Abstract
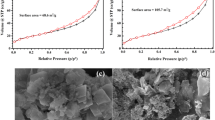
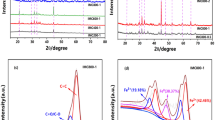
This study reports the eco-friendly preparation of a novel composite material consisting of red mud and carbon spheres, denoted as red mud@C composite, and its application for the removal of 2,4-dichlorophenoxyacetic acid herbicide (2,4-D) from aqueous solution. The preparation route has a green approach because it follows the low-energy consuming one-step hydrothermal process by using starch as a renewable carbon precursor and red mud as a waste from aluminum production industry. Characterization of the red mud@C composite was performed by FT-IR, TGA, SEM, TEM, BET, XRD, and Raman microscopy analyses. The batch adsorption studies revealed that the red mud@C composite has higher 2,4-D adsorption efficiency than those of the red mud and the naked carbon spheres. The maximum removal at initial pH of 3.0 is explained by considering the pKa of 2,4-D and pH of point of zero charge (pHpzc) of the composite material. The adsorption equilibrium time was 60 min, which followed the pseudo-second-order kinetic model together with intra-particle diffusion model. The isotherm analysis indicated that Freundlich isotherm model better represented the adsorption data, with isotherm parameters of k [15.849 (mg/g) (mg/L)−1/n] and n (2.985). The prepared composite is reusable at least 5 cycles of adsorption-desorption with no significant decrease in the adsorption capacity.








Similar content being viewed by others
References
Ai L, Li L (2013) Efficient removal of organic dyes from aqueous solution with ecofriendly biomass-derived carbon@montmorillonite nanocomposites by one-step hydrothermal process. Chem Eng J 223:688–695. doi:10.1016/j.cej.2013.03.015
Aksu Z, Kabasakal E (2004) Batch adsorption of 2,4-dichlorophenoxy-acetic acid (2,4-D) from aqueous solution by granular activated carbon. Sep Purif Technol 35:223–240. doi:10.1016/S1383-5866(03)00144-8
Aksu Z, Kabasakal E (2005) Adsorption characteristics of 2,4-dichlorophenoxyacetic acid (2,4-D) from aqueous solution on powdered activated carbon. J Environ Sci Heal Part B 40:545–570. doi:10.1081/PFC-200061533
Altundoğan HS, Altundoğan S, Tümen F, Bildik M (2002) Arsenic adsorption from aqueous solutions by activated red mud. Waste Manag 22:357–363. doi:10.1016/S0956-053X(01)00041-1
Apak R, Güçlü K, Turgut MH (1998) Modeling of copper(II), cadmium(II), and lead(II) adsorption on red mud. J Colloid Interface Sci 203:122–130. doi:10.1006/jcis.1998.5457
Atasoy A (2005) An investigation on characterization and thermal analysis of the Aughinish red mud. J Therm Anal Calorim 81:357–361. doi:10.1007/s10973-005-0792-5
Atasoy A (2011) Reduction of ferric oxides in the red mud by the aluminıothermic process. In: 6th international advanced technologies symposium. pp 16–18
Badellino C, Rodrigues CA, Bertazzoli R (2006) Oxidation of pesticides by in situ electrogenerated hydrogen peroxide: study for the degradation of 2,4-dichlorophenoxyacetic acid. J Hazard Mater 137:856–864. doi:10.1016/j.jhazmat.2006.03.035
Ballesteros Martín MM, Sánchez Pérez JA, Casas López JL et al (2009) Degradation of a four-pesticide mixture by combined photo-Fenton and biological oxidation. Water Res 43:653–660. doi:10.1016/j.watres.2008.11.020
Banerjee A, Gokhale R, Bhatnagar S et al (2012) MOF derived porous carbon–Fe3O4 nanocomposite as a high performance, recyclable environmental superadsorbent. J Mater Chem 22:19694. doi:10.1039/c2jm33798c
Cengeloglu Y, Tor A, Ersoz M, Arslan G (2006) Removal of nitrate from aqueous solution by using red mud. Sep Purif Technol 51:374–378. doi:10.1016/j.seppur.2006.02.020
Cengeloglu Y, Tor A, Arslan G et al (2007) Removal of boron from aqueous solution by using neutralized red mud. J Hazard Mater 142:412–417. doi:10.1016/j.jhazmat.2006.08.037
