Abstract
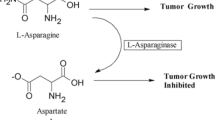
Asparaginases important role in the treatment of leukemia. It is part of chemotherapy in the treatment of leukemia in the last three decades. l-Asparaginase is isolated from Fusarium sp. isolated from soil and purified using ammonium sulfate precipitation and Sephadex G 100. Characterization of the crude enzyme revealed it is a metalloprotease inhibited by EDTA. Hg2+, Cd2+, and Pb2+ also inhibited the enzyme. Mg2+, Zn2+, and Ca2+ activated l-asparaginase. Furthermore, kinetic studies of purified enzyme were carried out. Vmax and Km were 0.031 M and 454 U/mL, respectively. The optimum temperature was 30 °C and the optimum pH was 7. Concerning substrate specificity, gelatin and casein in addition to l-asparagine were tested. The enzyme was found to be nonspecific that could hydrolyze all tested substrates at different rates. The maximum enzyme activity was recorded in the case of l-asparagine, followed by casein and gelatin, respectively. The molecular weight of l-asparaginase was 22.5 kDa. The antileukemic cytotoxicity assay of the enzyme against RAW2674 leukemic cell lines by MTT viability test was estimated. The enzyme exhibited antileukemic activity with IC50 of 50.1 UmL−1. The current work presents additional information regarding the purification and characterization of the enzyme produced by Fusarium sp. and its evaluation as a potential antileukemic chemotherapeutic agent.








Similar content being viewed by others
Data availability
Not applicable.
References
Adebo OA, Kayitesi E, AdeyinkaAdebiyi J, Gbashi S, Temba MC, AdeseyeLasekan, Phoku JZ and Njobeh PB (2017) Mitigation of Acrylamide in Foods: An African Perspective, Acrylic Polymers in Healthcare, Boreddy S.R. Reddy, IntechOpen. https://doi.org/10.5772/intechopen.68982. https://doi.org/10.5772/intechopen.68982
Aishwarya SS, Selvarajan E, Iyappan S, Rajnish KN (2019) Recombinant l -asparaginase II from Lactobacillus casei subsp. casei ATCC 393 and its anticancer activity. Indian J Microbiol 59:313–320. https://doi.org/10.1007/s12088-019-00806-0
Alam S, Pranaw K, Tiwari R, Khare SK (2019) Recent Development in the Uses of Asparaginase as Food Enzyme. In B. Parameswaran, S. Varjani, & S. Raveendran (Eds.), Green Bio-processes (pp. 55–81). Springer Singapore. https://doi.org/10.1007/978-981-13-3263-0_5
Ameen F, AlNadhari S, Al-Homaidan AA (2021) Marine microorganisms as an untapped source of bioactive compounds. Saudi J Biol Sci 28:224–231. https://doi.org/10.1016/j.sjbs.2020.09.052
Amena S, Vishalakshi N, Prabhakar M, Dayanand A, Lingappa K (2010) Production, purification and characterization of l-asparaginase from streptomyces gulbargensis. Brazilian Journal of Microbiology: [Publication of the Brazilian Society for Microbiology] 41(1):173–178. https://doi.org/10.1590/S1517-838220100001000025
Batool T, Makky EA, Jalal M, Yusoff MM (2016) A comprehensive review on l-asparaginase and its applications. Appl Biochem Biotechnol 178:900–923. https://doi.org/10.1007/s12010-015-1917-3
Battistel A, Rocha B, Santos M, et al (2020) Allergic reactions to asparaginase: retrospective cohort study in pediatric patients with acute lymphoid leukemia. Hematol Transfus Cell Ther 43. https://doi.org/10.1016/j.htct.2019.10.007
Borah D, Yadav R, Sangra A et al (2012) Production, purification and process optimization of asparagines. Int J Pharm Pharm Sci 4:560–563
