Abstract
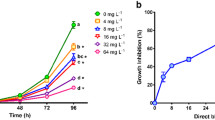
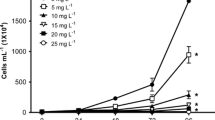
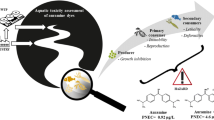
Global consumption of synthetic dyes is roughly 7 × 105 tons per year, of which the textile industry expends about two-thirds. Consumption of synthetic dyes produces large volumes of wastewater discharged into aquatic ecosystems. Colored effluents produce toxic effects in the hydrobionts, reduce light penetration, and alter the photosynthetic activity, causing oxygen depletion, among other effects. Some dyes, such as Congo red (CR), are elaborated with benzidine, a known carcinogenic compound. Information regarding dye toxicity in aquatic ecosystems is scarce; therefore, our study was aimed at evaluating the toxicity of CR on a battery of bioassays: the microalga Pseudokirchneriella subcapitata, the cladocerans Daphnia magna and Ceriodaphnia rigaudi, and the zebrafish Danio rerio. P. subcapitata was the most sensitive species to CR (IC50, 3.11 mg L−1); in exposed individuals, population growth was inhibited, but photosynthetic pigments and macromolecule concentrations were stimulated. D. magna was tolerant to high dye concentrations, the determined LC50 (322.9 mg L−1) is not an environmentally relevant value, but for C. rigaudi, LC50 was significantly lower (62.92 mg L−1). In zebrafish embryos, exposure to CR produced yolk sac edema, skeletal deformities, and stopped larvae hatching; lack of heart beating was the only observed lethal effect. CR affected organisms of different trophic levels diversely. Particularly, the effects observed in microalgae confirm the vulnerability of primary producers to dye-polluted wastewaters, because dyes produced toxic effects and interfered with photosynthesis. Different cladoceran species displayed different acute effects; thus, species sensitivity must also be considered when toxicity of dyes is assessed. Inhibition of fish larvae hatching is a significant effect not previously reported that warns about the toxicity of dyes in fish population dynamics. Synthetic azo colorants should be considered as emerging pollutants because they are discharged into the aquatic environment and are not currently included in the environmental regulation of several countries.





Similar content being viewed by others
References
Acuner A, Dilek FB (2004) Treatment of tectilon yellow 2G by Chlorella vulgaris. Process Biochem 39:623–631. https://doi.org/10.1016/S0032-9592(03)00138-9
Artal A, Albuquerque A, Lotufo G, Umbuzeiro G (2011) The toxicity of commercial dye disperse red 1 and his main fraction to estuarine copepod. In: 32nd Society of Environmental Toxicology and Chemistry (North America) Meeting, Boston, USA. http://www.setac.org/store/download.asp?id=D8FB98CE-3B61-4528-98A3-202D0D761E24
Bae JS, Freeman HS (2007) Aquatic toxicity evaluation of copper-complexed direct dyes to the Daphnia magna. Dyes Pigments 73(1):126–132. https://doi.org/10.1016/j.dyepig.2005.10.019
Bhaskar S, Kuldeep B, Faizal B (2015) Developments in applied phycology 7: algae and environmental sustainability. Springer, New Delhi, p 194. https://doi.org/10.1007/978-81-322-2641-3 ISBN 978-81-322-2639-0
Birhanli A, Ozmen M (2005) Evaluation of the toxicity and teratogenity of six commercial textile dyes using the frog embryo teratogenesis assay-Xenopus. Drug Chem Toxicol 28:51–65. https://doi.org/10.1081/DCT-39689