Chen L-F, Liang H-W, Lu Y et al (2011) Synthesis of an attapulgite clay@carbon nanocomposite adsorbent by a hydrothermal carbonization process and their application in the removal of toxic metal ions from water. Langmuir 27:8998–9004. doi:10.1021/la2017165
Deng S, Ma R, Yu Q et al (2009) Enhanced removal of pentachlorophenol and 2,4-D from aqueous solution by an aminated biosorbent. J Hazard Mater 165:408–414. doi:10.1016/j.jhazmat.2008.10.029
Deokar SK, Mandavgane SA (2015) Rice husk ash for fast removal of 2,4-dichlorophenoxyacetic acid from aqueous solution. Adsorpt Sci Technol 33:429–440. doi:10.1260/0263-6174.33.5.429
dos Santos DC, Adebayo MA, de Fátima Pinheiro Pereira S et al (2014) New carbon composite adsorbents for the removal of textile dyes from aqueous solutions: kinetic, equilibrium, and thermodynamic studies. Korean J Chem Eng 31:1470–1479. doi:10.1007/s11814-014-0086-3
Dupont L, Guillon E (2003) Removal of hexavalent chromium with a lignocellulosic substrate extracted from wheat bran. Environ Sci Technol 37:4235–4241. doi:10.1021/ES0342345
Feng Z, Liu D, Ma X (2016) The rectorite/carbon composites: fabrication, modification and adsorption. Chemosphere 144:621–627. doi:j.chemosphere.2015.09.032
Freundlich HMF (1906) Über die Adsorption in Lösungen. Z Phys Chem 385–470
Genç H, Tjell JC, McConchie D, Schuiling O (2003) Adsorption of arsenate from water using neutralized red mud. J Colloid Interface Sci 264:327–334. doi:10.1016/S0021-9797(03)00447-8
Genç-Fuhrman H, Tjell JC, McConchie D (2004) Increasing the arsenate adsorption capacity of neutralized red mud (Bauxsol). J Colloid Interface Sci 271:313–320. doi:10.1016/j.jcis.2003.10.011
Gerçel Ö, Özcan A, Özcan AS, Gerçel HF (2007) Preparation of activated carbon from a renewable bio-plant of Euphorbia rigida by H2SO4 activation and its adsorption behavior in aqueous solutions. Appl Surf Sci 253:4843–4852. doi:10.1016/j.apsusc.2006.10.053
Gong J, Liu T, Wang X et al (2011) Efficient removal of heavy metal ions from aqueous systems with the assembly of anisotropic layered double hydroxide nanocrystals@carbon nanosphere. Environ Sci Technol 45:6181–6187. doi:10.1021/es200668q
Gupta VK, Ali I, Suhas SVK (2006) Adsorption of 2,4-D and carbofuran pesticides using fertilizer and steel industry wastes. J Colloid Interface Sci 299:556–563. doi:10.1016/j.jcis.2006.02.017
Hameed BH, Salman JM, Ahmad AL (2009) Adsorption isotherm and kinetic modeling of 2,4-D pesticide on activated carbon derived from date stones. J Hazard Mater 163:121–126. doi:10.1016/j.jhazmat.2008.06.069
Hema M, Arivoli S (2007) International journal of physical sciences. Int J Phys Sci 2:10–17
Ho Y-S (2004) Selection of optimum sorption isotherm. Carbon N Y 42:2115–2116
Ho Y-S, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465. doi:10.1016/S0032-9592(98)00112-5
Horikoshi S, Hidaka H, Serpone N (2003) Environmental remediation by an integrated microwave/UV-illumination technique: IV. Non-thermal effects in the microwave-assisted degradation of 2,4-dichlorophenoxyacetic acid in UV-irradiated TiO2/H2O dispersions. J Photochem Photobiol A Chem 159:289–300. doi:10.1016/S1010-6030(03)00185-0
Kuśmierek K, Szala M, Świątkowski A (2016) Adsorption of 2,4-dichlorophenol and 2,4-dichlorophenoxyacetic acid from aqueous solutions on carbonaceous materials obtained by combustion synthesis. J Taiwan Inst Chem Eng 63:371–378. doi:10.1016/j.jtice.2016.03.036
Lagergren S (1898) Zur theorie der sogenannten absorption... - Google Akademik. K K Sven Vetenskapsad Handl 24:1–39
Landreau X, Lanfant B, Merle T et al (2012) A thorough FT-IR spectroscopy study on micrometric silicon oxide films deposited by atmospheric pressure microwave plasma torch. Eur Phys J D 66:160. doi:10.1140/epjd/e2012-20647-x
