Abstract
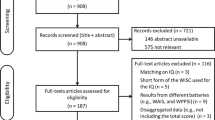
Trace elements persist in the environment, and their early exposure may adversely affect children's intellectual development. To clarify the influence of blood trace element levels in newborns and school-aged children, we used Wechsler Intelligence Scale for children (WISC-CR) to explore intellectual development level of 148 school-aged children based on a population cohort study. Lead (Pb), selenium (Se), arsenic (As), copper (Cu), manganese (Mn) and chromium (Cr) in cord blood and Pb, As, Cu in venous blood were determined by inductively coupled plasma mass spectrometry (ICP-MS) and atomic absorption spectrometer (AAS). Our analysis of the correlation between children's mental development and trace element content found children’s cognitive abilities negatively correlate with Pb (PIQ: β=-0.109, P=0.03737) and Cu (PIQ: β=-0.031, P=0.04431; FISQ: β=-0.031, P=0.02137) levels in cord blood. Prenatal low-level As exposure may negatively affect girls’ performance intelligence quotient (PIQ) and verbal intelligence quotient (VIQ). There were differences in Se levels in cord blood and venous blood between boys and girls (P=0.010; P=0.073). High Se levels were associated with a lower VIQ in boys and a higher VIQ in girls. Prenatal exposure to Pb, As and Cu may weaken children’s cognitive abilities at school age. Se exposure may have opposite effects on cognitive abilities affected by dose and gender.

Similar content being viewed by others
Data Availability
The data that support the findings of this study are not openly available due to the inclusion of information that could compromise the subject's privacy, but are available from the corresponding author on reasonable request.
Abbreviations
- Pb:
-
lead
- As:
-
arsenic
- Se:
-
selenium
- Mn:
-
manganese
- Cu:
-
copper
- Cr:
-
chromium
- WISC-CR:
-
Wechsler intelligence scale for children
- ICP-MS:
-
inductively coupled plasma emission mass spectroscopy
- DBH:
-
dopamine β-hydroxylase
- ROS:
-
reactive oxygen species
- LOD:
-
limit of detection
- IQR:
-
interquartile range
- C-Pb:
-
lead concentrations in cord blood
- C-Se:
-
selenium concentrations in cord blood
- C-Cu:
-
copper concentrations in cord blood
- C-Mn:
-
manganese concentrations in cord blood
- C-Cr:
-
chromium concentrations in cord blood
- C-As:
-
arsenic concentrations in cord blood
- V-Pb:
-
lead concentrations in venous blood
- V-Se:
-
selenium concentrations in venous blood
- V-Cu:
-
copper concentrations in venous blood
- CI:
-
confidence interval
- GM:
-
geometric mean
- GSD:
-
geometric standard deviation
- IQ:
-
intelligence quotient
- PIQ:
-
performance intelligence quotient
- VIQ:
-
verbal intelligence quotient
- FSIQ:
-
full-scale intelligence quotient
References
Amorós R, Murcia M, González L et al (2019) Maternal copper status and neuropsychological development in infants and preschool children. Int J Hygiene Environ Health 222:503–512. https://doi.org/10.1016/j.ijheh.2019.01.007
Aschner M, Erikson K (2017) Manganese. Adv Nutr 8:520–521. https://doi.org/10.3945/an.117.015305
Atsdr (2007) Toxicological profile for lead. Agency for Toxic Substances and Disease Registry, US
Atsdr (2012) Toxicological profile for cadmium. Agency for Toxic Substances and Disease Registry, US
Baksi S, Pradhan A (2021) Thyroid hormone: sex-dependent role in nervous system regulation and disease. Biol Sex Differ 12:25–38. https://doi.org/10.1186/s13293-021-00367-2
Banna HU, Anjum A, Biswas S et al (2021) Parental Lead Exposure Promotes Neurobehavioral Disorders and Hepatic Dysfunction in Mouse Offspring. Biol Trace Elem Res. https://doi.org/10.1007/s12011-021-02709-y
Bauer JA, Devick KL, Bobb JF et al (2020) Associations of a metal mixture measured in multiple biomarkers with IQ: Evidence from italian adolescents living near ferroalloy industry. Environ Health Perspect 128:097002-1-097002–12. https://doi.org/10.1289/EHP6803
