Abstract
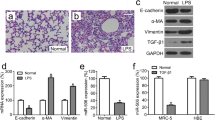
This research is designed to investigate the regulatory effect of miR-18a to the target gene connective tissue growth factor (CTGF, or CCN2), by participating in TGF-β1 signaling pathway and explore the pathogenic mechanism of miR-18a in pulmonary injury induced by nano-SiO2 based on our early study. miR-18a and expression of TGF-β1 in Chinese hamster lung (CHL) fibroblasts cells stimulated by supernatants of NR8383 cells exposed to 40 μg/ml nano-SiO2 for 24 h demonstrated 1.58 ± 0.22-fold and 1096.00 ± 2.60 pg/ml increase compared with blank control group analyzed by real-time quantitative PCR (RT-qPCR) and enzyme-linked immunosorbent assay (ELISA), respectively. Expression increase of miR-18a and reduction of CCN2 mRNA expression levels and protein gray value ratio detected by Western blotting in CHL cells transfect miR-18a mimics for 48 h. The reverse of CHL cell transfection miR-18a inhibit is also true. The result of miR-18a and CCN2 binding sites tested by luciferase reporter gene assay shows that the report relative fluorescence value of miR-18a mimics wild type on CCN2 is 0.50 ± 0.02 with the control of mimics NC and mutant relative fluorescence report value 0.86 ± 0.04 (P < 0.05). Expression levels of miR-18a, CCN2 mRNA, and protein gray value ratio decreased in CHL cells treated by TGF-β1, respectively, and vice versa treated by TGF-β1corepressor. The results suggest that CCN2 is the target gene regulated by miR-18a and miR-18a participates in TGF-β1 signaling pathway by regulating the expression of CCN2 negatively through CCN2 3’UTR site, and thus may be involved in the development process of pulmonary injury.










Similar content being viewed by others
References
Akhtar MJ, Ahamed M, Kumar S, Siddiqui H, Patil G, Ashquin M, Ahmad I (2010) Nanotoxicity of pure silica mediated through oxidant generation rather than glutathione depletion in human lung epithelial cells. Toxicology 276(2):95–102. https://doi.org/10.1016/j.tox.2010.07.010
Asano Y (2010) Future treatments in systemic sclerosis. J Dermatol 37(1):54–70. https://doi.org/10.1111/j.1346-8138.2009.00758.x
Basheerudeen SAS, Mani C, Kulkarni MAK, Pillai K, Rajan A, Venkatachalam P (2013) Human brain glioblastoma cells do not induce but do respond to the bleomycin-induced bystander response from lung adenocarcinoma cells. Mutat Res 757(2):114–119. https://doi.org/10.1016/j.mrgentox.2013.07.005
Bonauer A, Dimmeler S (2009) The microRNA-17-92 cluster: still a miracle? Cell Cycle 8(23):3866–3873. https://doi.org/10.4161/cc.8.23.9994
Brown DM, Kanase N, Gaiser B, Johnston H, Stone V (2014) Inflammation and gene expression in the rat lung after instillation of silica nanoparticles: effect of size, dispersion medium and particle surface charge. Toxicol Lett 224(1):147–156. https://doi.org/10.1016/j.toxlet.2013.10.019
Chen CM, Chou HC, Huang LT (2015) Maternal nicotine exposure during gestation and lactation induces kidney injury and fibrosis in rat offspring. Pediatr Res 77(1–1):56–63. https://doi.org/10.1038/pr.2014.148
Cho WS, Duffin R, Poland CA, Howie SE, MacNee W, Bradley M, Megson IL, Donaldson K (2010) Metal oxide nanoparticles induce unique inflammatory footprints in the lung: important implications for nanoparticle testing. Environ Health Perspect 118(12):1699–1706. https://doi.org/10.1289/ehp.1002201
Choi M, Cho WS, Han BS, Cho M, Kim SY, Yi JY, Ahn B, Kim SH, Jeong J (2008) Transient pulmonary fibrogenic effect induced by intratracheal instillation of ultrafine amorphous silica in A/J mice. Toxicol Lett 182(1–3):97–101. https://doi.org/10.1016/j.toxlet.2008.08.019
