Abstract
Purpose
Overexpression of the epidermal growth factor receptor (EGFR) occurs with high incidence in various carcinomas. The oncogenic expression of the receptor has been exploited for immunoglobulin-based diagnostics and therapeutics. We describe the use of a llama single-domain antibody fragment, termed Nanobody®, for the in vivo radioimmunodetection of EGFR overexpressing tumors using single photon emission computed tomography (SPECT) in mice.
Methods
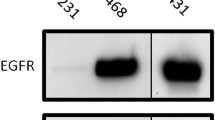
Fluorescence-activated cell sorting (FACS) analysis was performed to evaluate the specificity and selectivity of 8B6 Nanobody to bind EGFR on EGFR overexpressing cells. The Nanobody was then labeled with 99mTc via its C-terminal histidine tail. Uptake in normal organs and tissues was assessed by ex vivo analysis. In vivo tumor targeting of 99mTc-8B6 Nanobody was evaluated via pinhole SPECT in mice bearing xenografts of tumor cells with either high (A431) or moderate (DU145) overexpression of EGFR.
Results
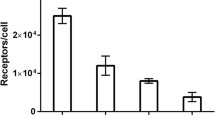
FACS analysis indicated that the 8B6 Nanobody only recognizes cells overexpressing EGFR. In vivo blood clearance of 99mTc-8B6 Nanobody is relatively fast (half-life, 1.5 h) and mainly via the kidneys. At 3 h postinjection, total kidney accumulation is high (46.6 ± 0.9%IA) compared to total liver uptake (18.9 ± 0.6%IA). Pinhole SPECT imaging of mice bearing A431 xenografts showed higher average tumor uptake (5.2 ± 0.5%IA/cm3) of 99mTc-8B6 Nanobody compared to DU145 xenografts (1.8 ± 0.3%IA/cm3, p < 0.001).
Conclusion
The EGFR-binding Nanobody investigated in this study shows high specificity and selectivity towards EGFR overexpressing cells. Pinhole SPECT analysis with 99mTc-8B6 Nanobody enabled in vivo discrimination between tumors with high and moderate EGFR overexpression. The favorable biodistribution further corroborates the suitability of Nanobodies for in vivo tumor imaging.






Similar content being viewed by others
References
Beekman F, Van der Have F (2006) The pinhole: gateway to ultra-high-resolution three-dimensional radionuclide imaging. Eur J Nucl Med Mol Imaging 34:151–161
Weissleder R (2006) Molecular imaging in cancer. Science 312:1168–1171
Van de Wiele C, Revets H, Mertens N (2004) Radioimmunoimaging. Advances and prospects. Q J Nucl Med Mol Imaging 48:317–325
Kenanova V, Wu AM (2006) Tailoring antibodies for radionuclide delivery. Expert Opin Drug Deliv 3:53–70
Mallender WD, Carrero J, Voss EWJ (1996) Comparative properties of the single chain antibody and Fv derivatives of mAb 4–4–20. Relationship between interdomain interactions and the high affinity for fluorescein ligand. J Biol Chem 271:5338–5346
Whitlow M, Bell BA, Feng SL, et al. (1993) An improved linker for single-chain Fv with reduced aggregation and enhanced proteolytic stability. Protein Eng 6:989–995
Hamers-Casterman C, Atarhouch T, Muyldermans S, et al. (1993) Naturally occurring antibodies devoid of light chains. Nature 363:446–448
Nguyen VK, Desmyter A, Muyldermans S (2001) Functional heavy-chain antibodies in Camelidae. Adv Immunol 79:261–296
Huang L, Reekmans G, Saerens D, et al. (2005) Prostate-specific antigen immunosensing based on mixed self-assembled monolayers, camel antibodies and colloidal gold enhanced sandwich assays. Biosens Bioelectron 21:483–490
Revets H, De Baetselier P, Muyldermans S (2005) Nanobodies as novel agents for cancer therapy. Expert Opin Biol Ther 5:111–124
Davies J, Riechmann L (1996) Single antibody domains as small recognition units: design and in vitro antigen selection of camelized, human VH domains with improved protein stability. Protein Eng 9:531–537
Arbabi Ghahroudi M, Desmyter A, Wyns L, Hamers R, Muyldermans S (1997) Selection and identification of single domain antibody fragments from camel heavy-chain antibodies. FEBS Lett 414:521–526