Bhattacharjee A, Chakraborty K, Shukla A (2017) Cellular copper homeostasis: Current concepts on its interplay with glutathione homeostasis and its implication in physiology and human diseases. Metallomics 9:1376–1388. https://doi.org/10.1039/c7mt00066a
Brady K, Herrera Y, Zenick H, Herrera Y (1975) Influence of parental lead exposure on subsequent learning ability of offspring. Pharmacol Biochem Behav 3:561–565. https://doi.org/10.1016/0091-3057(75)90173-2
Brown EE, Shah P, Pollock BG et al (2019) Lead (Pb) in Alzheimer’s Dementia: A Systematic Review of Human Case- Control Studies. Curr Alzheimer Res 16:353–361. https://doi.org/10.2174/1567205016666190311101445
Calamandrei G, Ricceri L, Meccia E, et al (2020) Pregnancy exposome and child psychomotor development in three European birth cohorts. Environ Res 181. https://doi.org/10.1016/j.envres.2019.108856
Castriotta L, Rosolen V, Biggeri A et al (2020) The role of mercury, selenium and the Se-Hg antagonism on cognitive neurodevelopment: A 40-month follow-up of the Italian mother-child PHIME cohort. Int J Hygiene Environ Health 230:113604. https://doi.org/10.1016/j.ijheh.2020.113604
Chung SE, Cheong HK, Ha EH et al (2015) Maternal blood manganese and early neurodevelopment: The mothers and children’s environmental health (MOCEH) study. Environ Health Perspect 123:717–722. https://doi.org/10.1289/ehp.1307865
Diana A, Purnamasari DM, Rahmannia S, et al (2019) Multimicronutrient biomarkers are related to anemia during infancy in Indonesia: A repeated cross-sectional study. Curr Dev Nutr 3. https://doi.org/10.1093/cdn/nzz022
Dion LA, Saint-Amour D, Sauvé S et al (2018) Changes in water manganese levels and longitudinal assessment of intellectual function in children exposed through drinking water. NeuroToxicology 64:118–125. https://doi.org/10.1016/j.neuro.2017.08.015
do Nascimento SN, Barth A, Göethel G, et al (2015) Cognitive deficits and ALA-D-inhibition in children exposed to multiple metals. Environ Res 136:387–395. https://doi.org/10.1016/j.envres.2014.10.003
Erfanizadeh M, Noorafshan A, Naseh M, Karbalay-Doust S (2021) The effects of copper sulfate on the structure and function of the rat cerebellum: A stereological and behavioral study. IBRO Neurosci Rep 11:119–127. https://doi.org/10.1016/j.ibneur.2021.09.001
Erikson KM, Thompson K, Aschner J, Aschner M (2007) Manganese neurotoxicity: a focus on the neonate. Pharmacol Ther 113:369–377. https://doi.org/10.1016/j.pharmthera.2006.09.002
Forns J, Fort M, Casas M et al (2014) Exposure to metals during pregnancy and neuropsychological development at the age of 4 years. NeuroToxicology 40:16–22. https://doi.org/10.1016/j.neuro.2013.10.006
Freire C, Amaya E, Gil F et al (2018) Prenatal co-exposure to neurotoxic metals and neurodevelopment in preschool children: The Environment and Childhood (INMA) Project. Sci Total Environ 621:340–351. https://doi.org/10.1016/j.scitotenv.2017.11.273
Gade M, Comfort N, Re DB (2021) Sex-specific neurotoxic effects of heavy metal pollutants: Epidemiological, experimental evidence and candidate mechanisms. Environ Res 201:111558. https://doi.org/10.1016/j.envres.2021.111558
Garí M, Grzesiak M, Krekora M, et al (2022a) Prenatal exposure to neurotoxic metals and micronutrients and neurodevelopmental outcomes in early school age children from Poland. Environ Res 204. https://doi.org/10.1016/j.envres.2021.112049
Garza-Lombó C, Pappa A, Panayiotidis MI et al (2019) Arsenic-induced neurotoxicity: a mechanistic appraisal. J Biol Inorg Chem 24:1305–1316. https://doi.org/10.1007/s00775-019-01740-8
Ge Hua-jiao (2019) Evaluation of heavy metal pollution in water, sediment and aquatic organisms in rural rivers and ecological health risks in Wujiang District, Suzhou. World Latest Medicine Information (Electronic Version) 19:293–296. https://doi.org/10.19613/j.cnki.1671-3141.2019.46.186