Coccini T, Barni S, Vaccarone R, Mustarelli P, Manzo L, Roda E (2013) Pulmonary toxicity of instilled cadmium-doped silica nanoparticles during acute and subacute stages in rats. Histol Histopathol 28(2):195–209
Crespo I, San-Miguel B, Fernández A, Ortiz de Urbina J, González-Gallego J, Tuñón MJ (2015) Melatonin limits the expression of profibrogenic genes and ameliorates the progression of hepatic fibrosis in mice. Transl Res 165(2):346–357. https://doi.org/10.1016/j.trsl.2014.10.003
Cushing L, Kuang P, Lü J (2014) The role of miR-29 in pulmonary fibrosis. Biochem Cell Biol 93(999):1–10. https://doi.org/10.1139/bcb-2014-0095
Cushing L, Kuang PP, Qian J, Shao F, Wu J, Little F, Thannickal VJ, Cardoso WV, Lü J (2011) miR-29 is a major regulator of genes associated with pulmonary fibrosis. Am J Respir Cell Mol Biol 45(2):287–294. https://doi.org/10.1165/rcmb.2010-0323OC
De Winter P, Leoni P, De Winter P (2008) Connective tissue growth factor: structure-function relationships of a mosaic, multifunctional protein. Growth Factors 26(2):80–91. https://doi.org/10.1080/08977190802025602
Downs TR, Crosby ME, Hu T, Kumar S, Sullivan A, Sarlo K, Reeder B, Lynch M, Wagner M, Mills T, Pfuhler S (2012) Silica nanoparticles administered at the maximum tolerated dose induce genotoxic effects through an inflammatory reaction while gold nanoparticles do not. Mutat Res 745(1–2):38–50. https://doi.org/10.1016/j.mrgentox.2012.03.012
Fox JL, Dews M, Minn AJ, Thomas-Tikhonenko A (2013) Targeting of TGFβ signature and its essential component CTGF by miR-18 correlates with improved survival in glioblastoma. RNA 19(2):177–190. https://doi.org/10.1261/rna.036467.112
Frazier K, Williams S, Kothapalli D, Klapper H, Grotendorst GR (1996) Stimulation of fibroblast cell growth, matrix production, and granulation tissue formation by connective tissue growth factor. J Invest Dermatol 107(3):404–411
Guo MC, Xu XL, Yan XC, Wang S, Gao S, Zhu S (2013) In vivo biodistribution and synergistic toxicity of silica nanoparticles and cadmium chloride in mice. J Hazard Mater 260:780–788. https://doi.org/10.1016/j.jhazmat.2013.06.040
He L, Thomson JM, Hemann MT, Hernando-Monge E, Mu D, Goodson S, Powers S, Cordon-Cardo C, Lowe SW, Hannon GJ, Hammond SM (2005) A microRNA polycistron as a potential human oncogene. Nature 435(7043):828–833. https://doi.org/10.1038/nature03552
He QJ, Zhang ZW, Gao F, Li Y, Shi J (2011) In vivo biodistribution and urinary excretion of mesoporous silica nanoparticles: effects of particle size and PEGylation. Small 7(2):271–280. https://doi.org/10.1002/smll.201001459
He Y, Huang C, Lin X, Li J (2013) MicroRNA-29 family, a crucial therapeutic target for fibrosis diseases. Biochimie 95(7):1355–1359. https://doi.org/10.1016/j.biochi.2013.03.010
Hwang J, Chang C, Kim JH, Oh CT, Lee HN, Lee C, Oh D, Lee C, Kim B, Hong SW, Lee DK (2016) Development of cell-penetrating asymmetric interfering RNA targeting connective tissue growth factor (CTGF). J Invest Dermatol. https://doi.org/10.1016/j.jid.2016.06.626
Jin M, Wu Y, Wang L, Zang B, Tan L (2016) Hydroxysafflor yellow A attenuates bleomycin-induced pulmonary fibrosis in mice. Phytother Res 30(4):577–587. https://doi.org/10.1002/ptr.5560
Jin Y, Kannan S, Wu M, Zhao JX (2007) Toxicity of luminescent silica nanoparticles to living cells. Chem Res Toxicol 20(8):1126–1133. https://doi.org/10.1021/tx7001959
Kubota S, Takigawa M (2015) Cellular and molecular actions of CCN2/CTGF and its role under physiological and pathological conditions. Clin Sci (Lond) 128(3):181–196
