Abstract
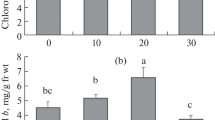
The present study was carried out to elucidate effects of synthesized magnetic nanoparticles (MNPs) on morphological and physiological parameters and main essential oil components of Calotropis procera seedlings. For this purpose, 21-day-old seedlings grown under hydroponic conditions were treated by the different MNP concentrations (0, 50, 100, 150, and 200 mg L-1). The results showed that the growth parameters, chlorophyll pigments, soluble sugars, and total proteins significantly increased in leaf under MNP treatment, except for the root length. As compared to the control, MNPs induced a substantial change in the activities of antioxidant enzymes, H2O2, and malondialdehyde contents. Ascorbate peroxidase activity showed a meaningful increase in leaf treated with 200 mg L-1 MNPs, while superoxide dismutase activity and concentration of H2O2 conspicuously decreased relative to the control. Moreover, MNPs enhanced geranial, 1,8-cineol, a-phellandrene, citronellal, camphor, and terpinen-4-ol contents as major components. These results suggest that MNPs could be a promising method of iron application in agricultural systems. Regarding the effects of MNPs, 200-mg L-1 MNPs were most effective on the production of main essential oils and plant growth that could serve as a favorable elicitor for plant improvement.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Abdi G, Salehi H, Khosh-Khui M (2008) Nano silver: a novel nanomaterial for removal of bacterial contaminants in valerian (Valeriana officinalis L.) tissue culture. Acta Physiol Plant 30(5):709–714. https://doi.org/10.1007/s11738-008-0169-z
Aebi H (1974) Catalases. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Academic Press, New York, pp 673–684. https://doi.org/10.1016/b978-0-12-091302-2.50032-3
Askary M, Talebi SM, Amini F, Dousti Balout Bangan A (2016) Effect of NaCl and iron oxide nanoparticles on Mentha piperita essential oil composition. Environ Exp Bot 14:27–32. https://doi.org/10.22364/eeb.14.05
Barhoumi L, Oukarroum A, Ben Taher L, Smiri L, Abdelmelek H, Dewez D (2015) Effects of superparamagnetic iron oxide nanoparticles on photosynthesis and growth of the aquatic plant lemna gibba. Arch Environ Contam Toxicol 68(3):510–520. https://doi.org/10.1007/s00244-014-0092-9
Bastani S, Hajiboland R, Khatamian M, Saket Oskoui M (2018) Nano iron (Fe) complex is an effective source of Fe for tobacco plants grown under low Fe supply. J Soil Sci Plant Nutr 18(2):524–541. https://doi.org/10.4067/S0718-95162018005001602
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assay applicable to acrylamide gels. Ann Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of proteindye binding. Annu Rev Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Choedon T, Mathan G, Arya S, Kumar VL, Kumar V (2006) Anticancer and cytotoxic properties of the latex of Calotropis procera in a transgenic mouse model of hepatocellular carcinoma. World J Gastroenterol 12:2517–2522. https://doi.org/10.3748/wjg.v12.i16.2517
Cvjetko P, Milošić A, Domijan AM, Vinković Vrček I, Tolić S, Peharec Štefanić S et al (2017) Toxicity of silver ions and differently coated silver nanoparticles in Allium cepa roots. Ecotoxicol Environ Saf 137:18–28. https://doi.org/10.1016/j.ecoenv.2016.11.009
Davies NW (1990) Gas chromatographic retention indices of monoterpenes and sesquiterpenes on methylsilicon and carbowax 20M Phases. J Chromatogr 503:1–24. https://doi.org/10.1016/S0021-9673(01)81487-4
Dubey VS, Bhalla R, Luthra R (2003) Sucrose mobilization in relation to essential oil biogenesis during palmarosa (Cymbopogon martinii Roxb. Wats. Var. Motia) inflorescence development. J Biosci 28(4):479–487. https://doi.org/10.1007/BF02705122
Fales FW (1951) The assimilation and degradation of carbohydrates by yeast cells. J Biol Chem 193:113–124
Ghafariyan MH, Malakouti MJ, Dadpour MR, Stroeve P, Madmoudi M (2013) Effects of magnetite nanoparticles on soybean chlorophyll. Environ Sci Technol 47(18):10645–10652. https://doi.org/10.1021/es402249b
Ghasemi R, Mirahmadi-zare SZ, Nasr-Esfahani MH, Allafchian A, Behmanesh M (2019) Optical biosensing of Streptococcus agalactiae based on core/shell magnetic nanoparticle-quantum dot. Anal Bioanal Chem 411:6733–6743. https://doi.org/10.1007/s00216-019-02046-z