Ge Hua-jiao, Zhou Ju-ping, Gu Hai-jin, Tian Hai-lin (2017) Investigation on lead (Pb), cadmium (Cd) and chromium (Cr) pollution in rural soils of Wujiang district in Suzhou city. Mod Prev Med 44:2912–2915. http://doi.org/CNKI:SUN:XDYF.0.2017-16-008
Grandjean P, Landrigan PJ (2014) Neurobehavioural effects of developmental toxicity. Lancet Neurol 13:330–338. https://doi.org/10.1016/S1474-4422(13)70278-3
Gunier RB, Arora M, Jerrett M et al (2015) Manganese in teeth and neurodevelopment in young Mexican-American children. Environ Res 142:688–695. https://doi.org/10.1016/j.envres.2015.09.003
Guo BQ, Cai SZ, Guo JL et al (2013) Levels of prenatal mercury exposure and their relationships to neonatal anthropometry in Wujiang City, China. Environ Pollut 182:184–189. https://doi.org/10.1016/j.envpol.2013.07.023
Guy M, Accrombessi M, Fievet N et al (2018) Toxics (Pb, Cd) and trace elements (Zn, Cu, Mn) in women during pregnancy and at delivery, South Benin, 2014–2015. Environ Res 167:198–206. https://doi.org/10.1016/j.envres.2018.06.054
Hamadani JD, Tofail F, Nermell B et al (2011) Critical windows of exposure for arsenic-associated impairment of cognitive function in pre-school girls and boys: A population-based cohort study. Int J Epidemiol 40:1593–1604. https://doi.org/10.1093/ije/dyr176
Hamilton EM, Young SD, Bailey EH, Watts MJ (2018) Chromium speciation in foodstuffs: a review. Food Chem 250:105–112. https://doi.org/10.1016/j.foodchem.2018.01.016
Horning KJ, Caito SW, Tipps KG et al (2015) Manganese is Essential for Neuronal Health. Ann Rev Nutr 35:71–108. https://doi.org/10.1146/annurev-nutr-071714-034419
Hsieh RL, Huang YL, Shiue HS et al (2014) Arsenic methylation capacity and developmental delay in preschool children in Taiwan. Int J Hygiene Environ Health 217:678–686. https://doi.org/10.1016/j.ijheh.2014.02.004
Huang X, Dong YL, Li T et al (2021) Dietary selenium regulates micrornas in metabolic disease: Recent progress. Nutrients 13:1527. https://doi.org/10.3390/nu13051527
Islam GMR, Rahman MM, Hasan MI, et al (2021) Hair, serum and urine chromium levels in children with cognitive defects: A systematic review and meta-analysis of case control studies. Chemosphere 133017. https://doi.org/10.1016/j.chemosphere.2021.133017
Jain RB (2014) Thyroid function and serum copper, selenium, and zinc in general U.S. population. Biol Trace Elem Res 159:87–98. https://doi.org/10.1007/s12011-014-9992-9
Kalita J, Kumar V, Misra UK, Bora HK (2018) Memory and Learning Dysfunction Following Copper Toxicity: Biochemical and Immunohistochemical Basis. Mol Neurobiol 55:3800–3811. https://doi.org/10.1007/s12035-017-0619-y
Karim Y, Siddique AE, Hossen F, et al (2019) Dose-dependent relationships between chronic arsenic exposure and cognitive impairment and serum brain-derived neurotrophic factor. Environ Int 131. https://doi.org/10.1016/j.envint.2019.105029
Koshy B, Srinivasan M, Zachariah SM et al (2020) Body iron and lead status in early childhood and its effects on development and cognition: A longitudinal study from urban Vellore. Public Health Nutr 23:1896–1906. https://doi.org/10.1017/S1368980019004622
Lee J, Freeman JL (2014) Zebrafish as a model for investigating developmental lead (Pb) neurotoxicity as a risk factor in adult neurodegenerative disease: A mini-review. NeuroToxicology 43:57–64. https://doi.org/10.1016/j.neuro.2014.03.008
Lee MS, do Eum K, Golam M, et al (2021) Umbilical cord blood metal mixtures and birth size in Bangladeshi children. Environ Health Perspect 129. https://doi.org/10.1289/EHP7502
Levin-Schwartz Y, Politis MD, Gennings C, et al (2021) Nephrotoxic metal mixtures and preadolescent kidney function. Children 8. https://doi.org/10.3390/children8080673
Li Z, Liang C, Huang K et al (2018) Umbilical Serum Copper Status and Neonatal Birth Outcomes: a Prospective Cohort Study. Biol Trace Elem Res 183:200–208. https://doi.org/10.1007/s12011-017-1144-6