Blaise C, Férard JF (2005) Small-scale freshwater toxicity investigations. Vol. 1–toxicity test methods. Springer, Berlin, p 422 ISBN 978-1-4020-3120-5
Busquet F, Strecker R, Rawlings JM, Belanger SE, Braunbeck T, Carr GJ, Cenijne P, Fochtmanf P, Gourmelong A, Hübler N, Kleensanga A, Knöbeli M, Kussatz C, Legler J, Lillicrap A, Martínez-Jerónimo F, Polleichtner C, Rzodeczko H, Salinas E, Schneider KE, Scholz S, van den Brandhof E-J, van der Ven LTM, Walter-Rohde S, Weigt S, Witters H, Halder M (2014) OECD validation study to assess intra- and inter-laboratory reproducibility of the zebrafish embryo toxicity test for acute aquatic toxicity testing. Regul Toxicol Pharmacol 69:496–511. https://doi.org/10.1016/j.yrtph.2014.05.018
Buthiyappan A, Aziz ARA, Daud WMAW (2016) Recent advances and prospects of catalytic advanced oxidation process in treating textile effluents. Rev Chem Eng 32(1):1–47. https://doi.org/10.1515/revce-2015-0034
Carfagna S, Lanza N, Salbitani G, Basile A, Sorbo S, Vona V (2013) Physiological and morphological responses of lead or cadmium exposed Chlorella sorokiniana 211-8K (Chlorophyceae). Springerplus 2(1):1–7. https://doi.org/10.1186/2193-1801-2-147
Chen CY, Zhao XQ, Yen HW, Ho SH, Cheng CL, Lee DJ, Bai FW, Chang JS (2013) Microalgae-based carbohydrates for biofuel production. Biochem Eng J 78:1–10. https://doi.org/10.1016/j.bej.2013.03.006
Cheng D, He Q (2014) Assessment of environmental stresses for enhanced microalgal biofuel production – an overview. Front Energy Res 2(26):1–8. https://doi.org/10.3389/fenrg.2014.00026
Cheriaa J, Bettaieb F, Denden I, Bakhrouf A (2009) Characterization of new algae isolated from textile wastewater plant. J Food Agric Environ 7(3&4):700–704
Chia MA, Musa RI (2014) Effect of indigo dye effluent on the growth, biomass, production and phenotypic plasticity of Scenedesmus quadricauda (Chlorococcales). An Acad Bras Cienc 86(1):419–428. https://doi.org/10.1590/0001-3765201420130225
Croce R, Ciná F, Lombardo A, Crispeyn G, Cappelli CI, Vian M, Maiorana S, Benfenati E, Baderna D (2017) Aquatic toxicity of several textile dye formulations: acute and chronic assays with Daphnia magna and Raphidocelis subcapitata. Ecotoxicol Environ Saf 37(10):2145–2151. https://doi.org/10.1016/j.ecoenv.2017.05.046
de Luna LA, Silva TH, Nogueira RF, Kummrow F, Umbuzeiro GA (2014) Aquatic toxicity of dyes before and after photo-Fenton treatment. J Hazard Mater 276:332–338. https://doi.org/10.1016/j.jhazmat.2014.05.047
Du M, Zhang D, Yan C, Zhang X (2012) Developmental toxicity evaluation of three hexabromocyclododecane diastereoisomers on zebrafish embryos. Aquat Toxicol 112–113:1–10. https://doi.org/10.1016/j.aquatox.2012.01.013
Dubois M, Gilles K, Hamilton JK, Rebers PA, Smith F (1951) A colorimetric method for the determination of sugars. Nature 168(4265):167. https://doi.org/10.1038/168167a0
Environment Canada and Health Canada (2009) Screening assessment for the challenge: five disazo dyes
Environment Canada and Health Canada (2013a) Draft screening assesment-aromatic azo and benzidine-based substance grouping-certain azo disperse dyes
Environment Canada and Health Canada (2013b) Draft screening assesment-aromatic azo and benzidine-based substance grouping- certain azo solvent dyes
Ferraz ERA, Umbuzeiro GA, de-Alameida G, Caloto-Oliveira A, Chequer FMD, Zanoni MVB, Dorta DJ, Oliveira DP (2010) Differential toxicity of Disperse Red 1 and Disperse Red 13 in the Ames test, HepG2 cytotoxicity assay, and Daphnia acute toxicity test. Environ Toxicol 26(5):489–497. https://doi.org/10.1002/tox.20576
