Abstract
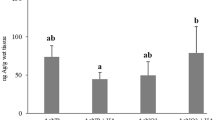
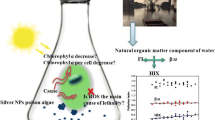
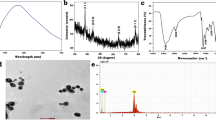
Silver nanoparticles (AgNPs) are one of the most produced nanoproducts due to their unique biocide properties. The natural organic matter has an important impact on nanoparticle’s dispersion as it may alter their fate and transport, as well as their bioavailability and toxicity. Therefore, this study aimed to evaluate the mitigatory effect of humic acids (HAs) on AgNP toxicity. For this purpose, we carried out an ex vivo exposure of gill of Piaractus mesopotamicus fish to 100 μg L−1 of AgNPs or AgNO3, alone and in combination with 10 mg L−1 of HAs. In parallel, a complete AgNP characterization in the media, including the presence of HAs, was provided, and the Ag+ release was measured. We analyzed Ag bioaccumulation, antioxidant enzymes activities, lipid peroxidation, antioxidant capacity against peroxyl radicals, and reduced glutathione levels in fish tissue. Our results indicated the Ag+ release from AgNPs decreased 28% when the HAs were present in the media. The Ag accumulation in gill tissue exposed to AgNPs alone was higher than the AgNO3 exposure, and sixfold higher than the treatment with the HA addition. Moreover, after both Ag forms, the catalase enzyme augmented its activity. However, those responses were mitigated when the HAs were present in the media. Then, our results suggested the mitigation by HAs under the exposure to both Ag forms, providing valuable information about the fate and behavior of this emergent pollutant.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abramenko N, Demidova TB, Krutyakov YA, Zherebin PM, Krysanov EY, Kustov LM, Peijnenburg W (2019) The effect of capping agents on the toxicity of silver nanoparticles to Danio rerio embryos. Nanotoxicology 13(1):1–13. https://doi.org/10.1080/17435390.2018.1498931
Ahmad SA, Das SS, Khatoon A, Ansari MT, Afzal M, Md H, Nayak AK (2020) Bactericidal activity of silver nanoparticles: a mechanistic review. Mater Sci Technol 3:756–769. https://doi.org/10.1016/j.mset.2020.09.002
Amado LL, Garcia ML, Ramos PB, Freitas RF, Zafalon B, Josencler Luis Ribas Ferreira JLR, Yunes JS, Monserrat JM (2009) A method to measure total antioxidant capacity against peroxyl radicals in aquatic organisms: application to evaluate microcystins toxicity. Sci Total Environ 407:2115–2123. https://doi.org/10.1016/j.scitotenv.2008.11.038
Ale A, Bacchetta C, Rossi AS, Galdopórpora J, Desimone MF, de la Torre FR, Gervasio S, Cazenave J (2018a) Nanosilver toxicity in gills of a neotropical fish: metal accumulation, oxidative stress, histopathology, and other physiological effects. Ecotox Environ Safe 148:976–984. https://doi.org/10.1016/j.ecoenv.2017.11.072
Ale A, Rossi AS, Bacchetta C, Gervasio S, de la Torre F, Cazenave J (2018b) Integrative assessment of silver nanoparticles toxicity in Prochilodus lineatus fish. Ecol Indic 93:1190–1198. https://doi.org/10.1016/j.ecolind.2018.06.023
AVMA (American Veterinary Medical Association) (2013) AVMA Guidelines for the Euthanasia of Animals, 2013 Edition. IL, USA.
