Abstract
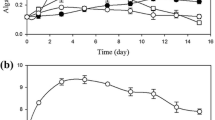
In a present study, nutrient removal from municipal wastewater by Chlorella vulgaris and Nannochloropsis oculata was investigated by using mixotrophic cultivation with glycerol (0 to 5 g/L). Performance parameters were assessed by estimating the removal of total nitrogen, total phosphorus, chemical oxygen demand (COD), biomass growth, chlorophyll content, lipid yield, and fatty acids. With the addition of 2 g/L glycerol, a maximum biomass productivity of 56 mg/L/day was achieved in the mixotrophic culture of C. vulgaris within 12 days. The mixotrophic culture showed a 30-fold increase in biomass productivity compared to the wastewater without any glycerol. However, the highest total nitrogen removal (80.62 %), total phosphate removal (60.72 %), and COD removal (96.3 %) was observed in the N. oculata culture supplemented with 3, 5, and 1 g/L glycerol, respectively. These results suggest that mixotrophic cultivation using glycerol offers great potential in the production of renewable biomass, waste water treatment, and consequent production of high-value microalgal oil.

Simultaneous biomass production and nutrient removal using microalgae cultivated in wastewater supplemented with glycerol






Similar content being viewed by others
References
Acién FG, Fernández JM, Magán JJ, Molina E (2012) Production cost of a real microalgae production plant and strategies to reduce it. Biotechnol Adv 30:1344–1353. doi:10.1016/j.biotechadv.2012.02.005
Singh A, Nigam PS, Rathore D (2015) Commercialization of marine algae-derived biofuels. In: Kim S-K, Lee C-G (eds) Marine bioenergy. CRC Press, Boca Raton, p 641–652
APHA (1998) APHA Standard Methods for the Examination of Water and Wastewater, Washington DC
Beuckels A, Smolders E, Muylaert K (2015) Nitrogen availability influences phosphorus removal in microalgae-based wastewater treatment. Water Res 77:98–106. doi:10.1016/j.watres.2015.03.018
Bhatnagar A, Chinnasamy S, Singh M, Das KC (2011) Renewable biomass production by mixotrophic algae in the presence of various carbon sources and wastewaters. Appl Energy 88:3425–3431. doi:10.1016/j.apenergy.2010.12.064
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917. doi:10.1139/o59-099
Cardozo KHM, Guaratini T, Barros MP et al (2007) Metabolites from algae with economical impact. Comp Biochem Physiol Part C Toxicol Pharmacol 146:60–78. doi:10.1016/j.cbpc.2006.05.007
Carreau JP, Dubacq JP (1978) Adaptation of a macro-scale method to the micro-scale for fatty acid methyl transesterification of biological lipid extracts. J Chromatogr A 151:384–390. doi:10.1016/S0021-9673(00)88356-9
Cerón-García MC, Macías-Sánchez MD, Sánchez-Mirón A et al (2013) A process for biodiesel production involving the heterotrophic fermentation of Chlorella protothecoides with glycerol as the carbon source. Appl Energy 103:341–349. doi:10.1016/j.apenergy.2012.09.054
Cheng Y, Lu Y, Gao C, Wu Q (2009) Alga-based biodiesel production and optimization using sugar cane as the feedstock. Energy Fuels 23:4166–4173. doi:10.1021/ef9003818
Chen Y-H, Walker TH (2011) Biomass and lipid production of heterotrophic microalgae Chlorella protothecoides by using biodiesel-derived crude glycerol. Biotechnol Lett 33:1973–1983. doi:10.1007/s10529-011-0672-y
Chinnasamy S, Bhatnagar A, Hunt RW, Das KC (2010) Microalgae cultivation in a wastewater dominated by carpet mill effluents for biofuel applications. Bioresour Technol 101:3097–3105. doi:10.1016/j.biortech.2009.12.026
Chi Z, Pyle D, Wen Z et al (2007) A laboratory study of producing docosahexaenoic acid from biodiesel-waste glycerol by microalgal fermentation. Process Biochem 42:1537–1545. doi:10.1016/j.procbio.2007.08.008