Ganesh R, Boardman GD, Michelson D (1994) Fate of azo dyes in sludges. Water Res 28:1367–1376. https://doi.org/10.1016/0043-1354(94)90303-4
George B, Pancha I, Desai C, Choskshi K, Paliwal C, Ghosh T, Mishra S (2014) Effects of different media composition, light intensity and photoperiod on morphology and physiology of freshwater microalgae Ankistrodesmus falcatus –a potential strain for bio-fuel production. Bioresour Technol 171:367–374. https://doi.org/10.1016/j.biortech.2014.08.086
Golka K, Kopps S, Myslak WZ (2004) Carcinogenicity of azo colorants: influence of solubility and bioavailability. Toxicol Lett 15:203–210. https://doi.org/10.1016/j.toxlet.2003.11.016
Gu A, Shi X, Yuan C, Ji G, Zhou Y, Long Y, Song L, Wang S, Wang X (2010) Exposure to fenvalerate causes brain impairment during zebrafish development. Toxicol Lett 197:188–192. https://doi.org/10.1016/j.toxlet.2010.05.021
Guo J, Selby K, Boxall ABA (2016) Effects of antibiotics on the growth and physiology of chlorophytes, cyanobacteria and diatom. Arch Environ Contam Toxicol 71(4):589–602. https://doi.org/10.1007/s00244-016-0305-5
Han Y, Wen Q, Chen Z, Li P (2011) Review of methods used for microalgal lipid-content analysis. Energy Procedia 12:944–950. https://doi.org/10.1016/j.egypro.2011.10.124
Hernández-Zamora M, Perales-Vela HV, Flores-Ortiz CM, Cañizares-Villanueva RO (2014) Physiological and biochemical responses of Chlorella vulgaris to Congo Red. Ecotoxicol Environ Saf 108:72–77. https://doi.org/10.1016/j.ecoenv.2014.05.030
Hernández-Zamora M, Cristiani-Urbina E, Martínez-Jerónimo F, Perales-Vela HV, Ponce-Noyola T, Montes-Horcasitas M, Cañizares-Villanueva RO (2015) Bioremoval of the azo dye Congo Red by the microalga Chlorella vulgaris. Environ Sci Pollut Res Int 14(22):10811–10823. https://doi.org/10.1007/s11356-015-4277-1
Hernández-Zamora M, Martínez-Jerónimo F, Cristiani-Urbina E, Cañizares-Villanueva RO (2016) Congo red dye affects survival and reproduction in the cladoceran Ceriodaphnia dubia. Effects of direct and dietary exposure. Ecotoxicology 25(10):1832–1840. https://doi.org/10.1007/s10646-016-1731-x
Heugens EH, Jager T, Creyghton R, Kraak MH, Hendriks AJ, Van Straalen NM, Admiraal W (2003) Temperature-dependent effects of cadmium on Daphnia magna: accumulation versus sensitivity. Environ Sci Technol 37(10):2145–2151
Hill AJ, Teraoka H, Heideman W, Peterson RE (2005) Zebrafish as a model vertebrate for investigating chemical toxicity. Toxicol Sci 86(1):6–19. https://doi.org/10.1093/toxsci/kfi110
International Organization for Standardization (1996) ISO-7346-1: water quality: determination of the acute lethal toxicity of substances to a freshwater fish (Brachidanio rerio Hamilton: Buchanan [Teleostei, Cyprinidae]): part 1: static method. Geneva, Switzerland. http://www.iso.org/iso/catalogue_detail.htm?csnumber=14026
Jang GH, Park IS, Lee SH, Huh TL, Lee YM (2009) Malachite green induces cardiovascular defects in developing zebrafish (Danio rerio) embryos by blocking VEGFR-2 signaling. Biochem Biophys Res Commun 382(3):486–491. https://doi.org/10.1016/j.bbrc.2009.01.118
Jena S, Acharya S, Mohapatra PK (2012) Variation in effects of our OP insecticides on photosynthetic pigment fluorescence of Chlorella Vulgaris Beij. Ecotoxicol Environ Saf 80:111–117. https://doi.org/10.1016/j.ecoenv.2012.02.016