Liang C Mei, Wu X Yan, Huang K, et al (2019) Trace element profiles in pregnant women’s sera and umbilical cord sera and influencing factors: Repeated measurements. Chemosphere 218:869–878. https://doi.org/10.1016/j.chemosphere.2018.11.115
Lidsky TI, Schneider JS (2003) Lead neurotoxicity in children: Basic mechanisms and clinical correlates. Brain 126:5–19. https://doi.org/10.1093/brain/awg014
Linares AM, Unrine JM, Thaxton Wigging A, et al (2021) Blood’s Concentration of Lead and Arsenic Associated with Anemia in Peruvian Children. J Environ Public Health 2021. https://doi.org/10.1155/2021/7283514
Liu X, Piao J, Zhang Y et al (2016) Serum Copper Status in School-Age Children and Pregnant Women in China Nutrition and Health Survey 2010–2012. Biol Trace Elem Res 173:268–274. https://doi.org/10.1007/s12011-016-0639-x
Llanos RM, Mercer JFB (2002) The molecular basis of copper homeostasis and copper-related disorders. DNA and Cell Biology 21:259–270. https://doi.org/10.1089/104454902753759681
Magrì A, la Mendola D, Rizzarelli E (2021) Nerve growth factor peptides bind copper(Ii) with high affinity: A thermodynamic approach to unveil overlooked neurotrophin roles. Int J Mol Sci 22. https://doi.org/10.3390/ijms22105085
Martin KV, Sucharew H, Dietrich KN, et al (2021) Co-exposure to manganese and lead and pediatric neurocognition in East Liverpool, Ohio. Environ Res 202. https://doi.org/10.1016/j.envres.2021.111644
Martins AC, Krum BN, Queirós L et al (2020) Manganese in the diet: bioaccessibility, adequate intake, and neurotoxicological effects. J Agric Food Chem 68:12893–12903. https://doi.org/10.1021/acs.jafc.0c00641
Mehdi Y, Hornick JL, Istasse L, Dufrasne I (2013) Selenium in the environment, metabolism and involvement in body functions. Molecules 18:3292–3311. https://doi.org/10.3390/molecules18033292
Michael K Georgieff (2007) Nutrition and the developing brain: nutrient priorities and measurement. Am J Clin Nutr 614S–620S. https://doi.org/10.1093/ajcn/85.2.614S
Mielke HW, Laidlaw MAS, Gonzales C (2010) Lead (Pb) legacy from vehicle traffic in eight California urbanized areas: Continuing influence of lead dust on children’s health. Sci Total Environ 408:3965–3975. https://doi.org/10.1016/j.scitotenv.2010.05.017
Močenić I, Kolić I, Nišević JR, et al (2019) Prenatal selenium status, neonatal cerebellum measures and child neurodevelopment at the age of 18 months. Environ Res 176. https://doi.org/10.1016/j.envres.2019.108529
Mochizuki H (2019) Arsenic neurotoxicity in humans. Int J Mol Sci 20. https://doi.org/10.3390/ijms20143418
Nordberg M, Nordberg GF (2016) Trace element research-historical and future aspects. J Trace Elem Med Biol 38:46–52. https://doi.org/10.1016/j.jtemb.2016.04.006
Petro A, Sexton HG, Miranda C, et al (2016) Persisting neurobehavioral effects of developmental copper exposure in wildtype and metallothionein 1 and 2 knockout mice. BMC Pharmacol Toxicol 17. https://doi.org/10.1186/s40360-016-0096-3
Polanska K, Hanke W, Pawlas N, et al (2018) Sex-dependent impact of low-level lead exposure during prenatal period on child psychomotor functions. Int J Environ Res Public Health 15. https://doi.org/10.3390/ijerph15102263
Polanska K, Krol A, Sobala W et al (2016) Selenium status during pregnancy and child psychomotor development - Polish Mother and Child Cohort study. Pediatr Res 79:863–869. https://doi.org/10.1038/pr.2016.32
Rihel J (2018) Copper on the brain. Nat Chem Biol 14:638–639. https://doi.org/10.1038/s41589-018-0089-1
Rocha A, Trujillo KA (2019) Neurotoxicity of low-level lead exposure: History, mechanisms of action, and behavioral effects in humans and preclinical models. NeuroToxicology 73:58–80. https://doi.org/10.1016/j.neuro.2019.02.021
Rodrigues EG, Bellinger DC, Valeri L, et al (2016a) Neurodevelopmental outcomes among 2- to 3-year-old children in Bangladesh with elevated blood lead and exposure to arsenic and manganese in drinking water. Environ Health 15https://doi.org/10.1186/s12940-016-0127-y
