Abstract
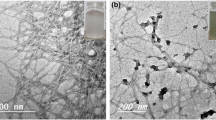
Porous magnesium oxide (MgO) films on carbon fiber paper (CF) have been successfully fabricated in a hydrothermal route at different calcination temperatures. The CF@MgO samples (CF@MgO-300, -400, and -500) show different morphologies with the increasing surface area from 3 for CF to 27 m2 g−1 for CF@MgO-400. Among the four investigated samples, the CF@MgO-400 exhibits the highest phosphate removal ability (~ 1230 mg g−1) with promising applications for the large-scale utilization at low cost.






Similar content being viewed by others
References
Ahmed S, Guo Y, Huang R, Li D, Tang P, Feng Y (2017) Hexamethylene tetramine-assisted hydrothermal synthesis of porous magnesium oxide for high-efficiency removal of phosphate in aqueous solution. J Environ Chem Eng 5:4649–4655 https://doi.org/10.1016/j.jece.2017.09.006
Ahmed S, Guo Y, Li D, Tang P, Feng Y (2018) Superb removal capacity of hierarchically porous magnesium oxide for phosphate and methyl orange. Environ Sci Pollut Res 25:24907–24916. https://doi.org/10.1007/s11356-018-2565-2
Shyla B, Mahadevaiah, Nagendrappa G (2011) A simple spectrohotometric method for the determination of phosphate in soil, detergents, water, bone and food samples through the formation of phosphomolybdate complex followed by its reduction with thiourea. Spectrochem Acta A 78:497–502
Guo L (2007) Doing battle with the green monster of taihu lake. Science 317:1166–1166. https://doi.org/10.1126/science.317.5842.1166
Chowdhury HA, Chowdhury HI, Naskar KM (2015) A facile synthesis of grainy rod-like porous MgO. Mater Lett 158:190–193. https://doi.org/10.1016/j.matlet.2015.05.116
Hiremath V, Trivino MLT, Shavi R, Gebresillase MN, Seo JG (2018) Sacrificial templating method for fabrication of MgO-Al2O3@C spheres and their application to CO2 capture. Mater Lett 211:304–307. https://doi.org/10.1016/j.matlet.2017.10.032
Huang R, He L, Zhang T, Li D, Tang P, Feng Y (2018) Novel carbon paper@magnesium silicate composite porous films: design, fabrication, and adsorption behavior for heavy metal ions in aqueous solution. ACS Appl Mater Interfaces 10:22776–22785. https://doi.org/10.1021/acsami.8b01557
Huang R, Wu M, Zhang T, Li D, Tang P, Feng Y (2017) Template-free synthesis of large-pore-size porous magnesium silicate hierarchical nanostructures for high-efficiency removal of heavy metal ions. ACS Sustain Chem Eng 5:2774–2780. https://doi.org/10.1021/acssuschemeng.7b00140
Kannan N, Sundaram MM (2001) Kinetics and mechanism of removal of methylene blue by adsorption on various carbons—a comparative study. Dyes Pigments 51:25–40 https://doi.org/10.1016/S0143-7208(01)00056-0
Li F, Li H, Wang L, He P, Cao Y (2015) Magnesium oxide nanosheets as effective catalysts for the synthesis of diethyl carbonate from ethyl carbamate and ethanol. Catal Sci Technol 5:1021–1034. https://doi.org/10.1039/C4CY01109K
Luo Y, Jiang J, Zhou W, Yang H, Luo J, Qi X, Zhang H, Yu DYW, Li CM, Yu T (2012) Self-assembly of well-ordered whisker-like manganese oxide arrays on carbon fiber paper and its application as electrode material for supercapacitors. J Mater Chem 22:8634–8640. https://doi.org/10.1039/C2JM16419A
Pan B, Han F, Nie G, Wu B, He K, Lu L (2014) New strategy to enhance phosphate removal from water by hydrous manganese oxide. Environ Sci Technol 48:5101–5107. https://doi.org/10.1021/es5004044
Sharma L, Kakkar R (2017) Hierarchical porous magnesium oxide (Hr-MgO) microspheres for adsorption of an organophosphate pesticide: kinetics, isotherm, thermodynamics, and DFT studies. ACS Appl Mater Interfaces 9:38629–38642. https://doi.org/10.1021/acsami.7b14370
Sing KSW (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl Chem 57:603–619. https://doi.org/10.1351/pac198557040603
Tian P, Han X, Ning G, Fang H, Ye J, Gong W, Lin Y (2013) Synthesis of porous hierarchical MgO and its superb adsorption properties. ACS Appl Mater Interfaces 5:12411–12418. https://doi.org/10.1021/am403352y
Guo W, Zhang F, Lin C, Lin WZ (2012) Direct growth of TiO2 nanosheet arrays on carbon fibers for highly efficient photocatalytic degradation of methyl orange. Adv Mater 24:4761–4764. https://doi.org/10.1002/adma.201201075
Xiong L, Lan D, Liang H, Chen L, Wang Q (2018) Grafting TiO2 nanoparticles onto carbon fiber via “thiol-ene” click chemistry and its photodegradation performance for methyl orange. Mater Lett 211:296–299. https://doi.org/10.1016/j.matlet.2017.10.028
Yan Z, Xingming G, Feng W et al (2016) Mesocarbon microbead carbon-supported magnesium hydroxide nanoparticles: turning spent Li-ion battery anode into a highly efficient phosphate adsorbent for wastewater treatment. ACS Appl Mater Interfaces 8:21315–21325. https://doi.org/10.1021/acsami.6b05458
Funding
This work is financially supported by the National Key R & D Program of China (No. 2016YFB0301600) and the National Natural Science Foundation of China. S. Ahmed received financial funding from the Chinese Scholarship Council.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Rights and permissions
About this article
Cite this article
Ahmed, S., Ashiq, M.N., Li, D. et al. Carbon fiber paper@MgO films: in situ fabrication and high-performance removal capacity for phosphate anions. Environ Sci Pollut Res 25, 34788–34792 (2018). https://doi.org/10.1007/s11356-018-3410-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-3410-3