Abstract
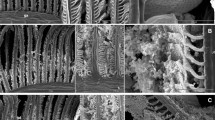
Scanning electron microscopic observations were made for the changes in the surface ultra morphology of gill of Labeo rohita as indicators of the stress of lethal (0.1, 0.2, 0.4, 0.6, and 0.8 mg/L dye) and sublethal (0.0225, 0.045, and 0.09 mg/L dye) doses of Basic Violet-1 (CI: 42535, Trade name- Methyl Violet-2B). Present study was taken up as insufficient data exist regarding safety of this dye. The dye was observed to be cytotoxic in nature during the short term (96 h) exposure to lethal doses and tumorogenic as well as cytotoxic during the long term (150 day) exposure to sublethal doses. The dye caused reduction or complete loss of microridges, proliferation of chloride cells, and degeneration of gill lamellae and rakers. The toxic effects became more pronounced with duration as 0.0225 mg/L dye caused remarkable distortion of the normal structure of gills after 150 days of exposure. Such changes may have become the underlying cause for 45–50 % mortality of fish exposed to even sublethal doses of dye as the gills of fish perform a range of vital functions. In the present study, changes in ultra morphology were observed on the 50th day whereas mortality was noticed between 100 and 150 days of subchronic exposure. Therefore, time to time monitoring of ultra morphology of gill will provide us early indicators for the stress of very low levels of pollutants which may later cause mortality of the fish.








Similar content being viewed by others
Abbreviations
- BV-1:
-
Basic Violet-1
- SEM:
-
Scanning electron microscopy
References
Alazemi BH, Lewis JW, Andrews EB (1996) Gill damage in the freshwater fish Gnathonemus petersii (Family: Mormyridae) exposed to selected pollutants: an ultra structural study. Environ Tech 17(3):225–238
Aljofan M, Sganga ML, Lo MK, Rootes CL, Porotto M, Meyer AG, Saubern S, Moscona A, Mungall BA (2009) Antiviral activity of gliotoxin, gentian violet and brilliant green against Nipah and Hendra virus in vitro. Virology J 6:187. doi:10.1186/1743-422X-6-187
APHA (1998) American standard methods for the examination of water and wastewater, 20th edn. American Publication of Health Association, Washington, DC, pp 2005–2605
Chung KT, Stevens JR (1993) Degradation of azo dyes by environmental microorganisms and helminthes. Environ Toxicol Chem 12:2121–2132
Combes RD, Haveland-Smith RB (1982) A review of the genotoxicity of food, drug and cosmetic colors and other also, triphenylmethane and xanthene dyes. Mutat Res 98:101–248
Culp SJ, Mellick PW, Trotter RW, Greenlees KJ, Kodell RL, Beland FA (2006) Carcinogenicity of malachite green chloride and leucomalachite green in B6C3F1 mice and F344 rats. Food Chem Toxicol 44:1204–1212
Dell Antone P, Colonna R, Azzone GF (1972) The membrane structure studied with cationic dyes. 1. The binding of cationic dyes to submitochondrial particles and the question of the polarity of the ion-translocation mechanism. Eur J Biochem 24:553–565
Diamante C, Bergfeld WF, Belsito DV, Klaassen CD, Marks JG Jr, Shank RC, Slaga TJ, Synder PW, Alan Andersen F (2009) Final report on the safety assessment of Basic Violet 1, Basic Violet 3 and Basic Violet 4. Int J Toxicol 28(6):193S–204S
Dutta HM, Zeno T, Richmonds C (1992) Toxic action of diazinon on the gills of bluegill sunfish Lepomis macrochirus. Ohio J Sci 92:09
Dutta HM, Munshi JSD, Roy PK, Singh NK, Adhikari S, Killius J (1996) Ultrastructural changes in the respiratory lamellae of the catfish, Heteropneustes fossilis, after sublethal exposure to malathion. Environ Pollut 9:329–341
Dutta HM, Munshi JSD, Roy PK, Singh NK, Motz L, Adhikari S (1997) Effects of diazinon on bluegill sunfish, Lepomis macrochirus, gills: scanning electron microscope observations. Exp Biol Online 2:17–31
Evans DH, Piermarini PM, Choe KP (2005) The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation and excretion of nitrogenous waste. Physiol Rev 85:97–177
Hao OJ, Kim H, Chiang PC (2000) Decolorization of wastewater. Crit Rev Env Sci Tec 30:449–505
Hart RK, Oglesby GB (1979) Toxin induced changes in the gill epithelium of rainbow trout (Salmo gairdneri). Scanning Electron Microsc 3:355–361
Jacobs D, Exmond EF, Melisky EL, Hocutt CH (1981) Morphological changes in gill epithelium of heat stressed rainbow trout, Salmo gairdneri: evidence in support of a temperature induced surface area hypothesis. Can J Fish Aquat Sci 38:16–22
Jagoe CH, Haines TA (1983) Alterations in gill epithelium morphology of yearling sunapee trout exposed to acute acid stress. Trans Am Fish Soc 112:689–695