Brumano LP, da Silva FVS, Costa-Silva TA et al (2019) Development of L-asparaginase biobetters: current research status and review of the desirable quality profiles. Front Bioeng Biotechnol 6. https://doi.org/10.3389/fbioe.2018.00212
Cachumba JJM, Antunes FAF, Peres GFD, et al (2016) Current applications and different approaches for microbial l-asparaginase production. Brazilian journal of microbiology : [publication of the Brazilian Society for Microbiology] 47 Suppl 1:77–85. 10.1016/j.bjm.2016.10.004
Cappelletti D, Chiarelli LR, Pasquetto MV et al (2008) Helicobacter pyloril-asparaginase: a promising chemotherapeutic agent. Biochem Biophys Res Commun 377:1222–1226. https://doi.org/10.1016/j.bbrc.2008.10.118
Chandrasekhar PA (2012) International journal of research in chemistry and environment an international peer reviewed quarterly e-journal ; (IJRCE). International Journal of Research in Chemistry and Environment (IJRCE)
Chohan SM, Rashid N (2013) TK1656, a thermostable l-asparaginase from Thermococcus kodakaraensis, exhibiting highest ever reported enzyme activity. J Biosci Bioeng 116:438–443. https://doi.org/10.1016/j.jbiosc.2013.04.005
Costa IM, Schultz L, de Araujo Bianchi Pedra B et al (2016) Recombinant L-asparaginase 1 from Saccharomyces cerevisiae: an allosteric enzyme with antineoplastic activity. Sci Rep 6:36239. https://doi.org/10.1038/srep36239
Covini D, Tardito S, Bussolati O, R. Chiarelli, L., V. Pasquetto, M., Digilio, R., Valentini, G., & Scotti, C (2012) Expanding Targets for a Metabolic Therapy of Cancer: L-Asparaginase. Recent Patents on Anti-Cancer Drug Discovery 7(1):4–13. https://doi.org/10.2174/157489212798358001
de Moura Sarquis MI, EMM O, Santos AS, da Costa GL (2004) Production of L-asparaginase by filamentous fungi. Mem Inst Oswaldo Cruz 99:489–492. https://doi.org/10.1590/s0074-02762004000500005
Distasio JA, Salazar AM, Nadji M, Durden DL (1982) Glutaminase-free asparaginase fromvibrio succinogenes: an antilymphoma enzyme lacking hepatotoxicity. Int J Cancer 30:343–347. https://doi.org/10.1002/ijc.2910300314
Duval M, Suciu S, Ferster A et al (2002) Comparison of Escherichia coli–asparaginase with Erwinia-asparaginase in the treatment of childhood lymphoid malignancies: results of a randomized European Organisation for Research and Treatment of Cancer—Children’s Leukemia Group phase 3 trial. Blood 99:2734–2739. https://doi.org/10.1182/blood.V99.8.2734
El-Nagga NE-A, El-Ewasy SM, El-Shweihy NM (2014) Microbial L-asparaginase as a potential therapeutic agent for the treatment of acute lymphoblastic leukemia: the pros and cons. Int J Pharmacol 10:182–199. https://doi.org/10.3923/ijp.2014.182.199
El-Naggar NE-A, Deraz SF, El-Ewasy SM, Suddek GM (2018) Purification, characterization and immunogenicity assessment of glutaminase free L-asparaginase from Streptomyces brollosae NEAE-115. BMC Pharmacol Toxicol 19:51. https://doi.org/10.1186/s40360-018-0242-1
Farag AM, Hassan SW, Beltagy EA, El-Shenawy MA (2015) Optimization of production of anti-tumor l-asparaginase by free and immobilized marine Aspergillus terreus. Egypt J Aquat Res 41:295–302. https://doi.org/10.1016/j.ejar.2015.10.002
Foda MS, Zedan HH and Hashem SA (1980) Formation of L-glutaminase and Lasparaginase activities in Pichia polymorpha.Acta Microbiol. Pol 29(4):343–352
Gupta N, Dash S, Basak U (2009) L- asparaginases from fungi of Bhitarkanika mangrove ecosystem. Aspac J Mol Biol Biotechnol 17