Frenken LG, van der Linden RH, Hermans PW, et al. (2000) Isolation of antigen specific llama VHH antibody fragments and their high level secretion by Saccharomyces cerevisiae. J Biotechnol 78:11–21
Joosten V, Gouka RJ, van den Hondel CA, Verrips CT, Lokman BC (2005) Expression and production of llama variable heavy-chain antibody fragments (VHHs) by Aspergillus awamori. Appl Microbiol Biotechnol 66:384–392
Ismaili A, Jalali-Javaran M, Rasaee MJ, Rahbarizadeh F, Forouzandeh-Moghadam M, Memari HR (2007) Production and characterization of anti-(mucin MUC1) single-domain antibody in tobacco (Nicotiana tabacum cultivar Xanthi). Biotechnol Appl Biochem 47:11–19
Abulrob A, Sprong H, en Henegouwen PVB, Stanimirovic D (2005) The blood-brain barrier transmigrating single domain antibody: mechanisms of transport and antigenic epitopes in human brain endothelial cells. J Neurochem 95:1201–1214
Cortez-Retamozo V, Lauwereys M, Gholamreza Hassanzadeh G, et al. (2002) Efficient tumor targeting by single-domain antibody fragments of camels. Int J Cancer 98:456–462
Nguyen VK, Hamers R, Wyns L, Muyldermans S (2000) Camel heavy-chain antibodies: diverse germline V(H)H and specific mechanisms enlarge the antigen-binding repertoire. EMBO J 19:921–930
Roovers RC, Laeremans T, Huang L, et al. (2006) Efficient inhibition of EGFR signalling and of tumour growth by antagonistic anti-EGFR Nanobodies. Cancer Immunol Immunother 56:303–317
Ellis LM (2004) Epidermal growth factor receptor in tumor angiogenesis. Hematol Oncol Clin North Am 18:1007–1021
Grandis JR, Sok JC (2004) Signaling through the epidermal growth factor receptor during the development of malignancy. Pharmacol Ther 102:37–46
Kim H, Muller WJ (1999) The role of the epidermal growth factor receptor family in mammary tumorigenesis and metastasis. Exp Cell Res 253:78–87
Nicholson RI, Gee JM, Harper ME (2001) EGFR and cancer prognosis. Eur J Cancer 37:S9–S15
Haigler H, Ash JF, Singer SJ, Cohen S (1978) Visualization by fluorescence of the binding and internalization of epidermal growth factor in human carcinoma cells A-431. Proc Natl Acad Sci U S A 75:3317–3321
MacDonald A, Chisholm GD, Habib FK (1990) Production and response of a human prostatic cancer line to transforming growth factor-like molecules. Br J Cancer 62:579–584
Honegger AM, Dull TJ, Felder S, et al. (1987) Point mutation at the ATP binding site of EGF receptor abolishes protein-tyrosine kinase activity and alters cellular routing. Cell 51:199–209
Honegger AM, Schmidt A, Ullrich A, Schlessinger J (1990) Evidence for epidermal growth factor (EGF)-induced intermolecular autophosphorylation of the EGF receptors in living cells. Mol Cell Biol 10:4035–4044
Aguilar Z, Akita RW, Finn RS, et al. (1999) Biologic effects of heregulin/neu differentiation factor on normal and malignant human breast and ovarian epithelial cells. Oncogene 18:6050–6062
Desmyter A, Transue TR, Ghahroudi MA, et al. (1996) Crystal structure of a camel single-domain VH antibody fragment in complex with lysozyme. Nat Struct Biol 3:803–811
Vanhove C, Defrise M, Franken PR, Everaert H, Deconinck F, Bossuyt A (2000) Interest of the ordered subsets expectation maximization (OS-EM) algorithm in pinhole single-photon emission tomography reconstruction: a phantom study. Eur J Nucl Med 27:140–146
Loening AM, Gambhir SS (2003) AMIDE: a free software tool for multimodality medical image analysis. Mol Imaging 2:131–137
De Genst E, Silence K, Decanniere K, et al. (2006) Molecular basis for the preferential cleft recognition by dromedary heavy-chain antibodies. Proc Natl Acad Sci U S A 103:4586–4591
Smith JM, Sporn MB, Roberts AB, Derynck R, Winkler ME, Gregory H (1985) Human transforming growth factor-alpha causes precocious eyelid opening in newborn mice. Nature 315:515–516
Sundberg AL, Gedda L, Orlova A, et al. (2004) [177Lu]Bz-DTPA-EGF: Preclinical characterization of a potential radionuclide targeting agent against glioma. Cancer Biother Radiopharm 19:195–204