Kant R (2012) Textile dyeing industry an environmental hazard. Nat Sci 4:22–26. https://doi.org/10.4236/ns.2012.41004
Kebede E (1997) Response of Spirulina platensis (=Arthrospira fusiformis) from Lake Chitu, Ethiopia, to salinity stress from sodium salts. J Appl Phycol 9:551–558. https://doi.org/10.1023/A:1007949021786
Lammer E, Carr GJ, Wendler K, Rawlings JM, Belanger SE, Braunbeck T (2009) Is the fish embryo toxicity test (FET) with the zebrafish (Danio rerio) a potential alternative for the fish acute toxicity test? Comp Biochem Physiol C 149:196–209. https://doi.org/10.1016/j.cbpc.2008.11.006
Levy JL, Stauber JL, Jolley DF (2007) Sensitivity of marine microalgae to copper: the effect of biotic factors on copper adsorption and toxicity. Sci Total Environ 387(1–3):141–154. https://doi.org/10.1016/j.scitotenv.2007.07.016
Li J, Liang Y, Zhang X, Lu J, Zhang J, Ruan T, Zhou Q, Jiang G (2011) Impaired gas bladder inflation in zebrafish exposed to a novel heterocyclic brominated flame retardant tris (2, 3-dibromopropyl) isocyanurate. Environ Sci Technol 45(22):9750–9757. https://doi.org/10.1021/es202420g
Lim SL, Chu WL, Phang SM (2010) Use of Chlorella vulgaris for bioremediation of textile wastewater. Bioresour Technol 101(19):7314–7322. https://doi.org/10.1016/j.biortech.2010.04.092
Lowry OH, Rosebrough NJ, Farr A, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mahapatra A, Mishra BG, Hota G (2013) Adsorptive removal of Congo res dye from wastewater by mixed iron oxide-alumina nanocomposites. Ceram Int 39:5443–5441. https://doi.org/10.1016/j.watres.2003.12.009
Marín N, Morales F, Tamigneaux E, Lodeiros C (1998) Effects of nitrate concentrations on the growth and pigment synthesis of a wild strain of Dunaliella salina preadapted to different salinities and cultivated under low illumination. J Appl Phycol 10:405–411. https://doi.org/10.1023/A:1008017928651
Martínez-Jerónimo F, Espinosa-Chávez F, Villaseñor-Córdova R (2000) Effect of culture volume and adult density on the neonate production of Daphnia magna, as test organisms for aquatic toxicity test. Environ Toxicol 15:155–159. https://doi.org/10.1002/1522-7278(2000)
Martínez-Jerónimo F, Rodríguez-Estrada J, Martínez-Jerónimo L (2008) Daphnia exilis Herrick, 1895 (Crustacea: Cladocera). Una especie zooplanctónica potencialmente utilizable como organismo de prueba en bioensayos de toxicidad aguda en ambientes tropicales y subtropicales. Rev Int Contam Ambient 24(4):153–159
Meric S, Selçuk H, Belgiorno V (2005) Acute toxicity removal in textile finishing wastewater by Fenton’s oxidation, ozone and coagulation-flocculation processes. Water Res 39(6):1147–1153. https://doi.org/10.1016/j.watres.2004.12.021
Novotný N, Dias A, Kapanen K, Malachová M, Vándrovcová M, Itävaara N, Lima N (2006) Comparative use of bacterial, algal and protozoan tests to study toxicity of azo and anthraquinone dyes. Chemosphere 63:1436–1442. https://doi.org/10.1016/j.chemosphere.2005.10.002
Ogawa T, Fuj H, Kawai K, Yatome C, Idaka E (1989) Growth inhibition of Bacillus subtilis upon interaction between basic dyes and DNA. Bull Environ Contam Toxicol 42:402–408
Organisation for Economic Co-operation and Development, OECD (2011) Test No. 201: Freshwater Alga and Cyanobacteria, Growth Inhibition Test, OECD Guidelines for the Testing of Chemicals, Section 2, OECD Publishing, Paris. https://doi.org/10.1787/9789264069923-en
