Abstract
Purpose
3′-deoxy-3′-[18F]fluorothymidine ([18F]FLT), a cell proliferation positron emission tomography (PET) tracer, has been shown in numerous tumors to be more specific than 2-deoxy-2-[18F]fluoro-d-glucose ([18F]FDG) but less sensitive. We studied the capacity of a nontoxic concentration of 5-fluoro-2′-deoxyuridine (FdUrd), a thymidine synthesis inhibitor, to increase uptake of [18F]FLT in tumor xenografts.
Methods
The duration of the FdUrd effect in vivo on tumor cell cycling and thymidine analogue uptake was studied by varying FdUrd pretreatment timing and holding constant the timing of subsequent flow cytometry and 5-[125I]iodo-2′-deoxyuridine biodistribution measurements. In [18F]FLT studies, FdUrd pretreatment was generally performed 1 h before radiotracer injection. [18F]FLT biodistributions were measured 1 to 3 h after radiotracer injection of mice grafted with five different human tumors and pretreated or not with FdUrd and compared with [18F]FDG tumor uptake. Using microPET, the dynamic distribution of [18F]FLT was followed for 1.5 h in FdUrd pretreated mice. High-field T2-weighted magnetic resonance imaging (MRI) and histology were used comparatively in assessing tumor viability and proliferation.
Results
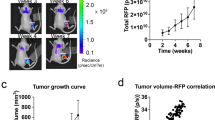
FdUrd induced an immediate increase in tumor uptake of 5-[125I]iodo-2′-deoxyuridine, that vanished after 6 h, as also confirmed by flow cytometry. Biodistribution measurements showed that FdUrd pretreatment increased [18F]FLT uptake in all tumors by factors of 3.2 to 7.8 compared with controls, while [18F]FDG tumor uptake was about fourfold and sixfold lower in breast cancers and lymphoma. Dynamic PET in FdUrd pretreated mice showed that [18F]FLT uptake in all tumors increased steadily up to 1.5 h. MRI showed a well-vascularized homogenous lymphoma with high [18F]FLT uptake, while in breast cancer, a central necrosis shown by MRI was inactive in PET, consistent with the histomorphological analysis.
Conclusion
We showed a reliable and significant uptake increase of [18F]FLT in different tumor xenografts after low-dose FdUrd pretreatment. These results show promise for a clinical application of FdUrd aimed at increasing the sensitivity of [18F]FLT PET.







Similar content being viewed by others
References
Bading JR, Shields AF (2008) Imaging of cell proliferation: status and prospects. J Nucl Med 49(Suppl 2):64S–80S
Sherley JL, Kelly TJ (1988) Regulation of human thymidine kinase during the cell cycle. J Biol Chem 263:8350–8358
Been LB, Elsinga PH, de Vries J et al (2006) Positron emission tomography in patients with breast cancer using (18)F-3′-deoxy-3′-fluoro-l-thymidine ((18)F-FLT)—a pilot study. Eur J Surg Oncol 32:39–43
Yamamoto Y, Nishiyama Y, Ishikawa S et al (2007) Correlation of 18F-FLT and 18F-FDG uptake on PET with Ki-67 immunohistochemistry in non-small cell lung cancer. Eur J Nucl Med Mol Imaging 34:1610–1616
Buck AK, Bommer M, Juweid ME et al (2008) First demonstration of leukemia imaging with the proliferation marker 18F-fluorodeoxythymidine. J Nucl Med 49:1756–1762
Vesselle H, Grierson J, Muzi M et al (2002) In vivo validation of 3′deoxy-3′-[(18)F]fluorothymidine ([(18)F]FLT) as a proliferation imaging tracer in humans: correlation of [(18)F]FLT uptake by positron emission tomography with Ki-67 immunohistochemistry and flow cytometry in human lung tumors. Clin Cancer Res 8:3315–3323
Kameyama R, Yamamoto Y, Izuishi K et al (2009) Detection of gastric cancer using 18F-FLT PET: comparison with 18F-FDG PET. Eur J Nucl Med Mol Imaging 36:382–388
Troost EG, Vogel WV, Merkx MA et al (2007) 18F-FLT PET does not discriminate between reactive and metastatic lymph nodes in primary head and neck cancer patients. J Nucl Med 48:726–735
Solit DB, Santos E, Pratilas CA et al (2007) 3′-Deoxy-3′-[18F]fluorothymidine positron emission tomography is a sensitive method for imaging the response of BRAF-dependent tumors to MEK inhibition. Cancer Res 67:11463–11469
Everitt S, Hicks RJ, Ball D et al (2009) Imaging cellular proliferation during chemo-radiotherapy: a pilot study of serial (18)F-FLT positron emission tomography/computed tomography imaging for non-small-cell lung cancer. Int J Radiat Oncol Biol Phys 75:1098–1104