Leask A, Parapuram SK, Shi-Wen X, Abraham DJ (2009) Connective tissue growth factor (CTGF, CCN2) gene regulation: a potent clinical bio-marker of fibroproliferative disease? J Cell Commun Signal 3(2):89–94. https://doi.org/10.1007/s12079-009-0037-7
Lin Z, Ma L, Zhu-ge X, Zhang H, Lin B (2013) A comparative study of lung toxicity in rats induced by three types of nanomaterials. Nanoscale Res Lett 8(1):521–521. https://doi.org/10.1186/1556-276X-8-521
Liu G, Friggeri A, Yang Y, Milosevic J, Ding Q, Thannickal VJ, Kaminski N, Abraham E (2010) miR-21 mediates fibrogenic activation of pulmonary fibroblasts and lung fibrosis. J Exp Med 207(8):1589–1597. https://doi.org/10.1084/jem.20100035
Liu J, Song C, Xiao Q, Hu G, Tao L, Meng J (2015) Fluorofenidone attenuates TGF-β1-induced lung fibroblast activation via restoring the expression of caveolin-1. Shock 43(2):201–207. https://doi.org/10.1097/SHK.0000000000000273
Maher TM, Wells AU, Laurent GJ (2007) Idiopathic pulmonary fibrosis: multiple causes and multiple mechanisms. Eur Respir J 30(5):835–839. https://doi.org/10.1183/09031936.00069307
Marshall RP, Gohlke P, Chambers RC, Howell DC, Bottoms SE, Unger T, McAnulty RJ, Laurent GJ (2004) Angiotensin II and the fibroproliferative response to acute lung injury. Am J Physiol Lung Cell Mol Physiol 286(1):L156–L164. https://doi.org/10.1152/ajplung.00313.2002
Moore BB, Hogaboam CM (2008) Murine models of pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol 294(2):L152–L160. https://doi.org/10.1152/ajplung.00313.2007
Niu YM, Zhu XL, Chang B, Tong ZH, Cao W, Qiao PH, Zhang LY, Zhao J, Song YG (2016) Nanosilica and polyacrylate/nanosilica: a comparative study of acute toxicity. Biomed Res Int 2016:9353275. https://doi.org/10.1155/2016/9353275
Nowak JS, Mehn D, Nativo P, García CP, Gioria S, Ojea-Jiménez I, Gilliland D, Rossi F (2014) Silica nanoparticle uptake induces survival mechanism in A549 cells by the activation of autophagy but not apoptosis. Toxicol Lett 224(1):84–92. https://doi.org/10.1016/j.toxlet.2013.10.003
Ono A, Utsugi M, Masubuchi K, Ishizuka T, Kawata T, Shimizu Y, Hisada T, Hamuro J, Mori M, Dobashi K (2009) Glutathione redox regulates TGF-β-induced fibrogenic effects through Smad3 activation. FEBS Lett 583(2):357–362. https://doi.org/10.1016/j.febslet.2008.12.021
Pandit KV, Corcoran D, Yousef H, Yarlagadda M, Tzouvelekis A, Gibson KF, Konishi K, Yousem SA, Singh M, Handley D, Richards T, Selman M, Watkins SC, Pardo A, Ben-Yehudah A, Bouros D, Eickelberg O, Ray P, Benos PV, Kaminski N (2010) Inhibition and role of let-7d in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 182(2):220–229. https://doi.org/10.1164/rccm.200911-1698OC
Ponticos M, Holmes AM, Shi WX, Leoni P, Khan K, Rajkumar VS, Hoyles RK, Bou-Gharios G, Black CM, Denton CP, Abraham DJ, Leask A, Lindahl GE (2009) Pivotal role of connective tissue growth factor in lung fibrosis: MAPK-dependent transcriptional activation of type I collagen. Arthritis Rheum 60(7):2142–2155. https://doi.org/10.1002/art.24620
Rabolli V, Badissi AA, Devosse R, Uwambayinema F, Yakoub Y, Palmai-Pallag M, Lebrun A, De Gussem V, Couillin I, Ryffel B, Marbaix E, Lison D, Huaux F (2014) The alarmin IL-1α is a master cytokine in acute lung inflammation induced by silica micro- and nanoparticles. Part Fibre Toxicol 11(1):69. https://doi.org/10.1186/s12989-014-0069-x
Sayes CM, Reed KL, Glover KP, Swain KA, Ostraat ML, Donner EM, Warheit DB (2010) Changing the dose metric for inhalation toxicity studies: short-term study in rats with engineered aerosolized amorphous silica nanoparticles. Inhal Toxicol 22(4):348–354. https://doi.org/10.3109/08958370903359992