Choi HJ, Lee SM (2014) Effect of the N/P ratio on biomass productivity and nutrient removal from municipal wastewater. Bioprocess Biosyst Eng 1–6. doi: 10.1007/s00449-014-1317-z
Christenson L, Sims R (2011) Production and harvesting of microalgae for wastewater treatment, biofuels, and bioproducts. Biotechnol Adv 29:686–702. doi:10.1016/j.biotechadv.2011.05.015
Costache TA, Fernández FGA, Morales MM et al (2013) Comprehensive model of microalgae photosynthesis rate as a function of culture conditions in photobioreactors. Appl Microbiol Biotechnol 97:7627–7637. doi:10.1007/s00253-013-5035-2
Dijkstra AJ (2006) Revisiting the formation of trans isomers during partial hydrogenation of triacylglycerol oils. Eur J Lipid Sci Technol 108:249–264. doi:10.1002/ejlt.200500335
EC (2009) Directive 2009/28/EC of the European Parliament of 23 April 2009 on the promotion of the use of energy from renewable sources and amending and subsequently repealing Directives 2001/77/EC and 2003/30/EC. Official Journal of the European Union. Off J Eur Union L140:16–62
European Standard EN 14214 (2008) Automotive fuels. Fatty acid methylesters (FAME) for diesel engines. Requirements and test methods.
Feng X, Walker TH, Bridges WC et al (2014) Biomass and lipid production of Chlorella protothecoides under heterotrophic cultivation on a mixed waste substrate of brewer fermentation and crude glycerol. Bioresour Technol 166:17–23. doi:10.1016/j.biortech.2014.03.120
Galhardo CX, Masini JC (2000) Spectrophotometric determination of phosphate and silicate by sequential injection using molybdenum blue chemistry. Anal Chim Acta 417:191–200. doi:10.1016/S0003-2670(00)00933-8
Gouveia L, Oliveira AC (2009) Microalgae as a raw material for biofuels production. J Ind Microbiol Biotechnol 36:269–274. doi:10.1007/s10295-008-0495-6
Guillard RRL, Ryther JH (1962) Studies of marine planktonic diatoms: I. Cyclotella Nana Hustedt, and Detonula Confervacea (Cleve) Gran. Can J Microbiol 8:229–239. doi:10.1139/m62-029
Heredia-Arroyo T, Wei W, Hu B (2010) Oil accumulation via heterotrophic/mixotrophic Chlorella protothecoides. Appl Biochem Biotechnol 162:1978–1995. doi:10.1007/s12010-010-8974-4
Heredia-Arroyo T, Wei W, Ruan R, Hu B (2011) Mixotrophic cultivation of Chlorella vulgaris and its potential application for the oil accumulation from non-sugar materials. Biomass Bioenergy 35:2245–2253. doi:10.1016/j.biombioe.2011.02.036
Huppe HC, Turpin DH (1994) Integration of carbon and nitrogen metabolism in plant and algal cells. Annu Rev Plant Physiol Plant Mol Biol 45:577–607. doi:10.1146/annurev.pp.45.060194.003045
Johnson X, Alric J (2013) Central carbon metabolism and electron transport in Chlamydomonas reinhardtii: metabolic constraints for carbon partitioning between oil and starch. Eukaryot Cell 12:776–793. doi:10.1128/EC.00318-12
Knothe G (2008) “Designer” biodiesel: optimizing fatty ester composition to improve fuel properties†. Energy Fuels 22:1358–1364. doi:10.1021/ef700639e
Koch FC, McMeekin TL (1924) A new direct nesslerization micro-Kjeldahl method and a modification of the Nessler-Folin reagent for ammonia. J Am Chem Soc 46:2066–2069. doi:10.1021/ja01674a013
Kruse O, Rupprecht J, Mussgnug JH et al (2005) Photosynthesis: a blueprint for solar energy capture and biohydrogen production technologies. Photochem Photobiol Sci Off J Eur Photochem Assoc Eur Soc Photobiol 4:957–970. doi:10.1039/b506923h
Liang Y, Sarkany N, Cui Y (2009) Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotechnol Lett 31:1043–1049. doi:10.1007/s10529-009-9975-7