Rodríguez-Barranco M, Lacasaña M, Gil F et al (2014) Cadmium exposure and neuropsychological development in school children in southwestern Spain. Environ Res 134:66–73. https://doi.org/10.1016/j.envres.2014.06.026
Rosado JL, Ronquillo D, Kordas K et al (2007) Arsenic exposure and cognitive performance in Mexican Schoolchildren. Environ Health Perspect 115:1371–1375. https://doi.org/10.1289/ehp.9961
Sanders AP, Claus Henn B, Wright RO (2015) Perinatal and Childhood Exposure to Cadmium, Manganese, and Metal Mixtures and Effects on Cognition and Behavior: A Review of Recent Literature. Curr Environ Health Rep 2:284–294. https://doi.org/10.1007/s40572-015-0058-8
Santa Maria MP, Hill BD, Kline J (2019) Lead (Pb) neurotoxicology and cognition. Appl Neuropsychol Child 8:272–293. https://doi.org/10.1080/21622965.2018.1428803
Sekovanić A, Piasek M, Orct T, et al (2020) Mercury exposure assessment in mother–infant pairs from continental and coastal croatia. Biomolecules 10https://doi.org/10.3390/biom10060821
Singh G, Singh V, Sobolewski M et al (2018) Sex-dependent effects of developmental lead exposure on the brain. Front Genet 9:89. https://doi.org/10.3389/fgene.2018.00089
Skröder H, Kippler M, de Loma J et al (2018) Predictors of selenium biomarker kinetics in 4–9-year-old Bangladeshi children. Environ Int 121:842–851. https://doi.org/10.1016/j.envint.2018.10.018
Skröder HM, Hamadani JD, Tofail F et al (2015) Selenium status in pregnancy influences children’s cognitive function at 1.5 years of age. Clin Nutr 34:923–930. https://doi.org/10.1016/j.clnu.2014.09.020
Skrökler H, Kippler M, Tofail F, Vahter M (2017) Early-life selenium status and cognitive function at 5 and 10 years of age in Bangladeshi children. Environ Health Perspect 125:117003-1-117003–13. https://doi.org/10.1289/EHP1691
Steinbrenner H, Sies H (2013) Selenium homeostasis and antioxidant selenoproteins in brain: Implications for disorders in the central nervous system. Arch Biochem Biophys 536:152–157. https://doi.org/10.1016/j.abb.2013.02.021
Stuss M, Michalska-Kasiczak M, Sewerynek E (2017) The role of selenium in thyroid gland pathophysiology. Endokrynol Pol 68:440–465. https://doi.org/10.5603/EP.2017.0051
Sun H, Chen W, Wang D et al (2014) The effects of prenatal exposure to low-level cadmium, lead and selenium on birth outcomes. Chemosphere 108:33–39. https://doi.org/10.1016/j.chemosphere.2014.02.080
Sun HJ, Xiang P, Luo J et al (2016) Mechanisms of arsenic disruption on gonadal, adrenal and thyroid endocrine systems in humans: A review. Environ Int 95:61–68. https://doi.org/10.1016/j.envint.2016.07.020
Takeuchi H, Taki Y, Nouchi R et al (2019) Association of copper levels in the hair with gray matter volume, mean diffusivity, and cognitive functions. Brain Struct Funct 224:1203–1217. https://doi.org/10.1007/s00429-019-01830-y
Theophanides T, Anastassopoulou J (2002) Copper and carcinogenesis. Crit Rev Oncol Hematol 42:57–64. https://doi.org/10.1016/s1040-8428(02)00007-0
Tolins M, Ruchirawat M, Landrigan P (2014) The developmental neurotoxicity of arsenic: Cognitive and behavioral consequences of early life exposure. Ann Glob Health 80:303–314. https://doi.org/10.1016/j.aogh.2014.09.005
Tsuji JS, Garry MR, Perez V, Chang ET (2015) Low-level arsenic exposure and developmental neurotoxicity in children: A systematic review and risk assessment. Toxicology 337:91–107. https://doi.org/10.1016/j.tox.2015.09.002
Vinceti M, Mandrioli J, Borella P et al (2014) Selenium neurotoxicity in humans: Bridging laboratory and epidemiologic studies. Toxicol Lett 230:295–303. https://doi.org/10.1016/j.toxlet.2013.11.016
Vlasak T, Jordakieva G, Gnambs T et al (2019) Blood lead levels and cognitive functioning: A meta-analysis. Sci Total Environ 668:678–684. https://doi.org/10.1016/j.scitotenv.2019.03.052