Ghasemzadeh A, Jaafar HZE, Rahmat A (2010) Antioxidant activities, total phenolics and flavonoids content in two varieties of Malaysia young ginger (Zingiber officinale Roscoe). Molecules 15:4324–4333. https://doi.org/10.3390/molecules15064324
Giannotolitis CN, Ries SK (1997) Superoxide dismutase: II. Purification and quantitative relationship with water-soluble protein in seedling. Plant Physiol 59:315–318. https://doi.org/10.1104/pp.59.2.315
Guo R, Yuan G, Wang Q (2011) Effect of sucrose and mannitol on the accumulation of health-promoting compounds and the activity of metabolic enzymes in broccoli sprouts. Sci Hortic 128:159–165. https://doi.org/10.1016/j.scienta.2011.01.014
Hao Y, Fang P, Ma C, White JC, Xiang Z, Wang H, Zhang Z, Rui Y, Xing B (2019) Engineered nanomaterials inhibit Podosphaera pannosa infection on rose leaves by regulating phytohormones. Environ Res 170:1–6. https://doi.org/10.1016/j.envres.2018.12.008
Hashemi S, Asrar Z, Pourseyedi S, Nadernejad N (2019) Investigation of ZnO nanoparticles on proline, anthocyanin contents and photosynthetic pigments and lipid peroxidation in the soybean. IET Nanobiotechnol 13(1):66–70. https://doi.org/10.1049/iet-nbt.2018.5212
Hatami M, Ghorbanpour M (2014) Defense enzymes activity and biochemical variations of Pelargonium zonale in response to nanosilver particles and dark storage. Turk J Biol 38:130–139. https://doi.org/10.3906/biy-1304-64
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Heidarvand L, Maali-Amiri R (2013) Physio-biochemical and proteome analysis of chickpea in early phases of cold stress. J Plant Physiol 170:459–469. https://doi.org/10.1016/j.jplph.2012.11.021
Ho TT, Murthy HN, Park SY (2020) Methyl jasmonate induced oxidative stress and accumulation of secondary metabolites in plant cell and organ cultures. Int J Mol Sci 21(3):1–18. https://doi.org/10.3390/ijms21030716
Hodges DM, DeLong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid reactive substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611. https://doi.org/10.1007/s004250050524
Iannone MF, Groppa MD, Sousa ME, Raap MBFV, Benavides MP (2016) Impact of magnetite iron oxide nanoparticles on wheat (Triticum aestivum L.) development: Evaluation of oxidative damage. Environ Exp Bot 131:77–88. https://doi.org/10.1016/j.envexpbot.2016.07.004
Iqbal Z, Lateef M, Jabbar A, Muhammad G, Khan MN (2005) Anthelmintic activity of Calotropis procera (Ait.) Ait. F. flowers in sheep. J Ethnopharmacol 102(2):256–261. https://doi.org/10.1016/j.jep.2005.06.022
Jasim B, Thomas R, Mathew J, Radhakrishnan EK (2017) Plant growth and diosgenin enhancement effect of silver nanoparticles in Fenugreek (Trigonella foenum-graecum L.). Saudi Pharm J 25:443–447. https://doi.org/10.1016/j.jsps.2016.09.012
Jiang HS, Qiu XN, Li GB, Li W, Yin LY (2014) Silver nanoparticles induced accumulation of reactive oxygen species and alteration of antioxidant systems in the aquatic plant Spirodela polyrhiza. Environ Toxicol Chem 33:1398–1405. https://doi.org/10.1002/etc.2577
Khatami M, Alijani H, Sharifi I, Sharifi F, Pourseyedi S, Kharazi S (2017) Leishmanicidal activity of biogenic Fe3O4 nanoparticles. Sci Pharm 85(4):36. https://doi.org/10.3390/scipharm85040036
Lafmejani ZN, Jafari AA, Moradi P, Moghadam AL (2018) Impact of foliar application of iron-chelate and iron nano particles on some morpho-physiological traits and essential oil composition of peppermint (Mentha piperita L.). J Essent Oil-Bear Plants 21:1374–1384. https://doi.org/10.1080/0972060X.2018.1556122
Li XM, Tian SL, Pang ZC, Shi JY (2009) Extraction of Cuminum cyminum essential oil by combination technology of organic solvent with low boiling point and system distillation. Food Chem 115:1114–1119. https://doi.org/10.1016/j.foodchem.2008.12.091
Li J, Chang P, Huang J, Wang Y, Yuan H, Ren H (2013a) Physiological effects of magnetic iron oxide nanoparticles towards watermelon. J Nanosci Nanotechnol 13(8):5561–5567. https://doi.org/10.1166/jnn.2013.7533
Li L, Mak KY, Leung CW, Chan KY, Chan WK, Zhong W, Pong PWT (2013b) Effect of synthesis conditions on the properties of citric–acid coated iron oxide nanoparticles. Microelectron Eng 110:329–334. https://doi.org/10.1016/j.mee.2013.02.045