Bacchetta C, Ale A, Simoniello MF, Gervasio S, Davico C, Rossi AS, Desimone MF, Poletta G, López G, Monserrat JM, Cazenave J (2017) Genotoxicity and oxidative stress in fish after a short-term exposure to silver nanoparticles. Ecol Indic 76:230–239. https://doi.org/10.1016/j.ecolind.2017.01.018
Bacchetta C, López G, Pagano G, Muratt DT, de Carvalho LM, Monserrat JM (2016) Toxicological effects induced by silver nanoparticles in zebra fish (Danio rerio) and in the bacteria communities living at their surface. Bull Environ Contam Toxicol 97:456–462. https://doi.org/10.1007/s00128-016-1883-7
Bacchetta C, Rossi A, Ale A, Campana M, Parma MJ, Cazenave J (2014) Combined toxicological effects of pesticides: a fish multi-biomarker approach. Ecol Indic 36:532–538. https://doi.org/10.1016/j.ecolind.2013.09.016
Banan A, Kalbassi MR, Bahmani M, Sotoudeh E, Johari SA, Ali JM, Kolok AS (2020) Salinity modulates biochemical and histopathological changes caused by silver nanoparticles in juvenile Persian sturgeon (Acipenser persicus). Environ Sci Pollut Res 27:10658–10671. https://doi.org/10.1007/s11356-020-07687-7
Batley GE, Kirby JK, McLaughlin MJ (2013) Fate and Risks of Nanomaterials in Aquatic and Terrestrial Environments. Accounts of Chemical Research 46(3):854–862
Bermejo-Nogales A, Fernández M, Fernández-Cruz ML, Navas JM (2016) Effects of a silver nanomaterial on cellular organelles and time course of oxidative stress in a fish cell line (PLHC-1). Comp Biochem Physiol C 190:54–65. https://doi.org/10.1016/j.cbpc.2016.08.004
Beutler E (1982) Catalase, in: Beutler, E. (Ed.), Red cell metabolism, a manual of biochemical methods. Grune and Stratton Inc., New York, 105-106. Chemosphere 82:308–317. https://doi.org/10.1016/j.chemosphere.2010.10.011
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of proteins utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Britto RS, Longaray Garcia M, Martins da Rocha A, Artigas Flores J, Brant Pinheiro MV, Monserrat JM, Ribas Ferreira JL (2012) Effects of carbon nanomaterials fullerene C60 and fullerol C60(OH)18-22 on gills of fish Cyprinus carpio (Cyprinidae) exposed to ultraviolet radiation. Aquat Toxicol 114-115:80–87. https://doi.org/10.1016/j.aquatox.2012.02.018
Cáceres-Vélez PR, Fascineli ML, Rojas E, Meyer T, Venus T, Koppe Grisolia C, Estrela-Lopis I, Moya S, Morais PC, Bentes Azevedo R (2019) Impact of humic acid on the persistence, biological fate and toxicity of silver nanoparticles: a study in adult zebrafish. Environ Nanotechnol Monit 12:100234. https://doi.org/10.1016/j.enmm.2019.100234
Cáceres-Vélez PR, Fascineli ML, Sousa ME, Koppe Grisolia C, Yate L, Narcizo de Souza PE, Estrela-Lopis I, Moya S, Bentes Azevedo R (2018) Humic acid attenuation of silver nanoparticle toxicity by ion complexation and the formation of a Ag3+ coating. J Hazard Mater 353:173–181. https://doi.org/10.1016/j.jhazmat.2018.04.019
Carrazco-Quevedo A, Römer I, Salamanca MJ, Poynter A, Lynch I, Valsami-Jones E (2019) Bioaccumulation and toxic effects of nanoparticulate and ionic silver in Saccostrea glomerata (rock oyster). Ecotoxicol Environ Saf 179:127–134. https://doi.org/10.1016/j.ecoenv.2019.04.032
Chappell MA, Miller LF, George AJ, Pettway BA, Price CL, Porter BE, Bednar AJ, Seiter JM, Kennedy AJ, Steevens JA (2011) Simultaneous dispersion-dissolution behavior of concentrated silver nanoparticle suspensions in the presence of model organic solutes. Chemosphere 84:1108–1116. https://doi.org/10.1016/j.chemosphere.2011.04.040
Clark NJ, Shaw BJ, Handy RD (2018) Low hazard of silver nanoparticles and silver nitrate to the haematopoietic system of rainbow trout. Ecotox Envion Safe 152:121–131. https://doi.org/10.1016/j.ecoenv.2018.01.030
CONICET (Consejo Nacional de Investigaciones Científicas y Técnicas) (2005). Marco Ético de Referencia para las Investigaciones Biomédicas en Animales de laboratorio, de granja y obtenidos de la naturaleza, Buenos Aires, Argentina. Retrieved from: www.conicet.gov.ar/wp-content/uploads/OCR-RD-20050701-1047.pdf.