Wang B, Liu J, Liu B et al (2018) Prenatal exposure to arsenic and neurobehavioral development of newborns in China. Environ Int 121:421–427. https://doi.org/10.1016/j.envint.2018.09.031
Wang J, Wu W, Li H et al (2019) Relation of prenatal low-level mercury exposure with early child neurobehavioral development and exploration of the effects of sex and DHA on it. Environ Int 126:14–23. https://doi.org/10.1016/j.envint.2019.02.012
Wang S, Hu C, Lu A, et al (2021a) Association between prenatal exposure to persistent organic pollutants and neurodevelopment in early life: A mother-child cohort (Shanghai, China). Ecotoxicol Environ Saf 208https://doi.org/10.1016/j.ecoenv.2020.111479
Wang SS, Lu AX, Cao LL, et al (2022) Effects of prenatal exposure to persistent organic pollutants on neonatal Outcomes:A mother-child cohort (Shanghai, China). Environ Res 203https://doi.org/10.1016/j.envres.2021.111767
Wang SX, Wang ZH, Cheng XT et al (2007) Arsenic and fluoride exposure in drinking water: Children’s IQ and growth in Shanyin Country, Shanxi Province, China. Environ Health Perspect 115:643–647. https://doi.org/10.1289/ehp.9270
Wang X, Huang X, Zhou L et al (2021) Association of arsenic exposure and cognitive impairment: A population-based cross-sectional study in China. NeuroToxicology 82:100–107. https://doi.org/10.1016/j.neuro.2020.11.009
Wasserman GA, Liu X, LoIacono NJ, et al (2014) A cross-sectional study of well water arsenic and child IQ in Maine schoolchildren. Environ Health 13https://doi.org/10.1186/1476-069X-13-23
Xiu-juan Yu, Yu Zeng, Jing-jing Na (2015) Research on the distribution characteristics of As, Cr, Pb, Cd in farmland soils and vegetables in south suburb of Suzhou. Adm Tech Environ Monit 27:29–33. https://doi.org/10.3969/j.issn.1006-2009.2015.05.008
Zhang Y, Lu W, Han M et al (2016) Biphasic effects of copper on rat learning and memory in the Morris Water Maze. Ann Clin Lab Sci 46:346–352
Zhou CC, He YQ, Gao ZY, et al (2020a) Sex differences in the effects of lead exposure on growth and development in young children. Chemosphere 250https://doi.org/10.1016/j.chemosphere.2020a.126294
Zhou T, Guo J, Zhang J et al (2020) Sex-Specific Differences in Cognitive Abilities Associated with Childhood Cadmium and Manganese Exposures in School-Age Children: a Prospective Cohort Study. Biol Trace Elem Res 193:89–99. https://doi.org/10.1007/s12011-019-01703-9
Funding
This work was supported by grants from the National Science Foundation of China (Grant No.81973062), the National Key R&D Program of China (Grant No. 2017YFC1600500), and the Science and Technology Commission of Shanghai Municipality (Grant No.09DZ2200900). The funders had no role in the conduct of the study, the analysis or interpretation of data, and the preparation, review, or approval of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Methodology, software and data curation were performed by Wang Yihong and Wang Yaqian. The first draft of the manuscript was written by Wang Yihong and all authors commented on previous versions of the manuscript. Yan Chonghuai contributed to conceptualizing, writing, reviewing and editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Ethics Approval
This research was approved by the Medical Ethics Committee of Xinhua Hospital affiliated with Shanghai Jiao Tong University School of Medicine and was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent to Participate
Pregnant women were provided with written informed consent prior to the study at delivery. We obtained written informed consent from the guardian before follow-up.
Consent to Publish
Not applicable.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Y., Wang, Y. & Yan, C. Gender differences in trace element exposures with cognitive abilities of school-aged children: a cohort study in Wujiang city, China. Environ Sci Pollut Res 29, 64807–64821 (2022). https://doi.org/10.1007/s11356-022-20353-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-20353-4