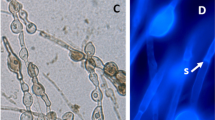
Abstract
For re-forestation of metal-contaminated land, ectomycorrhizal trees may provide a solution. Hence, the study of the interaction is necessary to allow for comprehensive understanding of the mutually symbiotic features. On a structural level, hyphal mantle and the Hartig’ net formed in the root apoplast are essential for plant protection and mycorrhizal functioning. As a model, we used the basidiomycete Tricholoma vaccinum and its host spruce (Picea abies). Using an optimized hydroponic cultivation system, both features could be visualized and lower stress response of the tree was obtained in non-challenged cultivation. Larger spaces in the apoplasts could be shown with high statistical significance. The easy accessibility will allow to address metal stress or molecular responses in both partners. Additionally, the proposed cultivation system will enable for other experimental applications like addressing flooding, biological interactions with helper bacteria, chemical signaling, or other biotic or abiotic challenges relevant in the natural habitat.



Similar content being viewed by others
References
Agerer R (1995) Anatomical characteristics of identified eczomycorrhizas: an attempt towards a natural classification. In: Varma A, Hock B (eds) Mycorrhiza structure, function, molecular biology and biotechnology. Springer, Berlin, pp 685–734
Arduini D, Godbold L, Onnis A (1994) Heavy metal uptake and distribution in tree seedlings. G Bot Ital 128(1):219
Asiimwe T, Krause K, Schlunk I, Kothe E (2010) Ectomycorrhiza in sustainable ecosystem functioning: A closer look at the symbiotic association. In: Behl RK (ed) Resource management towards sustainable agriculture and development. Agrobios Publishers, Jodhpur, pp 113–120
Asiimwe T, Krause K, Schlunk I, Kothe E (2012) Modulation of ethanol stress tolerance by aldehyde dehydrogenase in the mycorrhizal fungus Tricholoma vaccinum. Mycorrhiza 22:471–484
Brunner I (1993) Production of ectomycorrhizal Picea abies–Hebeloma crustuliniforme seedlings for ecological studies: effects of synthesis techniques on the morphology of the symbiosis. Water Air Soil Pollut 68:231–240
Büntgen U, Egli S (2014) Breaking new ground at the interface of dendroecology and mycology. Trends Plant Sci 19:613–614
Chilvers GA, Douglass PA, Lapeyrie FF (1986) A paper-sandwich technique for rapid synthesis of ectomycorrhizas. New Phytol 103:397–402
Clemmensen KE, Finlay RD, Dahlberg A, Stenlid J, Wardle DA, Lindahl BD (2015) Carbon sequestration is related to mycorrhizal fungal community shifts during long-term succession in boreal forests. New Phytol 205:1525–1536
de Freitas Pereira M, Betancourth BM, Teixeira JA, Zubieta MP, de Queiroz MV, Kasuya MC, Costa MD, de Araújo EF (2014) In vitro Scleroderma laeve and Eucalyptus grandis mycorrhization and analysis of atp6, 17S rDNA, and ras gene expression during ectomycorrhizal formation. J Basic Microbiol 54:1358–1366
Dore J, Marmeisse R, Combier JP, Gay G (2014) A fungal conserved gene from the basidiomycete Hebeloma cylindrosporum is essential for efficient ectomycorrhiza formation. Mol Plant Microbe Interact 27:1059–1069
Duponnois R, Plenchette C (2003) A mycorrhiza helper bacterium enhances ectomycorrhizal and endomycorrhizal symbiosis of Australian Acacia species. Mycorrhiza 13:85–91
Furrer G, Phillips BL, Ulrich KU, Pöthig R, Casey WH (2002) The origin of aluminum flocs in polluted streams. Science 297:2245–2247
Garcia K, Zimmermann SD (2014) The role of mycorrhizal associations in plant potassium nutrition. Front Plant Sci 5:337