Dasari TP, Hwang H-M (2012) The effect of humic acids on the cytotoxicity of silver nanoparticles to a natural aquatic bacterial assemblage. Sci Total Environ 408:5817–5823. https://doi.org/10.1016/j.scitotenv.2010.08.030
de Moraes FD, de Figueiredo JSL, Rossi PA, Venturini FP, Moraes G (2015) Acute toxicity and sublethal effects of phenol on hematological parameters of channel catfish Ictalurus punctatus and pacu Piaractus mesopotamicus. Ecotoxicol Environ Contam 10(1):31–36. https://doi.org/10.5132/eec.2015.01.05
de Souza TAJ, Souza LRR, Franchi LP (2019) Silver nanoparticles: an integrated view of green synthesis methods, transformation in the environment, and toxicity. Ecotoxicol Environ Saf 171:691–700. https://doi.org/10.1016/j.ecoenv.2018.12.095
Deshmukh SP, Patil SM, Mullani SB, Delekar SD (2019) Silver nanoparticles as an effective disinfectant: a review. Mater Sci Eng C 97:954–965. https://doi.org/10.1016/j.msec.2018.12.102
Di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW (2015). InfoStat versión 2015. Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. http://www.infostat.com.ar
Dong B, Liu G, Zhou J, Wang J, Jin R (2020) Transformation of silver ions to silver nanoparticles mediated by humic acid under dark conditions at ambient temperature. J Hazard Mater 383:121190. https://doi.org/10.1016/j.jhazmat.2019.121190
Drotar A, Phelps P, Fall R (1985) Evidence for glutathione peroxidase activities in cultured plant cells. Plant Sci 42:35–40. https://doi.org/10.1016/0168-9452(85)90025-1
Fabrega J, Fawcett SR, Renshaw JC, Lead JR (2009) Silver nanoparticle impact on bacterial growth: effect of pH, concentration, and organic matter. Environ Sci Technol 43(19):7285–7290. https://doi.org/10.1021/es803259g
Gao J, Powers K, Wang Y, Zhou H, Roberts SM, Moudgil BM, Koopman B, Barber DS (2012) Influence of Suwannee River humic acid on particle properties and toxicityof silver nanoparticles. Chemosphere 89:96–101. https://doi.org/10.1016/j.chemosphere.2012.04.024
Gao J, Youn S, Hovsepyan A, Llaneza VL, Wang Y, Bitton G, Jean-Claude JB (2009) Dispersion and toxicity of selected manufactured nanomaterials in natural river water samples: effects of water chemical composition. Environ Sci Technol 43(9):3322–3328. https://doi.org/10.1021/es803315v
Geranio L, Heuberger M, Nowack B (2009) The behavior of silver nanotextiles during washing. Environ Sci Technol 43:8113–8118. https://doi.org/10.1021/es9018332
Gottschalk F, Sun TY, Nowack B (2013) Environmental concentrations of engineered nanomaterials: review of modeling and analytical studies. Environ Pollut 181:287–300. https://doi.org/10.1016/j.envpol.2013.06.003
Govindasamy R, Rahuman AA (2012) Histopathological studies and oxidative stress of synthesized silver nanoparticles in Mozambique tilapia (Oreochromis mossambicus). J Environ Sci 24(6):1091–1098. https://doi.org/10.1016/S1001-0742(11)60845-0
Griffitt RJ, Hyndman K, Denslow ND, Barber DS (2009) Comparison of molecular and histological changes in zebrafish gills exposed to metallic nanoparticles. Toxicol Sci 107(2):404–415. https://doi.org/10.1093/toxsci/kfn256
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first step in mercapturic acid formation. J Biol Chem 249:7130–7139
Halliwell B, Gutteridge JMC (2007) Free radicals in biology and medicine Fourth Ed. In: The Toxicology of Fishes (Eds. Di Giulio RT y Hinton DE). Oxford University, Press New York.