Kaur R, Dua A (2015) 96 h LC50, behavioural alterations and histopathological effects due to wastewater toxicity in a freshwater fish Channa punctatus. Environ Sci Pollut Res 22:5100–5110
Khanna SK, Das M (1991) Toxicity, carcinogenic potential and clinic epidemiological studies on dyes and dye intermediate. J Sci Ind Res 50:965–974
Khidr MB, Mekkawy IAA (2008) Effect of separate and combined lead and selenium on the liver of the cichlid fish Oreochromis niloticus: ultrastructural study. Egypt J Zool Vol 50:89–119
Laurent P (1984) Gill internal morphology. In: Hoar WS, Randall DJ (eds) Fish physiology. Academic, New York, pp 73–183
Mallatt J (1985) Fish gill structural changes induced by toxicants and other irritants: a statistical review. Can J Fish Aquat Sci 42:630–648
Mathur N, Bhatnagar P, Nagar P, Bijarnia MK (2005) Mutagenicity assessment of from textile/dye industries of Sanganer, Jaipur (India): a case study. Ecotoxicol Environ Saf 61:105–113
McCarthy B (1997) Biotechnology and coloration. Rev Prog Color Relat Top 27:26–31
McCoy EC, Speck WT, Rosenkranz HS (1977) Activation of a procarcinogen to a mutagen by cell-free extracts of anaerobic bacteria. Mutat Res 46:261–264
Moawad H, Abdel-Rahim WM, Khalafallah M (2003) Evaluation of bio-toxicity of textile dyes using two bioassays. J Basic Microbiol 43:218–229
Morgan M, Tovell PWA (1973) The structure of the gill of the trout, Salmo gairdneri (Richardson). Z Zellfo Mikros Anat 142:147–162
Nehls S, Segner H (2005) Comet assay with the fish cell line rainbow trout gonad-2 for in vitro genotoxicity testing of xenobiotics and surface waters. Environ Toxicol Chem 24:2078–2087
OECD (1992) Test No. 203: Fish, Acute Toxicity Test, OECD Guidelines for the Testing of Chemicals, Section 2, OECD Publishing, Paris. doi:10.1787/9789264069961-en
Part P, Lock RAC (1983) Diffusion of calcium, cadmium and mercury in a mucous solution from rainbow trout. Comp Biochem Physiol C 76:259–263
Roy PK, Munshi JSD (1991) Malathion induced structural and morphometric changes of gills of a freshwater major carp, Cirrhinus mrigala (Ham.). J Environ Biol 12:79–87
Roy PK, Munshi JSD, Munshi JD (1986) Scanning electron microscopy evaluation of effects of saponin on the gills of the climbing perch, Anabas testudeneus (Bloch) (Anabantidae: Pisces). Ind J Exp Biol 24:511–516
Shepherd KL (1994) Functions for fish mucous. Rev Fish Biol Fish 4:401–429
Singh AK (2007) Chrome black-T induced hyper pigmentation in the skin of an air breathing fish Colisa Chuna. J Exp Biol 10(1):99–102
Slokar YM, Lemarechal AM (1998) Methods for decoloration of textile wastewaters. Dyes Pigments 37:335–356
Souza Pietra RCC, Rodrigues LF, Teixeira E, Fried L, Lefkove B (2013) Triphenylmethane derivatives have high in vitro and in vivo activity against the main causative agents of cutaneous leishmaniasis. PLoS ONE 8(1), e51864
Temmink J, Bouweister P, De Jong P, Vander Berg J (1983) An ultrastructural study of chromate induced hyperplasia in the gill of rainbow trout (Salmo gairdneri). Aquat Toxicol 4:165–179
Trindade RC, Angelis DF, Camargo AT (1986) Studies about adsorpion of the azoic compounds by yeast of the Rhodotorula genus. Simpósio Nacional de Fermentação, Programas e Resumos de Comunicações, Londrina, p 10
Wong CKC, Wong MH (2000) Morphological and biochemical changes in the gills of Tilapia (Oreochromis mossambicus) to ambient cadmium exposure. Aquat Toxicol 48:517–527
Zachowski A, Durand P (1988) Biphasic nature of the binding of cationic amphipaths with artificial and biological membranes. Biochem Biophys Acta 937:411–416
Acknowledgment
Financial support from UGC as SRF to K Kaur (Vide Letter No. F.40-50(M/S)/2009(SA-III/MANF) and UPE to A Kaur is greatly acknowledged. We are thankful to Mr Kawal, Technician at the Central Instrumentation Facility, Guru Nanak Dev University, Amritsar for help in SEM photography.
Author contributions
The study was designed by A Kaur. K Kaur and S Kaur performed the experiment. A Kaur oversaw the study and provided overall supervision and management of the work. K Kaur wrote the first draft. All authors were involved in interpretation of results, critical evaluation, and approval of the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Responsible editor: Thomas Braunbeck
Rights and permissions
About this article
Cite this article
Kaur, K., Kaur, S. & Kaur, A. Scanning electron microscopic observations of Basic Violet-1 induced changes in the gill morphology of Labeo rohita . Environ Sci Pollut Res 23, 16579–16588 (2016). https://doi.org/10.1007/s11356-016-6764-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6764-4