Garcia K, Delteil A, Conéjéro G, Becquer A, Plassard C, Sentenac H, Zimmermann SD (2014) Potassium nutrition of ectomycorrhizal Pinus pinaster: overexpression of the Hebeloma cylindrosporum HcTrk1 transporter affects the translocation of both K+ and phosphorus in the host plant. New Phytol 201:951–960
Gehring CA, Müller RC, Haskins KE, Rubow TK, Whitham TG (2014) Convergence in mycorrhizal fungal communities due to drought, plant competition, parasitism, and susceptibility to herbivory: consequences for fungi and host plants. Front Microbiol 5:306
Hacskaylo E (1953) Pure culture syntheses of pine mycorrhizae in Terra-Lite. Mycologia 45(6):971–975
Haferburg G, Kothe E (2010) Metallomics: lessons for metalliferous soil remediation. Appl Microbiol Biotechnol 87:1271–1280
Hayward J, Horton TR (2014) Phylogenetic trait conservation in the partner choice of a group of ectomycorrhizal trees. Mol Ecol 23:4886–4898
Heim A, Brunner I, Frey B, Frossard E, Luster J (2001) Root exudation, organic acids, and element distribution in roots of Norway spruce treated with aluminium in hydroponics. J Plant Nutr Soil Sci 164:519–526
Iordache V, Gherghel F, Kothe E (2009) Assessing the effect of disturbances on ectomycorrhiza diversity. Int J Environ Res Publ Health 6:414–432
Knabe N, Jung E-M, Freihorst D, Hennicke F, Horton JS, Kothe E (2013) A central role for Ras1 in morphogenesis of the basidiomycete Schizophyllum commune. Eukaryot Cell 12:941–952
Kottke I, Guttenberger M, Hampp R, Oberwinkler F (1987) An in vitro method for establishing mycorrhizae on coniferous tree seedlings. Trees 1:191–194
Kozlowski TT (1997) Responses of woody plants to flooding and salinity. Tree Physiol Monogr 1:1–29
Krause K, Kothe E (2006) Use of RNA fingerprinting to identify fungal genes specifically expressed during ectomycorrhizal interaction. J Basic Microbiol 46:387–399
Kuo A, Kohler A, Martin FM, Grigoriev IV (2014) Expanding genomics of mycorrhizal symbiosis. Front Microbiol 5:582
Langella F, Grawunder A, Stark R, Weist A, Merten D, Haferburg G, Büchel G, Kothe E (2014) Microbially assisted phytoremediation approaches for two multi-element contaminated sites. Environ Sci Pollut Res 21:6845–6858
Lepp NW (1970) The potential of tree-ring analysis for monitoring heavy metal pollution patterns. Environ Pollut 9:49–61
Liao HL, Chen Y, Bruns TD, Peay KG, Taylor JW, Branco S, Talbot JM, Vilgalys R (2014) Metatranscriptomic analysis of ectomycorrhizal roots reveals genes associated with Piloderma-Pinus symbiosis: improved methodologies for assessing gene expression in situ. Environ Microbiol 16(12):3730–3742
Nicoară A, Neagoe A, Stancu P, de Giudici G, Langella F, Sprocati AR, Iordache V, Kothe E (2014) Coupled pot and lysimeter experiments assessing plant performance in microbially assisted phytoremediation. Environ Sci Pollut Res 21:6905–6920
Nylund J-E, Wallander H, Sundberg B, Gay G (1994) IAA-overproducer mutants of Hebeloma cylindrosporum Romagnesi mycorrhizal with Pinus pinaster (Ait.) Sol. and P. sylvestris L. in hydroponic culture. Mycorrhiza 4:247–250
Osundina MA (1998) Nodulation and growth of mycorrhizal Casuarina equisetifolia J.R. and G. First in response to flooding. Biol Fertil Soils 26:95–99
Pérez Rodríguez N, Langella F, Rodushkin I, Engström E, Kothe E, Alakangas L, Öhlander B (2014) The role of bacterial consortium and organic amendment in Cu and Fe isotope fractionation in plants on a polluted mine site. Environ Sci Pollut Res 21:6836–6844
Pfeiffer F (1898) Beiträge zur Fixierung und Präparation der Süßwasseralgen. Österr Bot Z 48:53–59
