have read this manuscript and consent for publication in Environmental Science and Pollution Research.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fu, S., Di, J., Guo, X. et al. Preparation of lignite-loaded nano-FeS and its performance for treating acid Cr(VI)-containing wastewater. Environ Sci Pollut Res 30, 3351–3366 (2023). https://doi.org/10.1007/s11356-022-22411-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22411-3