Chen W, Cloughesy T, Kamdar N et al (2005) Imaging proliferation in brain tumors with 18F-FLT PET: comparison with 18F-FDG. J Nucl Med 46:945–952
Kenny L, Coombes RC, Vigushin DM, Al-Nahhas A, Shousha S, Aboagye EO (2007) Imaging early changes in proliferation at 1 week post chemotherapy: a pilot study in breast cancer patients with 3′-deoxy-3′-[18F]fluorothymidine positron emission tomography. Eur J Nucl Med Mol Imaging 34:1339–1347
Herrmann K, Wieder HA, Buck AK et al (2007) Early response assessment using 3′-deoxy-3′-[18F]fluorothymidine-positron emission tomography in high-grade non-Hodgkin's lymphoma. Clin Cancer Res 13:3552–3558
Kostakoglu L, Agress H Jr, Goldsmith SJ (2003) Clinical role of FDG PET in evaluation of cancer patients. Radiographics 23:315–340, quiz 533
Higashi K, Clavo AC, Wahl RL (1993) Does FDG uptake measure proliferative activity of human cancer cells? In vitro comparison with DNA flow cytometry and tritiated thymidine uptake. J Nucl Med 34:414–419
Juweid ME, Cheson BD (2006) Positron-emission tomography and assessment of cancer therapy. N Engl J Med 354:496–507
Tian J, Yang X, Yu L et al (2008) A multicenter clinical trial on the diagnostic value of dual-tracer PET/CT in pulmonary lesions using 3′-deoxy-3′-18F-fluorothymidine and 18F-FDG. J Nucl Med 49:186–194
Serlin O, Wolkoff JS, Amadeo JM, Keehn RJ (1969) Use of 5-fluorodeoxyuridine (FUDR) as an adjuvant to the surgical management of carcinoma of the stomach. Cancer 24:223–228
Kim DW, Ahn DS, Oh YH et al (2006) A new class of SN2 reactions catalyzed by protic solvents: facile fluorination for isotopic labeling of diagnostic molecules. J Am Chem Soc 128:16394–16397
Foulon CF, Adelstein SJ, Kassis AI (1996) Kit formulation for the preparation of radiolabeled iododeoxyuridine by demetallation. J Nucl Med 37:1S–3S
Dupertuis YM, Vazquez M, Mach JP et al (2001) Fluorodeoxyuridine improves imaging of human glioblastoma xenografts with radiolabeled iododeoxyuridine. Cancer Res 61:7971–7977
Marques JP, Maddage R, Mlynarik V, Gruetter R (2009) On the origin of the MR image phase contrast: an in vivo MR microscopy study of the rat brain at 14.1 T. Neuroimage 46:345–352
Yamamoto Y, Nishiyama Y, Ishikawa S et al (2008) 3′-Deoxy-3′-18F-fluorothymidine as a proliferation imaging tracer for diagnosis of lung tumors: comparison with 2-deoxy-2-18f-fluoro-d-glucose. J Comput Assist Tomogr 32:432–437
Barwick T, Bencherif B, Mountz JM, Avril N (2009) Molecular PET and PET/CT imaging of tumour cell proliferation using F-18 fluoro-l-thymidine: a comprehensive evaluation. Nucl Med Commun 30:908–917
Direcks WG, Berndsen SC, Proost N et al (2008) [18F]FDG and [18F]FLT uptake in human breast cancer cells in relation to the effects of chemotherapy: an in vitro study. Br J Cancer 99:481–487
Dupertuis YM, Xiao WH, De Tribolet N et al (2002) Unlabelled iododeoxyuridine increases the rate of uptake of [125I]iododeoxyuridine in human xenografted glioblastomas. Eur J Nucl Med Mol Imaging 29:499–505
Dittmann H, Dohmen BM, Kehlbach R et al (2002) Early changes in [18F]FLT uptake after chemotherapy: an experimental study. Eur J Nucl Med Mol Imaging 29:1462–1469
Leichman L, Silberman H, Leichman CG et al (1992) Preoperative systemic chemotherapy followed by adjuvant postoperative intraperitoneal therapy for gastric cancer: a University of Southern California pilot program. J Clin Oncol 10:1933–1942
Sullivan RD, Miller E (1965) The clinical effects of prolonged intravenous infusion of 5-fluoro-2′-deoxyuridine. Cancer Res 25:1025–1033
Acknowledgments
Major support of this work by the Swiss National Science Foundation Grant Nr3100AO-110023 is thankfully acknowledged. We express our gratitude to Mrs. Frances Godson for reviewing the manuscript. We also acknowledge the technical assistance of the laboratory of the Institute of Pathology at the University Hospital of Lausanne. We gratefully acknowledge Drs. Sandrine Ding and Sébastien Baechler for their assistance in the statistical analysis.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Viertl, D., Bischof Delaloye, A., Lanz, B. et al. Increase of [18F]FLT Tumor Uptake In Vivo Mediated by FdUrd: Toward Improving Cell Proliferation Positron Emission Tomography. Mol Imaging Biol 13, 321–331 (2011). https://doi.org/10.1007/s11307-010-0368-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-010-0368-z