Phieler R, Voit A, Kothe E (2014) Microbially supported phytoremediation of heavy metal contaminated soils: strategies and applications. Adv Biochem Eng Biotechnol 141:211–235
Plett JM, Tisserant E, Brun A, Morin E, Grigoriev IV, Kuo A, Martin FM, Kohler A (2015) The mutualist Laccaria bicolor expresses a core gene regulon during the colonization of diverse host plants and a variable regulon to counteract host-specific defenses. Mol Plant Microbe Interact 28(3):261–273
Pohjanen J, Koskimäki JJ, Sutela S, Ardanov P, Suorsa M, Niemi K, Sarjala T, Häggman H, Pirttilä AM (2014) Interaction with ectomycorrhizal fungi and endophytic Methylobacterium affects nutrient uptake and growth of pine seedlings in vitro. Tree Physiol 34:993–1005
Raudaskoski M, Kothe E (2014) Novel findings on the role of signal exchange in arbuscular and ectomycorrhizal symbioses. Mycorrhiza. doi:10.1007/s00572-014-0607-2
Rovira AD (1959) Root excretions in relation to the rhizosphere effect. Plant Soil 11:53–64
Rutto KL, Mizutani F, Kadoya K (2002) Effect of root-zone flooding on mycorrhizal and non-mycorrhizal peach (Prunus persica Batsch) seedlings. Sci Hortic 94:285–295
Scheidegger C, Brunner I (1993) Freeze-fracturing for low-temperature scanning electron microscopy of Hartig net in synthesized Picea abies - Hebeloma crustuliniforme and -Tricholoma vaccinum ectomycorrhizas. New Phytol 123:123–132
Schütze E, Klose M, Merten D, Nietzsche S, Senftleben D, Roth M, Kothe E (2014) Growth of streptomycetes in soil and their impact on bioremediation. J Hazard Mater 267:128–135
Sebastiana M, Vieira B, Lino-Neto T, Monteiro F, Figueiredo A, Sousa L, Pais MS, Tavares R, Paulo OS (2014) Oak root response to ectomycorrhizal symbiosis establishment: RNA-Seq derived transcript identification and expression profiling. PLoS One 9(5):e98376. doi:10.1371/journal.pone.0098376
Seven J, Polle A (2014) Subcellular nutrient element localization and enrichment in ecto- and arbuscular mycorrhizas of field-grown beech and ash trees indicate functional differences. PLoS One 9(12):e114672. doi:10.1371/journal.pone.0114672
Smith SE, Read DJ (1997) Mycorrhizal symbiosis. Academic Press Inc., San Diego
Ugawa S, Fukuda K (2005) The response of ectomycorrhizal fungi on Pinus densiflora seedling roots to liquid culture. J For Res 10:233–237
Veneault-Fourrey C, Commun C, Kohler A, Morin E, Balestrini R, Plett J, Danchin E, Coutinho P, Wiebenga A, de Vries RP, Henrissat B, Martin F (2014) Genomic and transcriptomic analysis of Laccaria bicolor CAZome reveals insights into polysaccharides remodelling during symbiosis establishment. Fungal Genet Biol 72:168–181
Wenkart S, Roth-Bejerano N, Mills D, Kagan-Zur V (2001) Mycorrhizal associations between Tuber melanosporum mycelia and transformed roots of Cistus incanus. Plant Cell Rep 20:369–373
Acknowledgments
This research was funded by the University of Jena and the Research Training Group “Alteration and element mobility at microbe-mineral interfaces". We would like to thank the Graduate School “Jena School for Microbial Communication” (JSMC) as part of the German Excellence Initiative and the German National Academic Foundation for support in this work. The authors especially thank Annekatrin Voigt for technical support.
Conflict of interest
The authors state that no potential conflicts of interests exist.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Henke, C., Jung, EM. & Kothe, E. Hartig’ net formation of Tricholoma vaccinum-spruce ectomycorrhiza in hydroponic cultures. Environ Sci Pollut Res 22, 19394–19399 (2015). https://doi.org/10.1007/s11356-015-4354-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-4354-5