Organisation for Economic Co-operation and Development, OECD (2013) Test No. 236: Fish embryo acute toxicity (FET) test. OECD Guidelines for the Testing of Chemicals. Paris, France. https://doi.org/10.1787/9789264203709-en
Parrott JL, Bartlett AJ, Balakrishnan VK (2016) Chronic toxicity of azo and anthracenedione dyes to embryo-larval fathead minnow. Environ Pollut 210:40–47. https://doi.org/10.1016/j.envpol.2015.11.037
Petit AN, Eullaffroy P, Debenest T, Gagne F (2010) Toxicity of PAMAM dendrimers to Chlamydomonas reinhardtii. Aquat Toxicol 100:187–193. https://doi.org/10.1016/j.aquatox.2010.01.019
Pielesz A (1999) The process of the reduction of azo dyes used in dyeing textiles on the basis of infrared spectroscopy analysis. J Mol Struct 511-512:337–344. https://doi.org/10.1016/S0022-2860(99)00176-3
Purkait MK, Maiti A, Das Gupta S, De S (2007) Removal of Congo Red using activated carbon and its regeneration. J Hazard Mater 145(1–2):287–295. https://doi.org/10.1016/j.jhazmat.2006.11.021
Qian HF, Chen W, Sheng GD, Xu XY, Liu WP, Fu ZW (2008) Effects of glufosinate on antioxidant enzymes, subcellular structure, and gene expression in the unicellular green alga Chlorella vulgaris. Aquat Toxicol 88:301–307. https://doi.org/10.1016/j.aquatox.2008.05.009
Qu R, Xu B, Meng L, Wang L, Wang Z (2015) Ozonation of indigo enhanced by carboxylated carbon nanotubes: performance optimization, degradation products, reaction mechanism and toxicity evaluation. Water Res 68:316–327. https://doi.org/10.1016/j.watres.2014.10.017
Rekha RD, Amali AA, Her GM, Yeh YH, Gong HY, Hu SY, Lin GH, Wu JL (2008) Thioacetamide accelerates steatohepatitis, cirrhosis and HCC by expressing HCV core protein in transgenic zebrafish Danio rerio. Toxicology 243(1–2):11–22. https://doi.org/10.1016/j.tox.2007.09.007
Richardson AD, Duigan SP, Berlyn GP (2002) An evaluation of noninvasive methods to estimate foliar chlorophyll content. New Phytol 153(1):185–194. https://doi.org/10.1046/j.0028-646X.2001.00289.x
Rodrigues de Oliveira GA, de Lapuente J, Teixidó E, Porredón C, Borràs M, de Oliveira DP (2016) Textile dyes induce toxicity on zebrafish early life stages. Environ Toxicol Chem 35(2):429–434. https://doi.org/10.1002/etc.3202
Roy R, Fakhruddin ANM, Khatun R, Islam MS, Ahsan MA, Neger AJMT (2010) Characterization of textile industrial effluents and its effects on aquatic macrophytes and algae. Bangladesh J Sci Ind Res 45(1):79–84. https://doi.org/10.3329/bjsir.v45i1.5187
Selderslaghs IWT, Blust R, Witters HE (2012) Feasibility study of the zebrafish assay as an alternative method to screen for developmental toxicity and embryotoxicity using a training set of 27 compounds. Reprod Toxicol 33:142–154. https://doi.org/10.1016/j.reprotox.2011.08.003
Sharma KK, Schuhmann H, Schenk PM (2012) High lipid induction in microalgae for biodiesel production. Energies 5(5):532–1553. https://doi.org/10.3390/en5051532
Soni P, Sharma S, Sharma S, Kumar S, Sharma KP (2006) A comparative study on the toxic effects of textile dye wastewaters (untreated and treated) on mortality and RBC of a freshwater fish Gambusia affinis (Baird and Gerard). J Environ Biol 27:623–628
Sponza DT, Isik M (2005) Toxicity and intermediates of C.I. Direct Red 28 dye through sequential anaerobic/aerobic treatment. Process Biochem 40:2735–2744. https://doi.org/10.1016/j.procbio.2004.12.016
