Abstract
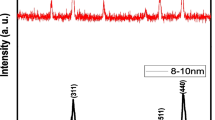
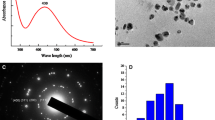
Within the last years, nanogold has become more and more important in nanotechnology, for example, as catalyst or in medical applications. Its rising production, application, and disposal inevitably lead to an increased emission of gold nanoparticles (Au-NPs) in the environment. However, only little is known about the uptake and effects of Au-NPs on biota. The objective of this study was to investigate the reversibility and effects of citrate-coated Au-NP uptake into the model organism barley (Hordeum vulgare L.). For this purpose, barley seeds were cultivated in Au-NP-containing nutrient solution for 2 weeks before the seedlings were transferred into Au-NP-free media and grown for another 3 weeks. Stability of Au-NPs in the cultivation media was investigated over the 2-week exposure time. Gold content in the leaves and roots of the plants was measured after 2 weeks of exposure and after 7, 14, and 21 days of regeneration by means of total reflection X-ray fluorescence (TXRF) analysis after microwave-assisted digestion. Moreover, Au-NPs within plant material were localized by transmission electron microscopy (TEM) of ultrathin cross sections. The obtained results reveal that Au-NPs accumulate in the plant roots. Concentration-dependent effects on the uptake of macronutrients and micronutrients, as well as on biomass production of exposed plants, in particular, on root growth were observed. Even though exposed barley plants were able to regenerate to a certain extent, their root growth was permanently decreased.






Similar content being viewed by others
References
Asli S, Neumann PM (2009) Colloidal suspensions of clay or titanium dioxide nanoparticles can inhibit leaf growth and transpiration via physical effects on root water transport. Plant Cell Environ 32(5):577–584
Barrena R, Casals E, Colón J, Font X, Sánchez A, Puntes V (2009) Evaluation of the ecotoxicity of model nanoparticles. Chemosphere 75(7):850–857
Battke F, Schramel P, Ernst D (2003) A novel method for in vitro culture of plants: cultivation of barley in a floating hydroponic system. Plant Mol Biol Report 21(4):405–409
Battke F, Leopold K, Maier M, Schmidhalter U, Schuster M (2008) Palladium exposure of barley: uptake and effects. Plant Biol 10(2):272–276
Biddulph O, Cory R, Biddulph S (1959) Translocation of calcium in the bean plant. Plant Physiol 34:512–519
Chithrani BD, Ghazani AA, Chan WCW (2006) Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett 6(4):662–668
Ferry VE, Munday JN, Atwater HA (2010) Design considerations for plasmonic photovoltaics. Adv Mater 22(43):4794–4808
Foote BD, Hanson JB (1964) Ion uptake by soyabean root tissue depleted of calcium by EDTA. Plant Physiol 39:450–460
Giljohann DA, Seferos DS, Daniel WL, Massich MD, Patel PC, Mirkin CA (2010) Gold nanoparticles for biology and medicine. Angew Chem Int Ed 49(19):3280–3294
Glenn JB, White SA, Klaine SJ (2012) Interactions of gold nanoparticles with freshwater aquatic macrophytes are size and species dependent. Environ Toxicol Chem 31(1):194–201
Grisel R, Weststrate K-J, Gluhoi A, Nieuwenhuys BE (2002) Catalysis by gold nanoparticles. Gold Bull 35(2):39–45
Judy JD, Unrine JM, Bertsch PM (2011) Evidence for biomagnification of gold nanoparticles within a terrestrial food chain. Environ Sci Technol 45(2):776–781
Judy JD, Unrine JM, Rao W, Wirick S, Bertsch PM (2012) Bioavailability of gold nanomaterials to plants: importance of particle size and surface coating. Environ Sci Technol 46(15):8467–8474