Hayhurst LD, Martin JD, Wallace SJ, Langlois VS, Xenopoulos MA, Metcalfe CD, Rennie MD (2020) Multi-level responses of yellow perch (Perca flavescens) to a whole-lake nanosilver addition study. Arch Environ Contam Toxicol 79:283–297. https://doi.org/10.1007/s00244-020-00764-5
Kataoka C, Ariyoshi T, Kawaguchi H, Nagasakaa S, Kashiwada S (2014) Salinity increases the toxicity of silver nanocolloids to Japanese medaka embryos. Environ Sci Nano 2(1):94–103. https://doi.org/10.1039/C4EN00175C
Johari SA, Kalbassi MR, Lee SB, Dong MS, Yu J (2016) Silver nanoparticles affects the expression of biomarker genes mRNA in rainbow trout (Oncorhynchus mykiss). Comp Clin Pathol 25:85–90. https://doi.org/10.1007/s00580-015-2144-6
Joo HS, Kalbassi MR, Johari SA (2018) Hematological and histopathological effects of silver nanoparticles in rainbow trout (Oncorhynchus mykiss)-how about increase of salinity? Environ Sci Pollut Res 25(16):15449–15461. https://doi.org/10.1007/s11356-018-1663-5
Kalbassi MR, Johari SA, Soltani M, Yu J (2013) Particle size and agglomeration affect the toxicity levels of silvernanoparticle types in aquatic environment. Ecopersia 1(3):273–290
Khan MS, Qureshi NA, Jabeen F, Shakeel M, Asghar MS (2017) Assessment of waterborne amine-coated silver nanoparticle (Ag-NP)-induced toxicity in Labeo rohita by histological and hematological profiles. Biol Trace Elem Res 182:130–139. https://doi.org/10.1007/s12011-017-1080-5
Ki HY, Kim JH, Kwon SC, Jeong SH (2007) A study on multifunctional wool textiles treated with nano-sized silver. J Mater Sci 42:8020–8024. https://doi.org/10.1007/s10853-007-1572-3
Kim JY, Kim K-T, Lee BG, Lim BJ, Kim SD (2013) Developmental toxicity of Japanese medaka embryos by silver nanoparticles and released ions in the presence of humic acid. Ecotoxicol Environ Saf 92:57–63. https://doi.org/10.1016/j.ecoenv.2013.02.004
Kleiven M, Rosseland BO, Teien H-C, Joner EJ, Oughton DH (2018) Route of exposure has a major impact on uptake of silver nanoparticles in Atlantic salmon (Salmo salar). Environ Toxicol Chem 37(11):2895–2903. https://doi.org/10.1002/etc.4251
Kvitek L, Panacek A, Soukupova J, Kolar M, Vecerova R, Prucek R, Holecová M, Zboril R (2008) Effect of surfactants and polymers on stability and antibacterial activity of silver nanoparticles. J Phys Chem C 112(5):5825–5834. https://doi.org/10.1021/jp711616v
Lee B, Duong CN, Cho J, Lee J, Kim K, Seo Y, Kim P, Choi K, Yoon J (2012) Toxicity of citrate-capped silver nanoparticles in Common carp (Cyprinus carpio). J Biomed Biotechnol 262670:1–14. https://doi.org/10.1155/2012/262670
Levard C, Hotze EM, Lowry GV, Brown GEJ (2012) Environmental transformations of silver nanoparticles: impact on stability and toxicity. Environ Sci Technol 46:6900–6914. https://doi.org/10.1021/es2037405
Liu JY, Hurt RH (2010) Ion release kinetics and particle persistence in aqueous nano-silver colloids. Environ Sci Technol 44(6):2169–2175. https://doi.org/10.1021/es9035557
Magesky A, Pelletier E (2018) Cytotoxicity and physiological effects of silver nanoparticles on marine invertebrates. Q. Saquib et al. (eds.), Cellular and Molecular Toxicology of Nanoparticles, Advances in Experimental Medicine and Biology. https://doi.org/10.1007/978-3-319-72041-8_17
Massarsky A, Dupuis L, Taylor J, Eisa-Beygi S, Strek L, Trudeau VL, Moon TW (2013) Assessment of nanosilver toxicity during zebrafish (Danio rerio) development. Chemosphere 92:59–66. https://doi.org/10.1016/j.chemosphere.2013.02.060
Martin JD, Colson T-LL, Langlois VS, Metcalfe CD (2016) Biomarkers of exposure to nanosilver and silver accumulation in yellow perch (Perca flavescens). Environ Toxicol Chem 9999:1–10. https://doi.org/10.1002/etc.3644