Shen Z, Wu X, Wang Z, Li B, Zhu X (2015) Effect of miR-18a overexpression on the radiosensitivity of non-small cell lung cancer. Int J Clin Exp Pathol 8(1):643–648
Shi Y, Yadav S, Wang F, Wang H (2010) Endotoxin promotes adverse effects of amorphous silica nanoparticles on lung epithelial cells in vitro. J Toxicol Environ Health A 73(11):748–756. https://doi.org/10.1080/15287391003614042
Sonnylal S, Shi WX, Leoni P, Naff K, Van Pelt CS, Nakamura H, Leask A, Abraham D, Bou-Gharios G, de Crombrugghe B (2010) Selective expression of connective tissue growth factor in fibroblasts in vivo promotes systemic tissue fibrosis. Arthritis Rheum 62(5):1523–1532. https://doi.org/10.1002/art.27382
Strieter RM, Mehrad B (2009) New mechanisms of pulmonary fibrosis. Chest 136(5):1364–1370. https://doi.org/10.1378/chest.09-0510
Sung DK, Kong WH, Park K, Kim JH, Kim MY, Kim H, Hahn SK (2013) Noncovalenly PEGylated CTGF siRNA/PDMAEMA complex for pulmonary treatment of bleomycin-induced lung fibrosis. Biomaterials 34(4):1261–1269. https://doi.org/10.1016/j.biomaterials.2012.09.061
Vance ME, Kuiken T, Vejerano EP, Mc Ginnis SP, Hochella MF Jr, Rejeski D, Hull MS (2015) Nanotechnology in the real world: redeveloping the nanomaterial consumer products inventory. Beilstein J Nanotechnol 6:1769–1780, doi: 10.3762/bjnano.6.181.
Wang Q, Usinger W, Nichols B, Gray J, Xu L, Seeley TW, Brenner M, Guo G, Zhang W, Oliver N, Lin A, Yeowell D (2011) Cooperative interaction of CTGF and TGF-β in animal models of fibrotic disease. Fibrogenesis Tissue Repair 4(1):4. https://doi.org/10.1186/1755-1536-4-4
Wynn TA (2008) Cellular and molecular mechanisms of fibrosis. J Pathol 214(2):199–210
Yang H, Wu Q, Tang M, Cao S, Dong Y, Sheng J, Lu Z (2008) In vitro study of cytotoxicity of nanosized silica. Dongnan Daxue Xuebao (Ziran Kexue Ban) 38(6):1054–1060
Yang H, Wu Q, Tang M, Liu X, Deng H, Kong L, Lu Z (2010) In vitro study of silica nanoparticle-induced cytotoxicity based on real-time cell electronic sensing system. J Nanosci Nanotechnol 10(1):561–568. https://doi.org/10.1166/jnn.2010.1735
Yang H, Zhang Y, Li W, Lao C, Li M, Zheng Y (2016) Screening of different expression miRNAs in pulmonary injury induced by nanosized SiO2. Nanomedicine 12(2):549. https://doi.org/10.1016/j.nano.2015.12.287
Yang P, Zhang J, Xia Y, Huang F, Xu YY, Zheng YF, Yang J, Zhu XQ (2013) The pulmonary toxicity study of nano-silica particles on rats through dynamic inhalation. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi 31(7):487–491
Zhang Y, Li W, Zheng Y, Wang X, Li G, Yang H (2014) Dynamic changes of pathological morphology and ultrastructure of lung injury in rats induced by SiO2 nanoparticles. Zhonghua lao dong wei sheng zhi ye bing za zhi 32:504–510
Zhu X, Cao W, Chang B, Zhang L, Qiao P, Li X, Si L, Niu Y, Song Y (2016) Polyacrylate/nanosilica causes pleural and pericardial effusion, and pulmonary fibrosis and granuloma in rats similar to those observed in exposed workers. Int J Nanomedicine 11:1593–1605. https://doi.org/10.2147/IJN.S102020
Funding
[Fund Project] Supported by the National Natural Science Foundation of China (Grant No.81273046), the Fundamental Research Funds for the Central Universities, the Preventive Medicine Research Projects of Jiangsu Province (Grant No.Y2012039), and the Major State Basic Research Development Program of China (973Program, Grant No.2011CB933404).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Yang, H., Li, W., Zhang, Y. et al. Regulatory role of miR-18a to CCN2 by TGF-β1 signaling pathway in pulmonary injury induced by nano-SiO2 . Environ Sci Pollut Res 25, 867–876 (2018). https://doi.org/10.1007/s11356-017-0344-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0344-0