Tolmachev V, Orlova A, Wei Q, Bruskin A, Carlsson J, Gedda L (2004) Comparative biodistribution of potential anti-glioblastoma conjugates [111In]DTPA-hEGF and [111In]Bz-DTPA-hEGF in normal mice. Cancer Biother Radiopharm 19:491–501
Behr TM, Behe M, Becker W (1999) Diagnostic applications of radiolabeled peptides in nuclear endocrinology. Q J Nucl Med 43:268–280
Decristoforo C, Mather SJ (2002) The influence of chelator on the pharmacokinetics of 99mTc-labelled peptides. Q J Nucl Med 46:195–205
Ballinger JR, Cooper MS, Mather SJ (2004) Re: controversies–[Tc(CO)3] chemistry: a promising new concept for SPET? Eur J Nucl Med Mol Imaging 31:304–305
Orlova A, Magnusson M, Eriksson TLJ, et al. (2006) Tumor imaging using a picomolar affinity HER2 binding affibody molecule. Cancer Res 66:4339–4348
Cai W, Chen K, He L, Cao Q, Koong A, Chen X (2007) Quantitative PET of EGFR expression in xenograft-bearing mice using (64)Cu-labeled cetuximab, a chimeric anti-EGFR monoclonal antibody. Eur J Nucl Med Mol Imaging 34:850–858
Senekowitsch-Schmidtke R, Steiner K, Haunschild J, Mollenstadt S, Truckenbrodt R (1996) In vivo evaluation of epidermal growth factor (EGF) receptor density on human tumor xenografts using radiolabeled EGF and anti-(EGF receptor) mAb 425. Cancer Immunol Immunother 42:108–114
Reilly RM, Kiarash R, Sandhu J, et al. (2000) A comparison of EGF and MAb 528 labeled with 111In for imaging human breast cancer. J Nucl Med 41:903–911
Meenakshi A, Ganesh V, Suresh Kumar R, Siva Kumar N (2003) Radioimmuno targetting (99 m)technetium labeled anti-epidermal growth factor receptor monoclonal antibodies in experimental tumor models. Q J Nucl Med 47:139–144
Schechter NR, Yang DJ, Azhdarinia A, et al. (2003) Assessment of epidermal growth factor receptor with 99mTc-ethylenedicysteine-C225 monoclonal antibody. Anticancer Drugs 14:49–56
Pnwar P, Iznaga-Escobar N, Mishra P, et al. (2005) Radiolabeling and biological evaluation of DOTA-Ph-Al derivative conjugated to anti-EGFR antibody ior egf/r3 for targeted tumor imaging and therapy. Cancer Biol Ther 4:854–860
Torres LA, Perera A, Batista JF, et al. (2005) Phase I/II clinical trial of the humanized anti-EGF-r monoclonal antibody h-R3 labeled with 99mTc in patients with tumour of epithelial origin. Nucl Med Commun 26:1049–1057
Goldenberg A, Masui H, Divgi C, Kamrath H, Pentlow K, Mendelsohn J (1989) Imaging of human tumor xenografts with an indium-111-labeled anti-epidermal growth factor receptor monoclonal antibody. J Natl Cancer Inst 81:1616–1625
Steiner K, Haunschild J, Faro HP, Senekowitsch R (1995) Distribution of humanized MAb 425 (EMD 62,000) in rats and specific localization in tumor-bearing nude mice. Cell Mol Biol (Noisy-le-grand) 41:179–184
Scopinaro F, De Vincentis G, Banci M, et al. (1997) In vivo study of a technetium labeled anti-EGFr MoAB. Anticancer Res 17:1761–1765
Morales-Morales A, Duconge J, Caballero-Torres I, Nunez-Gandolff G, Fernandez E, Iznaga-Escobar N (1999) Biodistribution of 99mTc-labeled anti-human epidermal growth factor receptor (EGF-R) humanized monoclonal antibody h-R3 in a xenograft model of human lung adenocarcinoma. Nucl Med Biol 26:275–279
Schillaci O, Danieli R, Picardi V, Bagni O, Di Loreto M, Scopinaro F (2001) Immunoscintigraphy with a technetium-99m labeled anti-epithelial growth factor receptor antibody in patients with non-small cell lung cancer. Anticancer Res 21:3571–3574
Panousis C, Rayzman VM, Johns TG, et al. (2005) Engineering and characterisation of chimeric monoclonal antibody 806 (ch806) for targeted immunotherapy of tumors expressing de2–7 EGFR or amplified EGFR. Br J Cancer 92:1069–1077
Perk LR, Visser GWM, Vosjan MJWD, et al. (2005) (89)Zr as a PET surrogate radioisotope for scouting biodistribution of the therapeutic radiometals (90)Y and (177)Lu in tumor-bearing nude mice after coupling to the internalizing antibody cetuximab. J Nucl Med 46:1898–1906