He Q, Peng H, Sheng M, Hu S, Qiu J, Gu J (2019) Humidity Control Strategies for Solid-State Fermentation: Capillary Water Supply by Water-Retention Materials and Negative-Pressure Auto-controlled Irrigation. Frontiers in Bioengineering and Biotechnology 7:263. https://doi.org/10.3389/fbioe.2019.00263
Hölker U, Lenz J (2005) Solid-state fermentation — are there any biotechnological advantages? Curr Opin Microbiol 8:301–306. https://doi.org/10.1016/j.mib.2005.04.006
Huang L, Liu Y, Sun Y, Yan Q, Jiang, Z (2014) Biochemical characterization of a novel L-Asparaginase with low glutaminase activity from Rhizomucor miehei and its application in food safety and leukemia treatment. Applied and Environmental Microbiology 80(5):1561–1569. https://doi.org/10.1128/AEM.03523-13
Husain I, Sharma A, Kumar S, Malik F (2016) Purification and characterization of glutaminase free asparaginase from Pseudomonas otitidis: Induce apoptosis in human leukemia MOLT-4 cells. Biochimie 121:38–51. https://doi.org/10.1016/j.biochi.2015.11.012
Im C, L S, B de ABP, et al (2016) Recombinant L-asparaginase 1 from Saccharomyces cerevisiae: an allosteric enzyme with antineoplastic activity. Sci Rep 6:36239–36239. https://doi.org/10.1038/srep36239
Imada A, Igarasi S, Nakahama K, Isono M (2009) Asparaginase and glutaminase activities of micro-organisms. J Gen Microbiol 76:85–99. https://doi.org/10.1099/00221287-76-1-85
Karamitros CS, Labrou NE (2014) Extracellular expression and affinity purification of L-asparaginase from E. chrysanthemi in E. coli. Sustain Chem Process 2:1–8. https://doi.org/10.1186/s40508-014-0016-z
Koerholz D, Brueck M, Nuernberger W et al (2009) Chemical and immunological characteristics of four different L-asparaginase preparations. Eur J Haematol 42:417–424. https://doi.org/10.1111/j.1600-0609.1989.tb01464.x
Kornbrust BA, Stringer MA, Lange NEK, Hendriksen HV (2009) Asparaginase—an enzyme for acrylamide reduction in food products. In: Whitehurst RJ, Oort MV (eds) Enzymes in food technology, 2nd edn. Wiley-Blackwell, Hoboken, pp 59–87
Kumar S, Pakshirajan K, Venkata Dasu V (2009) Development of medium for enhanced production of glutaminase-free l-asparaginase from Pectobacterium carotovorum MTCC 1428. Appl Microbiol Biotechnol 84:477–486. https://doi.org/10.1007/s00253-009-1973-0
Kumar S, Venkata Dasu V, Pakshirajan K (2011) Purification and characterization of glutaminase-free L-asparaginase from Pectobacterium carotovorum MTCC 1428. Bioresour Technol 102:2077–2082. https://doi.org/10.1016/j.biortech.2010.07.114
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Lincoln L, Niyonzima F, More S (2019) Purification and properties of a fungal L-asparaginase from Trichoderma viride Pers: Sf Grey. J Microbiol Biotechnol Food Sci 9:310–316. https://doi.org/10.15414/jmbfs.2014.4.4.310-316
Lizardi-Jiménez MA, Hernández-Martínez R (2017) Solid state fermentation (SSF): diversity of applications to valorize waste and biomass. 3 Biotech 7. https://doi.org/10.1007/s13205-017-0692-y
Lopes AM, de Oliveira-Nascimento L, Ribeiro A et al (2017) Therapeutic l-asparaginase: upstream, downstream and beyond. Crit Rev Biotechnol 37:82–99. https://doi.org/10.3109/07388551.2015.1120705
Lowry OH, Rosebrough NJ, Farr AL, and Randall RJ (1951) Protein measurement with the Folin phenol reagent. J. Biol. Chem 193:265–275
Luhana K, Dave A, Patel K (2013) Production, purification and characterization of extracellular L-asparaginase (anti cancerous enzyme) from Aspergillus Niger. International Journal of Chemtech Applicaions 2:14–25