Stein JR (1973) Handbook of phycological methods. Culture Methods and Growth Measurements. Cambridge University Press, London, pp 7–24
US Environmental Protection Agency (2002) Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organism. EPA-821-R-02-012. 5th Ed., Office of Research and Development, Cincinnati, OH. https://www3.epa.gov/region6/water/npdes/wet/wet_methods_manuals/atx.pdf
Vacchi FI, Von der Ohe PC, Albuquerque AF, Vendemiatti JA, Azevedo CC, Honório JG, Silva BF, Zanoni MV, Henry TB, Nogueira AJ, de Umbuzeiro GA (2016) Occurrence and risk assessment of an azo dye - the case of disperse red 1. Chemosphere 156:95–100. https://doi.org/10.1016/j.chemosphere.2016.04.121
Verma Y (2008) Acute toxicity assessment of textile dyes and textile and dye industrial effluents using Daphnia magna bioassay. Toxicol Ind Health 24(7):491–500. https://doi.org/10.1177/0748233710380218
Walthall WK, Stark JD (1999) The acute and chronic toxicity of two xanthene fluorescein sodium salt and phloxine B, to Daphnia pulex. Environ Pollut 104(2):207–215. https://doi.org/10.1016/S0269-7491(98)00189-4
Wellburn AR (1994) The spectral determination of chlorophyll a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313. https://doi.org/10.1016/S0176-1617(11)81192-2
Wen YZ, Chen H, Shen CS, Zhao MR, Liu WP (2011) Enantioselectivity tuning of chiral herbicide dichlorprop by copper: roles of reactive oxygen species. Environ Sci Technol 45:4778–4784. https://doi.org/10.1021/es2003793
Wong K, Liu XJ, Lee AOK, Wong PK (2006) Effect of azo dyes on survivorship, oxygen consumption rate, and filtration rate of the freshwater cladoceran Moina macrocopa. Hum Ecol Risk Assess 12:89–300. https://doi.org/10.1080/10807030500531604
Xu XR, Li HB, Wang WH, Gu JD (2005) Decolorization of dyes and textile wastewater by potassium permanganate. Chemosphere 59:893–898. https://doi.org/10.1016/j.chemosphere.2004.11.013
Xu D, Li C, Chen H, Shao B (2013) Cellular response of freshwater green algae to perfluorooctanoic acid toxicity. Ecotoxicol Environ Saf 88:103–107. https://doi.org/10.1016/j.ecoenv.2012.10.027
Yu WL, Ansari W, Schoepp NG, Hannon MJ, Mayfield SP, Burkart MD (2011) Modifications of the metabolic pathways of lipid and triacylglycerol production in microalgae. Microb Cell Factories 10:91–101. https://doi.org/10.1186/1475-2859-10-91
Zöllner N, Kirsch K (1962) Microdetermination of lipids by the sulphophosphovanillin reaction. Zeitschrift fur die Gesamte Experimentelle Medizin einschliesslich Experimenteller Chirurgie 135:545–561
Acknowledgments
Miriam Hernández-Zamora received a postdoctoral scholarship from the Consejo Nacional de Ciencia y Tecnología, and thanks to the Escuela Nacional de Ciencias Biológicas for providing the support and facilities to conduct the present study. Fernando Martínez-Jerónimo acknowledges the Secretaria de Investigación y Posgrado I. P. N., and the Comisión de Operación y Fomento de Actividades Académicas del I. P. N. for the support provided.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Cinta Porte
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hernández-Zamora, M., Martínez-Jerónimo, F. Congo red dye diversely affects organisms of different trophic levels: a comparative study with microalgae, cladocerans, and zebrafish embryos. Environ Sci Pollut Res 26, 11743–11755 (2019). https://doi.org/10.1007/s11356-019-04589-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-04589-1