Rusckowski M, Qu T, Chang F, Hnatowich DJ (1997) Technetium-99m labeled epidermal growth factor-tumor imaging in mice. J Pept Res 50:393–401
Reilly RM, Kiarash R, Cameron RG, et al. (2000) 111In-labeled EGF is selectively radiotoxic to human breast cancer cells overexpressing EGFR. J Nucl Med 41:429–438
Abourbeh G, Dissoki S, Jacobson O, et al. (2007) Evaluation of radiolabeled ML04, a putative irreversible inhibitor of epidermal growth factor receptor, as a bioprobe for PET imaging of EGFR-overexpressing tumors. Nucl Med Biol 34:55–70
Friedman M, Nordberg E, Höidén-Guthenberg I, et al. (2007) Phage display selection of Affibody molecules with specific binding to the extracellular domain of the epidermal growth factor receptor. Protein Eng Des Sel 20:189–199
Tolmachev V, Orlova A, Nilsson FY, Feldwisch J, Wennborg A, Abrahmsen L (2007) Affibody molecules: potential for in vivo imaging of molecular targets for cancer therapy. Expert Opin Biol Ther 7:555–568
Lundberg E, Hoiden-Guthenberg I, Larsson B, Uhlen M, Graslund T (2007) Site-specifically conjugated anti-HER2 Affibody molecules as one-step reagents for target expression analyses on cells and xenograft samples. J Immunol Methods 319:53–63
Orlova A, Tolmachev V, Pehrson R, et al. (2007) Synthetic affibody molecules: a novel class of affinity ligands for molecular imaging of HER2-expressing malignant tumors. Cancer Res 67:2178–2186
Steffen A-C, Orlova A, Wikman M, et al. (2006) Affibody-mediated tumour targeting of HER-2 expressing xenografts in mice. Eur J Nucl Med Mol Imaging 33:631–638
Britten CD (2004) Targeting ErbB receptor signaling: a pan-ErbB approach to cancer. Mol Cancer Ther 3:1335–1342
Cunningham D, Humblet Y, Siena S, et al. (2004) Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med 351:337–345
Vincenzi B, Santini D, Rabitti C, et al. (2006) Cetuximab and irinotecan as third-line therapy in advanced colorectal cancer patients: a single centre phase II trial. Br J Cancer 94:792–797
Chung KY, Shia J, Kemeny NE, et al. (2005) Cetuximab shows activity in colorectal cancer patients with tumors that do not express the epidermal growth factor receptor by immunohistochemistry. J Clin Oncol 23:1803–1810
Dei Tos AP, Ellis I (2005) Assessing epidermal growth factor receptor expression in tumors: what is the value of current test methods? Eur J Cancer 41:1383–1392
Fruehauf J (2006) EGFR function and detection in cancer therapy. J Exp Ther Oncol 5:231–246
Penault-Llorca F, Cayre A, Arnould L, et al. (2006) Is there an immunohistochemical technique definitively valid in epidermal growth factor receptor assessment? Oncol Rep 16:1173–1179
Atkins D, Reiffen K-A, Tegtmeier CL, Winther H, Bonato MS, Storkel S (2004) Immunohistochemical detection of EGFR in paraffin-embedded tumor tissues: variation in staining intensity due to choice of fixative and storage time of tissue sections. J Histochem Cytochem 52:893–901
Hoff J (2000) Methods of blood collection in the mouse. Lab Anim 29:47–53
Acknowledgments
L.H. is supported by the Institute for the Promotion of Innovation through Science and Technology in Flanders (IWT-Vlaanderen). L.O.T.G. is a Ph.D. fellow of the Horizontal Onderzoek Axis (HOA) of the VUB. T.L. is a Senior Clinical Investigator of the Research Foundation–Flanders (Belgium) (FWO). The research at ICMI is funded by the Interuniversity Attraction Poles Programme–Belgian State–Belgian Science Policy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, L., Gainkam, L.O.T., Caveliers, V. et al. SPECT Imaging with 99mTc-Labeled EGFR-Specific Nanobody for In Vivo Monitoring of EGFR Expression. Mol Imaging Biol 10, 167–175 (2008). https://doi.org/10.1007/s11307-008-0133-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-008-0133-8