Manna S, Sinha A, Sadhukhan R, Chakrabarty SL (1995) Purification, characterization and antitumor activity of l-asparaginase isolated from Pseudomonas stutzeri MB-405. Curr Microbiol 30:291–298. https://doi.org/10.1007/BF00295504
Mashburn LT, Wriston JC (1964) Tumor inhibitory effect of l-asparaginase from Escherichia coli. Arch Biochem Biophys 105:450–453
Miskovic L, Tokic M, Savoglidis G, Hatzimanikatis V (2019) Control theory concepts for modeling uncertainty in enzyme kinetics of biochemical networks. Ind Eng Chem Res 58:13544–13554. https://doi.org/10.1021/acs.iecr.9b00818
More S (2013) Isolation, Purification and Characterization of Fungal Extracellular L-Asparaginase from Mucor hiemalis. Journal of Biocatalysis & Biotransformation 02(02). https://doi.org/10.4172/2324-9099.1000108
MOOLA ZB, et al (1994) Erwinia chrysanthemi L-asparaginase: epitope mapping and production of antigenically modified enzymes. Biochemical Journal 302(3):921–927
Moreno-Enriquez A, Evangelista-Martinez Z, Gonzalez-Mondragon EG et al (2012) Biochemical characterization of recombinant L-asparaginase (AnsA) from Rhizobium etli, a member of an increasing rhizobial-type family of L-asparaginases. J Microbiol Biotechnol 22:292–300. https://doi.org/10.4014/jmb.1107.07047
Narta UK, Kanwar SS, Azmi W (2007) Pharmacological and clinical evaluation of l-asparaginase in the treatment of leukemia. Crit Rev Oncol Hematol 61:208–221. https://doi.org/10.1016/j.critrevonc.2006.07.009
Orabi HM (2019) L-ASPARAGINASE AND L-GLUTAMINASE: SOURCES, PRODUCTION, AND APPLICATIONS IN MEDICINE AND INDUSTRY. Journal of Microbiology, Biotechnology and Food Sciences 9(2):179–190. https://doi.org/10.15414/jmbfs.2019.9.2.179-190
Orabi HM, El-Fakharany EM, Abdelkhalek ES, Sidkey NM (2019b) L-Asparaginase and L-glutaminase: sources, production, and applications in medicine and industry. J Microbiol Biotechnol Food Sci:179–190. https://doi.org/10.15414/jmbfs.2019.9.2.179-190
Oza VP, Parmar PP, Kumar S, Subramanian RB (2010) Anticancer properties of highly purified l-asparaginase from Withania somnifera L. against acute lymphoblastic leukemia. Appl Biochem Biotechnol 160:1833–1840. https://doi.org/10.1007/s12010-009-8667-z
Pedreschi F, Kaack K, Granby K (2008) The effect of asparaginase on acrylamide formation in French fries. Food Chem 109:386–392. https://doi.org/10.1016/j.foodchem.2007.12.057
Pieters R, Hunger SP, Boos J et al (2011) L-Asparaginase treatment in acute lymphoblastic leukemia. Cancer 117:238–249. https://doi.org/10.1002/cncr.25489
Prakasham RS, Hymavathi M, Subba Rao C et al (2010) Evaluation of antineoplastic activity of extracellular asparaginase produced by isolated Bacillus circulans. Appl Biochem Biotechnol 160:72–80. https://doi.org/10.1007/s12010-009-8679-8
Pritsa AA, Kyriakidis DA (2001) L-asparaginase of Thermus thermophilus: purification, properties and identification of essential amino acids for its catalytic activity. Mol Cell Biochem 216:93–101. https://doi.org/10.1023/A:1011066129771
Richards NGJ, Kilberg MS (2006) Asparagine synthetase chemotherapy. Annu Rev Biochem 75:629–654. https://doi.org/10.1146/annurev.biochem.75.103004.142520
Ruma K, George TK, Aswani P, Jisha MS (2017) Production and optimization of extra cellular L-asparaginase by Fusarium solani Isolated from Withania sominifera. J Biol Act Prod Nat 7:81–88. https://doi.org/10.1080/22311866.2017.1325007