Li J, Hu J, Ma C, Wang Y, Wu C, Huang J, Xing B (2016) Uptake, translocation and physiological effects of magnetic iron oxide (g-Fe2O3) nanoparticles in corn (Zea mays L.). Chemosphere 159:326–334. https://doi.org/10.1016/j.chemosphere.2016.05.083
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic bio membranes. Methods Enzymol 148:350–382. https://doi.org/10.1016/0076-6879(87)48036-1
Ma C, Chhikara S, Minocha R, Long S, Musante C, White JC, Xing B, Dhankher Parkash O (2015a) Reduced silver nanoparticle phytotoxicity in crambe abyssinica with enhanced glutathione production by overexpressing bacterial g-glutamylcysteine synthase. Environ Sci Technol 49(16):10117. https://doi.org/10.1021/acs.est.5b02007
Ma C, White J, Dhankher O, Xing B (2015b) Metal-based nanotoxicity and detoxification pathways in higher plants. Environ Sci Technol 49(12):7109–7122. https://doi.org/10.1021/acs.est.5b00685
Makarov VV, Love AJ, Sinitsyna OV, Makarova SS, Yaminsky IV, Taliansky ME, Kalinina NO (2014) Green nanotechnologies: synthesis of metal nanoparticles using plants. Acta Nat 6(1):35–44
Malik LA, Bashir A, Ahmad N, Qureashi A, Pandith AH (2020) Exploring metal Ion adsorption and antifungal properties of carbon-Coated magnetite composite. Chem Select 11:3208–3216. https://doi.org/10.1002/slct.201904830
Medinger J, Nedyalkova M, Lattuada M (2021) Solvothermal synthesis combined with design of experiments—optimization approach for magnetite nanocrystal clusters. Nanomaterials (Basel) 11(2):360. https://doi.org/10.3390/nano11020360
Mittler R (2017) ROS are good. Trends Plant Sci 22:11–19. https://doi.org/10.1016/j.tplants.2016.08.002
Moharrami F, Hosseini B, Sharafi A, Farjaminehad M (2017) Enhanced production of hyoscyamine and scopolamine from genetically transformed root culture of Hyoscyamus reticulatus L. elicited by iron oxide nanoparticles. In Vitro Cell Dev Biol Plant 53:104–111. https://doi.org/10.1007/s11627-017-9802-0
Mohasseli V, Farbood F, Moradi A (2020) Antioxidant defense and metabolic responses of lemon balm (Melissa officinalis L.) to Fe-nano-particles under reduced irrigation regimes. Ind Crops. Prod 149:112338. https://doi.org/10.1016/j.indcrop.2020.112338
Mohebodini M, Fathi R, Mehri N (2017) Optimization of hairy root induction in chicory (Cichorium intybus L.) and effects of nanoparticles on secondary metabolites accumulation. Iran J Genet Plant Breed 6:60–68. https://doi.org/10.30479/ijgpb.2017.1491
Mozafari AA, Havas F, Ghaderi N (2018) Application of iron nanoparticles and salicylic acid in in vitro culture of strawberries (Fragaria × ananassa Duch.) to cope with drought stress. Plant Cell Tissue Org Cult 132:511–523. https://doi.org/10.1007/s11240-017-1347-8
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Plant Physiol 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x|
Nair PMG, Chung IM (2014) Physiological and molecular level effects of silver nanoparticles exposure in rice (oryza sativa L.) seedlings. Chemosphere 112:105–113. https://doi.org/10.1016/j.chemosphere.2014.03.056
Nair PMG, Kim S, Chung IM (2014) Copper oxide nanoparticle toxicity in mung bean (vigna radiata L.) seedlings: physiological and molecular level responses of in vitro grown plants. Acta Physiol Plant 36(11):2947–2958. https://doi.org/10.1007/s11738-014-1667-9
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Ngan HTM, Tung HT, Le BV, Nhut DT (2020) Evaluation of root growth, antioxidant enzyme activity and mineral absorbability of carnation (Dianthus caryophyllus “Express golem”) plantlets cultured in two culture systems supplemented with iron nanoparticles. Sci Hortic 272:109612. https://doi.org/10.1016/j.scienta.2020.109612
Oktay M, Gülҫin İ, Küfrevioğlu Öİ (2003) Determination of in vitro antioxidant activity of fennel (Foeniculum vulgare) seed extracts. LWT-Food Sci Technol 36:263–271. https://doi.org/10.1016/S0023-6438(02)00226-8
Olson BJSC, Markwell J (2007) Assays for determination of protein concentration. Curr Protoc Protein Sci 48:3.4.1–3.4.29. https://doi.org/10.1002/0471140864.ps0304s48|