Masouleh FF, Amiri BM, Mirvaghefi A, Ghafoori H, Madsen SS (2017) Silver nanoparticles cause osmoregulatory impairment and oxidative stress in Caspian kutum (Rutilus kutum, Kamensky 1901). Environ Monit Assess 189:448. https://doi.org/10.1007/s10661-017-6156-3
Mekkawy IA, Mahmoud UM, Hana MN, Alaa El-Din H, Sayed AE-DH (2019) Cytotoxic and hemotoxic effects of silver nanoparticles on the African Catfish, Clarias gariepinus (Burchell, 1822). Ecotox Environ Safe 171:638–646. https://doi.org/10.1016/j.ecoenv.2019.01.011
McShan D, Ray PC, Yu H (2014) Molecular toxicity mechanism of nanosilver. J Food Drug Anal 22(1):116–127. https://doi.org/10.1016/j.jfda.2014.01.010
Mieiro CL, Martins M, da Silva M, Coelho JP, Lopes CB, Alves da Silva A, Alves J, Pereira E, Pardal M, Costa MH, Pacheco M (2019) Advances on assessing nanotoxicity in marine fish - the pros and cons of combining an ex vivo approach and histopathological analysis in gills. Aquat Toxicol 217:105322. https://doi.org/10.1016/j.aquatox.2019.105322
Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247(10):3170–3175
Mock JJ, Barbic M, Smith DR, Schultz DA, Schultz S (2002) Shape effects in plasmon resonance of individual colloidal silver nanoparticles. J Chem Phys 116:6755–6759. https://doi.org/10.1063/1.1462610
Monserrat JM, Garcia ML, Ventura-Lima J, González M, Ballesteros ML, Miglioranza KSB, Améd MV, Wunderlin DA (2014) Antioxidant, phase II and III responses induced by lipoic acid in the fish Jenynsia multidentata (Anablapidae) and its influence on endosulfan accumulation and toxicity. Pestic Biochem Physiol 108:8–15. https://doi.org/10.1016/j.pestbp.2013.10.009
Nabi MM, Wang J, Meyer M, Croteau M-N, Ismail N, Baalousha M (2021) Concentrations and size distribution of TiO2 and Ag engineered particles in five wastewater treatment plants in the United States. Sci Total Environ 753:142017. https://doi.org/10.1016/j.scitotenv.2020.142017
Navarro E, Piccapetra F, Wagner B, Marconi F, Kaegi R, Odzak N, Sigg L, Behra R (2008) Toxicity of silver nanoparticles to Chlamydomonas reinhardtii. Environ Sci Technol 42(23):8959–8964. https://doi.org/10.1021/es801785m
Parma de Croux MJ (1990) Benzocaine (ethyl-p-aminobenzoate) as an anaesthetic for Prochilodus lineatus, valenciennes (pisces, curimatidae). J Appl Ichthyol 6:189–192. https://doi.org/10.1111/j.1439-0426.1990.tb00578.x
Peter PKJ, Manu N, Chacko J, Nair SM (2012) Synthesis of silver nanoparticles and of self assembled supramolecules from marine humic acid. J Exp Nanosci 9(7):719–727. https://doi.org/10.1080/17458080.2012.702932
Pham T-L (2019) Effect of silver nanoparticles on tropical freshwater and marine microalgae. J Chemother 479:9658386–9658387. https://doi.org/10.1155/2019/9658386
Sampaio FG, Boijink CL, dos Santos LRB, Oba ET, Kalinin AL, Luiz AJ, Rantin FT (2012) Antioxidant defenses and biochemical changes in the neotropical fish pacu, Piaractus mesopotamicus: responses to single and combined copper and hypercarbia exposure. Comp Biochem Physiol C 156:178–186. https://doi.org/10.1016/j.cbpc.2012.07.002
Sardella D, Gatt R, Valdramidis VP (2019) Metal nanoparticles for controlling fungal proliferation: quantitative analysis and applications. Curr Opin Food Sci 30:49–59. https://doi.org/10.1016/j.cofs.2018.12.001
Sharan BR, Nagaraja MS, Kadalli GG, Champa BV (2018). Fourier transform infrared (FTIR) spectroscopy of soil humic and fulvic acids extracted from paddy land use System. Int J Curr Microbiol App Sci 7(5): 834-837. https://doi.org/10.20546/ijcmas.2018.705.102
Schlenk D, Benson WH (2001). New perspectives: toxicology and the environment. Target organ toxicity in marine and freshwater teleosts. Taylor & Francis Group. Boca Raton, FL.372 pp.