Langmuir I (1916) The constitution and fundamental properties of solids and liquids. Part I Solids J Am Chem Soc 38:2221–2295. doi:10.1021/ja02268a002
Lee J, Kim J, Hyeon T (2006) Recent progress in the synthesis of porous carbon materials. Adv Mater 18:2073–2094. doi:10.1002/adma.200501576
Li T, Shen J, Li N, Ye M (2012) Facile and novel hydrothermal preparation of functionalised carbon microspheres from glucose by using graphene sheets as a substrate. Mater Lett 89:202–205. doi:10.1016/j.matlet.2012.08.132
Lim X, Sanna A, Andrésen JM (2014) Influence of red mud impregnation on the pyrolysis of oil palm biomass-EFB. Fuel 119:259–265. doi:10.1016/j.fuel.2013.11.057
Liu Y, Cai Q, Li H, Zhang J (2013) Fabrication and characterization of mesoporous carbon nanosheets using halloysite nanotubes and polypyrrole via a template-like method. J Appl Polym Sci 128:517–522. doi:10.1002/app.38208
Liu Y, Naidu R, Ming H et al (2016) Effects of thermal treatments on the characterisation and utilisation of red mud with sawdust additive. Waste Manag Res 34:518–526. doi:10.1177/0734242X16634197
López-Ayala S, Rincón ME, Quiroz Alfaro MA et al (2015) Nanocrystalline titania xerogels doped by metal precursors in the photocatalytic degradation of 2,4-D sodium salts. J Photochem Photobiol A Chem 311:166–175. doi:10.1016/j.jphotochem.2015.06.019
Mckay G (1984) The adsorption of basic dye onto silica from aqueous solution-solid diffusion model. Chem Eng Sci 39:129–138. doi:10.1016/0009-2509(84)80138-4
Nath H, Sahoo A (2014) A study on the characterization of red mud. Int J Appl bio-engineering 8:1–4. doi:10.18000/ijabeg.10118
Nidheesh PV, Gandhimathi R, Ramesh ST, Amatha Singh TS (2012) Kinetic analysis of crystal violet adsorption on to bottom ash. Turkish J Eng Env Sci 36:249–262. doi:10.3906/muh-1110-3
Ozcan S, Tor A, Aydin ME (2011) Removal of organochlorine pesticides from aqueous solution by using neutralized red mud. CLEAN - Soil, Air, Water 39:972–979. doi:10.1002/clen.201000596
Pirsaheb M, Dargahi A, Hazrati S, Fazlzadehdavil M (2014) Removal of diazinon and 2,4-dichlorophenoxyacetic acid (2,4-D) from aqueous solutions by granular-activated carbon. Desalin Water Treat 52:4350–4355. doi:10.1080/19443994.2013.801787
Pradhan J, Das J, Das S, Thakur RS (1998) Adsorption of phosphate from aqueous solution using activated red mud. J Colloid Interface Sci 204:169–172. doi:10.1006/jcis.1998.5594
Rafatullah M, Sulaiman O, Hashim R, Ahmad A (2010) Adsorption of methylene blue on low-cost adsorbents: a review. J Hazard Mater 177:70–80. doi:10.1016/j.jhazmat.2009.12.047
Redlich O, Peterson DL (1959) A useful adsorption isotherm. J Phys Chem 63:1024–1024. doi:10.1021/j150576a611
Ruiz de Arcaute C, Soloneski S, Larramendy ML (2016) Toxic and genotoxic effects of the 2,4-dichlorophenoxyacetic acid (2,4-D)-based herbicide on the Neotropical fish Cnesterodon decemmaculatus. Ecotoxicol Environ Saf 128:222–229. doi:10.1016/j.ecoenv.2016.02.027
Sarkar B, Liu E, McClure S et al (2015) Biomass derived palygorskite–carbon nanocomposites: synthesis, characterisation and affinity to dye compounds. Appl Clay Sci 114:617–626. doi:10.1016/j.clay.2015.07.001
Sevilla M, Fuertes AB (2009) Chemical and structural properties of carbonaceous products obtained by hydrothermal carbonization of saccharides. Chem - A Eur J 15:4195–4203. doi:10.1002/chem.200802097
Sglavo VM, Campostrini R, Maurina S et al (2000) Bauxite “red mud” in the ceramic industry. Part 1: thermal behaviour. J Eur Ceram Soc 20:235–244. doi:10.1016/S0955-2219(99)00088-6
Song X, Boily J-F (2016) Surface and bulk thermal dehydroxylxation of FeOOH polymorphs. J Phys Chem A 120:6249–6257. doi:10.1021/acs.jpca.6b04294