Pu S, Ma H, Zinchenko A, Chu W (2017) Novel highly porous magnetic hydrogel beads composed of chitosan and sodium citrate: an effective adsorbent for the removal of heavy metals from aqueous solutions. Environ Sci Pollut Res 24:16520–16530. https://doi.org/10.1007/s11356-017-9213-0
Răcuciu M, Creangă DE (2007) Influence of water-based ferrofluid upon chlorophylls in cereals. J Magn Magn Mater 311(1):291–294. https://doi.org/10.1016/j.jmmm.2006.10.1185
Razavizadeh R, Farahzadianpoor F, Adabavazeh F, Komatsu S (2019) Physiological and morphological analyses of Thymus vulgaris L. in vitro cultures under polyethylene glycol (PEG)-induced osmotic stress. In Vitro Cell Dev Biol Plant 55:342–357. https://doi.org/10.1007/s11627-019-09979-1
Razavizadeh R, Adabavazeh F, Komatsu S (2020) Chitosan effects on the elevation of essential oils and antioxidant activity of Carum copticum L. seedlings and callus cultures under in vitro salt stress. J Plant Biochem Biotechnol 29:473–483. https://doi.org/10.1007/s13562-020-00560-1
Ren H, Liu L, Liu C, He S, Huang J, Li J, Zhang Y, Huang XJ, Gu N (2011) Physiological investigation of magnetic iron oxide nanoparticles towards chinese mung bean. J Biomed Nanotechnol 7(5):677–684. https://doi.org/10.1166/jbn.2011.1338
Sagner S, Kneer R, Wanner G, Cosson JP, Deus-Neumann B, Zenk MH (1998) Hyperaccumulation, complexation and distribution of nickel in Sebertia acuminata. Phytochemistry 47:339–347. https://doi.org/10.1016/S0031-9422(97)00593-1
Sehgal R, Roy S, Kumar V (2006) Evaluation of cytotoxic potential of latex of Calotropis procera and podophyllotoxin in Allium cepa model. Biocell 30:9–13
Shankramma K, Yallappa S, Shivanna MB, Manjanna J (2015) Fe2O3 magnetic nanoparticles to enhance S. lycopersicum (tomato) plant growth and their biomineralization. Appl Nanosci 6(7):983–990. https://doi.org/10.1007/s13204-015-0510-y
Sheykhbaglou R, Sedghi M, Fathi-Achachlouie B (2018) The effect of ferrous nano-oxide particles on physiological traits and nutritional compounds of soybean (Glycine max L.) seed. An Acad Bras Cienc 90(1):485–494. https://doi.org/10.1590/0001-3765201820160251
Tripathi DK, Singh S, Singh S, Srivastava PK, Singh VP, Singh S et al (2017) Nitric oxide alleviates silver nanoparticles (AgNps)-induced phytotoxicity in Pisum sativum seedlings. Plant Physiol Biochem 110:167–177. https://doi.org/10.1016/j.plaphy.2016.06.015
Vadivel N, Yuvakkumar R, Suriyaprabha R, Rajendran V (2012) Catalytic effect of iron nanoparticles on heterocyst, protein and chlorophyll content of Anabaena sp. Int J Green Nanotechnol Biol 4(3):326–338. https://doi.org/10.1080/19430892.2012.706185
Wang H, Kou X, Pei Z, Xiao J, Shan X, Xing B (2011) Physiological effects of magnetite (Fe3O4) nanoparticles on perennial ryegrass (lolium perenne L.) and pumpkin (cucurbita mixta) plants. Nanotoxicology 5(1):30–42. https://doi.org/10.3109/17435390.2010.489206
Yang D, Hu J, Fu S (2009) Controlled synthesis of magnetite−silica nanocomposites via a seeded sol−gel approach. J Phys Chem C 113(18):7646–7651. https://doi.org/10.1021/jp900868d
Zhang B, Zheng LP, Yi Li W, Wen Wang J (2013) Stimulation of artemisinin production in Artemisia annua hairy roots by Ag-SiO2 core-shell nanoparticles. Curr Nanosci 9:363–370. https://doi.org/10.2174/1573413711309030012
Funding
This study was supported by Shahid Bahonar University of Kerman, Iran.
Author information
Authors and Affiliations
Contributions
F.A, N.N., and S.P designed this study. F.A performed the experiment and analyzed the data. F.A wrote the manuscript. N.N., S.P., R.R. and H.M reviewed and edited the manuscript. All authors supervised the project and discussed the results and approved the final version of manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All authors carefully read and approved the study.
Consent for publication
All authors are agreed to publish the manuscript.
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Gangrong Shi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Adabavazeh, F., Nadernejad, N., Pourseyedi, S. et al. Synthesis of magnetic nanoparticles and their effects on growth and physiological parameters of Calotropis procera seedlings. Environ Sci Pollut Res 29, 59027–59042 (2022). https://doi.org/10.1007/s11356-022-19660-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-19660-7