Scown TM, Santos EM, Johnston BD, Gaiser B, Baalousha M, Mitov S, Lead JR, Stone V, Fernandes TF, Jepson M, van Aerle R, Tyler CR (2010) Effects of aqueous exposure to silver nanoparticles of different sizes in rainbow trout. Toxicol Sci 115(2):521–534. https://doi.org/10.1093/toxsci/kfq076
Taju G, Abdul Majeed S, Nambi KSN, Sahul Hameed AS (2014) In vitro assay for the toxicity of silver nanoparticles using heart and gill cell lines of Catla catla and gill cell line of Labeo rohita. Comp Biochem Physiol C 161:41–52
Tanaka K, Sano T, Ishizuka K, Kitta K, Kawamura Y (1994) Comparison of properties of leaf and root glutathione reductases from spinach. Physiol Plant 91:353–358. https://doi.org/10.1111/j.1399-3054.1994.tb02960.x
Tao S, Long A, Dawson RW, Xu F, Li B, Cao J, Fang J (2002) Copper speciation and accumulation in the gill microenvironment of carp (Cyprinus carpio) in the presence of kaolin particles. Arch Environ ContamToxicol 42:325–331. https://doi.org/10.1007/s00244-001-0022-5
Vance ME, Kuiken T, Vejerano EP, McGinnis SP, Hochella MF, Rejeski D, Hull MS (2015) Nanotechnology in the real world: redeveloping the nanomaterial consumer products inventory. Beilstein J Nanotechnol 6:1769–1780. https://doi.org/10.3762/bjnano.6.181
Venturino A, Anguiano OL, Gauna L, Cocca C, Bergoc RM, Pechen de D’Angelo AM (2001) Thiols and polyamines in the potentiation of malathion toxicity in larval stages of the toad Bufo arenarum. Comp Biochem Physiol C Toxicol Pharmacol 130(2):191–198. https://doi.org/10.1016/S1532-0456(01)00241-1
Wang H, Qiao X, Chen J, Wang X, Ding S (2005) Mechanisms of PVP in the preparation of silver nanoparticles. Mater Chem Phys 94:449–453. https://doi.org/10.1016/j.matchemphys.2005.05.005
Westerhoff P, Atkinson A, Fortner J, Wong MS, Zimmerman J, Gardea-Torresdey J, Ranville J, Herckes P (2018) Low risk posed by engineered and incidental nanoparticles in drinking water. Nat Nanotechnol 13:661–669. https://doi.org/10.1038/s41565-018-0217-9
Yagi K (1976) A simple fluorometric assay for lipoperoxide in blood plasma. Biochem Med 15(2):212–216. https://doi.org/10.1016/0006-2944(76)90049-1
Yin Y, Yang X, Zhou X, Wang W, Yu S, Liu J, Jiang G (2015a) Water chemistry controlled aggregation and photo-transformation of silver nanoparticles. J Environ Sci 116-125. https://doi.org/10.1016/j.jes.2015.04.005
Yin Y, Shen M, Tan Z, Yu S, Liu J, Jiang G (2015b) Particle coating-dependent interaction of molecular weight fractionated natural organic matter: impacts on the aggregation of silver nanoparticles. Environ Sci Technol 49(11):6581–6589. https://doi.org/10.1021/es5061287
Acknowledgements
The authors thank Gerardo López (Nanotek S.A.) for providing the nanosilver stock solution.
Funding
Universidad Nacional del Litoral (CAID-UNL), CONICET, and Agencia Nacional de Promoción Científica y Técnica PICT 2018-01271.
Author information
Authors and Affiliations
Contributions
AA: formal analysis, investigation, methodology, writing (original draft, review, and editing). JG: methodology, writing (review and editing). MM: methodology. FT: methodology, supervision, resources, writing (review and editing). MD: methodology, supervision, resources, writing (review and editing). JC: conceptualization, funding acquisition, investigation, methodology, resources, writing (review and editing).
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All experiments were performed according to national and institutional guidelines for the protection of animal welfare (CONICET 2005) and approved by the Committee of Ethics and Safety in Experimental Work (Scientific-Technological Center, CONICET, Santa Fe, Argentina).
Consent for publication
Not applicable.
Competing interests
The authors declare competing interests.
Additional information
Responsible editor: Bruno Nunes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ale, A., Galdopórpora, J.M., Mora, M.C. et al. Mitigation of silver nanoparticle toxicity by humic acids in gills of Piaractus mesopotamicus fish. Environ Sci Pollut Res 28, 31659–31669 (2021). https://doi.org/10.1007/s11356-021-12590-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12590-w