Szymański GS, Karpiński Z, Biniak S, Świątkowski A (2002) The effect of the gradual thermal decomposition of surface oxygen species on the chemical and catalytic properties of oxidized activated carbon. Carbon N Y 40:2627–2639. doi:10.1016/S0008-6223(02)00188-4
Tang L, Zhang S, Zeng G-M et al (2015) Rapid adsorption of 2,4-dichlorophenoxyacetic acid by iron oxide nanoparticles-doped carboxylic ordered mesoporous carbon. J Colloid Interface Sci 445:1–8. doi:10.1016/j.jcis.2014.12.074
Tang J, Mu B, Wang W et al (2016) Fabrication of manganese dioxide/carbon/attapulgite composites derived from spent bleaching earth for adsorption of Pb(II) and brilliant green. RSC Adv 6:36534–36543. doi:10.1039/C5RA26362J
Tor A, Cengeloglu Y (2006) Removal of Congo red from aqueous solution by adsorption onto acid activated red mud. J Hazard Mater 138:409–415. doi:10.1016/j.jhazmat.2006.04.063
Tor A, Cengeloglu Y, Aydin ME, Ersoz M (2006) Removal of phenol from aqueous phase by using neutralized red mud. J Colloid Interface Sci 300:498–503. doi:10.1016/j.jcis.2006.04.054
Ünlü N, Ersoz M (2006) Adsorption characteristics of heavy metal ions onto a low cost biopolymeric sorbent from aqueous solutions. J Hazard Mater 136:272–280. doi:10.1016/j.jhazmat.2005.12.013
Unur E (2013) Functional nanoporous carbons from hydrothermally treated biomass for environmental purification. Microporous Mesoporous Mater 168:92–101. doi:10.1016/j.micromeso.2012.09.027
US EPA (2012) 2012 Edition of the Drinking Water Standards and Health Advisories (EPA 822-S-12-001). pp 1–12
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng 89:31–59
WHO (2009) The WHO Recommended Classification of Pesticides by Hazard and Guidelines to Classification 2009
Wu X, Gao P, Zhang X et al (2014) Synthesis of clay/carbon adsorbent through hydrothermal carbonization of cellulose on palygorskite. Appl Clay Sci 95:60–66. doi:10.1016/j.clay.2014.03.010
Xu YH, Cai QQ, Ma HX et al (2013) Optimisation of electrocatalytic dechlorination of 2,4-dichlorophenoxyacetic acid on a roughened silver–palladium cathode. Electrochim Acta 96:90–96. doi:10.1016/j.electacta.2013.02.068
Zhang Z, Kong J (2011) Novel magnetic Fe3O4@C nanoparticles as adsorbents for removal of organic dyes from aqueous solution. J Hazard Mater 193:325–329. doi:10.1016/j.jhazmat.2011.07.033
Zhang W, Mu B, Wang A, Shao S (2014) Attapulgite oriented carbon/polyaniline hybrid nanocomposites for electrochemical energy storage. Synth Met 192:87–92. doi:10.1016/j.synthmet.2014.03.021
Zhou J, Tang C, Cheng B et al (2012) Rattle-type carbon–alumina core–shell spheres: synthesis and application for adsorption of organic dyes. ACS Appl Mater Interfaces 4:2174–2179. doi:10.1021/am300176k
Acknowledgements
The authors are grateful to the Scientific Research Projects of Necmettin Erbakan University (151219003) for the financial support.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. All authors contributed equally.
Corresponding author
Additional information
Responsible editor: Guilherme L. Dotto
Electronic supplementary material
ESM 1
Supporting Information. XRD patterns of the materials, the removal efficiencies, the graphs of kinetic model, diffusion models, Langmuir, Freundlich, and Redlich-Peterson isotherms, and the changes in adsorption capacity of regenerated red mud@C composite.(DOCX 1257 kb)
Rights and permissions
About this article
Cite this article
Kazak, O., Eker, Y.R., Akin, I. et al. Green preparation of a novel red mud@carbon composite and its application for adsorption of 2,4-dichlorophenoxyacetic acid from aqueous solution. Environ Sci Pollut Res 24, 23057–23068 (2017). https://doi.org/10.1007/s11356-017-9937-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9937-x