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Li T, Zheng Y, Yu L, Chen S (2013) High productivity cultivation of a heat-resistant microalga Chlorella sorokiniana for biofuel production. Bioresour Technol 131:60–67. doi:10.1016/j.biortech.2012.11.121
Liu N, Li F, Ge F et al (2015) Mechanisms of ammonium assimilation by Chlorella vulgaris F1068: isotope fractionation and proteomic approaches. Bioresour Technol 190:307–314. doi:10.1016/j.biortech.2015.04.024
Li Y, Fei X, Deng X (2012) Novel molecular insights into nitrogen starvation-induced triacylglycerols accumulation revealed by differential gene expression analysis in green algae Micractinium pusillum. Biomass Bioenergy 42:199–211. doi:10.1016/j.biombioe.2012.03.010
Meng X, Yang J, Xu X et al (2009) Biodiesel production from oleaginous microorganisms. Renew Energy 34:1–5. doi:10.1016/j.renene.2008.04.014
Neilson AH, Lewin RA (1974) The uptake and utilization of organic carbon by algae: an essay in comparative biochemistry. Phycologia 13:227–264. doi:10.2216/i0031-8884-13-3-227.1
Ogawa T, Aiba S (1981) Bioenergetic analysis of mixotrophic growth in Chlorella vulgaris and Scenedesmus acutus. Biotechnol Bioeng 23:1121–1132. doi:10.1002/bit.260230519
Ogbonna JC, Masui H, Tanaka H (1997) Sequential heterotrophic/autotrophic cultivation—an efficient method of producing Chlorella biomass for health food and animal feed. J Appl Phycol 9:359–366. doi:10.1023/A:1007981930676
Pancha I, Chokshi K, George B et al (2014) Nitrogen stress triggered biochemical and morphological changes in the microalgae Scenedesmus sp. CCNM 1077. Bioresour Technol 156:146–154. doi:10.1016/j.biortech.2014.01.025
Perez-Garcia O, Escalante FME, de-Bashan LE, Bashan Y (2011) Heterotrophic cultures of microalgae: metabolism and potential products. Water Res 45:11–36. doi:10.1016/j.watres.2010.08.037
Quispe CAG, Coronado CJR, Carvalho JA Jr (2013) Glycerol: production, consumption, prices, characterization and new trends in combustion. Renew Sustain Energy Rev 27:475–493. doi:10.1016/j.rser.2013.06.017
Rai MP, Nigam S, Sharma R (2013) Response of growth and fatty acid compositions of Chlorella pyrenoidosa under mixotrophic cultivation with acetate and glycerol for bioenergy application. Biomass Bioenergy 58:251–257. doi:10.1016/j.biombioe.2013.08.038
Spolaore P, Joannis-Cassan C, Duran E, Isambert A (2006) Commercial applications of microalgae. J Biosci Bioeng 101:87–96. doi:10.1263/jbb.101.87
Tompkins J, Culture Collection of Algae and Protozoa, Natural Environment Research Council (Great Britain) (1995) Catalogue of strains, 1995. Culture Collection of Algae and Protozoa, Ambleside, Cumbria, U.K., p 204
Wan M, Liu P, Xia J et al (2011) The effect of mixotrophy on microalgal growth, lipid content, and expression levels of three pathway genes in Chlorella sorokiniana. Appl Microbiol Biotechnol 91:835–844. doi:10.1007/s00253-011-3399-8
Xiong W, Gao C, Yan D et al (2010) Double CO2 fixation in photosynthesis–fermentation model enhances algal lipid synthesis for biodiesel production. Bioresour Technol 101:2287–2293. doi:10.1016/j.biortech.2009.11.041
Acknowledgments
This work was supported by the National Research Foundation (NRF) of Korea grant funded by the Korean Government (MEST) (2012R1A2A4A01001539).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 3004 kb)
Rights and permissions
About this article
Cite this article
Gupta, P.L., Choi, H.J. & Lee, SM. Enhanced nutrient removal from municipal wastewater assisted by mixotrophic microalgal cultivation using glycerol. Environ Sci Pollut Res 23, 10114–10123 (2016). https://doi.org/10.1007/s11356-016-6224-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6224-1