Lasagna-Reeves C, Gonzalez-Romero D, Barria MA, Olmedo I, Clos A, Sadagopa Ramanujam VM, Urayama A, Vergara L, Kogan MJ, Soto C (2010) Bioaccumulation and toxicity of gold nanoparticles after repeated administration in mice. Biochem Biophys Res Commun 393(4):649–655
Leopold K, Schuster M (2011) Pd particles as standardized test material for bioavailability studies of traffic related Pd emissions to barley plants. In: Zereini F, Wiseman CLS (eds) Urban airborne particulate matter. Springer Berlin Heidelberg, Heidelberg, pp 399–410
Li L, Leopold K (2012) Ligand-assisted extraction for separation and preconcentration of gold nanoparticles from waters. Anal Chem 84(9):4340–4349
Lin D, Xing B (2007) Phytotoxicity of nanoparticles: inhibition of seed germination and root growth. Environ Pollut 150(2):243–250
Nel A, Xia T, Mädler L, Li N (2006) Toxic potential of materials at the nanolevel. Science 622(5761):622–627
Nowack B, Bucheli TD (2007) Occurrence, behavior and effects of nanoparticles in the environment. Environ Pollut 150(1):5–22
Ojea-Jiménez I, López X, Arbiol J, Puntes V (2012) Citrate-coated gold nanoparticles as smart scavengers for mercury(II) removal from polluted waters. ACS Nano 6(3):2253–2260
Pernodet N, Fang X, Sun Y, Bakhtina A, Ramakrishnan A, Sokolov J, Ulman A, Rafailovich M (2006) Adverse effects of citrate/gold nanoparticles on human dermal fibroblasts. Small 2(6):766–773
Racuciu M, Creanga D-E (2007) TMA-OH coated magnetic nanoparticles internalized in vegetal tissue. Rom J Physiol 52(3–4):395–402
Roco MC (2005) Environmentally responsible development of nanotechnology. Environ Sci Technol 39(5):106A–112A
Sabo-Attwood T, Unrine JM, Stone JW, Murphy CJ, Ghoshroy S, Blom D, Bertsch PM, Newman L (2012) Uptake, distribution and toxicity of gold nanoparticles in tobacco (Nicotiana xanthi) seedlings. Nanotoxicology 6(4):353–360
Sun L, Zhang Z, Wang S, Zhang J, Li H, Ren L, Weng J, Zhang Q (2009) Effect of pH on the interaction of gold nanoparticles with DNA and application in the detection of human p53 gene mutation. Nanoscale Res Lett 4:216–220
Tiede K, Hasselloev M, Breitbarth E, Chaudhry Q, Boxall ABA (2009) Considerations for environmental fate and ecotoxicity testing to support environmental risk assessments for engineered nanoparticles. J Chromatogr A 1216:503–509
True RH (1914) The harmful action of distilled water. Am J Bot 1:255–273
Zhang X-D, Wu H-Y, Wu D, Wang Y-Y, Chang J-H, Zhai Z-B, Meng A-M, Liu P-X, Zhang L-A, Fan F-Y (2010) Toxicologic effects of gold nanoparticles in vivo by different administration routes. Int J Nanomedicine 5:771–781
Zhu Z-J, Carboni R, Quercio MJ, Yan B, Miranda OR, Anderton DL, Arcaro KF, Rotello VM, Vachet RW (2010) Surface properties dictate uptake, distribution, excretion, and toxicity of nanoparticles in fish. Small 6(20):2261–2265
Acknowledgments
The authors would like to thank Josef Breun GmbH & Co. KG for providing barley seeds. We also acknowledge R. Schindl and K. Wörle (Institute for Analytical and Bioanalytical Chemistry, University of Ulm) for assistance with TEM measurements.
Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Elena Maestri
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 224 kb)
Rights and permissions
About this article
Cite this article
Feichtmeier, N.S., Walther, P. & Leopold, K. Uptake, effects, and regeneration of barley plants exposed to gold nanoparticles. Environ Sci Pollut Res 22, 8549–8558 (2015). https://doi.org/10.1007/s11356-014-4015-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-4015-0