Safary A, Moniri R, Hamzeh-Mivehroud M, Dastmalchi S (2019) Highly efficient novel recombinant L-asparaginase with no glutaminase activity from a new halo-thermotolerant Bacillus strain. Bioimpacts 9:15–23. https://doi.org/10.15171/bi.2019.03
Sakamoto T, Araki C, Beppu T, Arima K (1977) Extracellular asparaginase from Candida utilis, its properties as glycoprotein and antitumor activities. Agric Biol Chem 41:1365–1371. https://doi.org/10.1080/00021369.1977.10862699
Saxena RK, Sinha U (1981) L-asparaginase and glutaminase activities in the culture filtrates of Aspergillus nidulans. CurSci 50: 218–219
Shrivastava A, Khan AA, Shrivastav A et al (2012) Kinetic studies of L-asparaginase from Penicillium digitatum. Prep Biochem Biotechnol 42:574–581. https://doi.org/10.1080/10826068.2012.672943
Siddalingeshwara KG, Lingappa K (2011) Production and characterization of L-asparaginase - a tumour inhibitor. Int J PharmTech Res 3:314–319
Singh Y, Srivastav SK (2013) Statistical and evolutionary optimization for enhanced production of an antileukemic enzyme, L-asparaginase, in a protease-deficient Bacillus aryabhattai ITBHU02 isolated from the soil contaminated with hospital waste. Indian J Exp Biol 51:322–335
Taubeneck U (2007) J. R. NORRIS and D. W. RIBBONS (Editors), Methods in microbiology, Vols. 4-7. Vol. 4: 795 S., £ 11.00; Vol. 5A: 450 S., £ 7.75; Vol. 5B: 695 S., £ 9.50; Vol. 6A: 593 S., £ 9.00; Vol. 6B: 385 S., £ 7.00; Vol. 7A: 479 S., £ 8.50; Vol. 7B: 388 S., £ 7.00. Za. Zeitschrift für allgemeine Mikrobiologie 14:261–261. https://doi.org/10.1002/jobm.19740140317
Thirunavukkarasu N, Suryanarayanan TS, Murali TS et al (2011) L-Asparaginase from marine derived fungal endophytes of seaweeds. Mycosphere 2:147–155. https://doi.org/10.1111/j.1365-313X.2009.03887.x.zation
Ueno T, Ohtawa K, Mitsui K et al (1997) Cell cycle arrest and apoptosis of leukemia cells induced by L-asparaginase. Leukemia 11:1858–1861
Wardani NMS, Osseva N, Suryawan N (2020) Allergic reaction to Escherichia coli-asparaginase in childhood acute lymphoblastic leukemia: a study in a tertiary hospital in Indonesia. Asian J Pharm Clin Res 13:142–146. https://doi.org/10.22159/ajpcr.2020.v13i4.36807
Xu F, Oruna-Concha M, Elmore J (2016) The use of asparaginase to reduce acrylamide levels in cooked food. Food Chem 210:163–171. https://doi.org/10.1016/j.foodchem.2016.04.105
Yadav N, Sarkar S (2014) Production of L-asparaginase by Fusarium oxysporum using submerged fermentation. Int J Pharm Sci Invent 3:32–40
Zhang J-F, Shi L-Y, Wei D-Z (2004) Chemical modification of L-asparaginase from Escherichia coli with a modified polyethyleneglycol under substrate protection conditions. Biotechnol Lett 26:753–756. https://doi.org/10.1023/b:bile.0000024100.49716.3d
Zuo S, Xue D, Zhang T et al (2014) Biochemical characterization of an extremely thermostable L-asparaginase from Thermococcus gammatolerans EJ3. J Mol Catal B Enzym 109. https://doi.org/10.1016/j.molcatb.2014.08.021
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, analysis and investigation, original draft preparation, review, and editing: S.A.A.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 139 kb)
Rights and permissions
About this article
Cite this article
Al Yousef, S.A. Fusarium sp. l-asparaginases: purification, characterization, and potential assessment as an antileukemic chemotherapeutic agent. Environ Sci Pollut Res 29, 11243–11254 (2022). https://doi.org/10.1007/s11356-021-16175-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-16175-5