Abstract
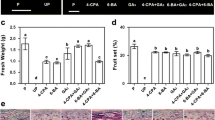
Application of exogenous gibberellin (GA) has been widely used to improve fruit set in grapevine, although the underlying molecular mechanism remains unknown. In this work, GA application at 10 days before anthesis promoted fruit set of grapevine “Fenghou” (Vitis vinifera × V. labrusca) and increased sugar content and acid invertase activity in the ovary, especially vacuolar invertase (VIN) activity during fruit set, compared with the control. However, the GA biosynthesis inhibitor paclobutrazol (PAC) had the opposite effect. Tissue-specific expression analysis and in situ hybridization showed that transcripts of three acid invertase genes were primarily detected in the phloem of the flower and fruitlet. In contrast to the expression of the cell wall invertase gene, which was only induced before blooming, transcript accumulation of the VIN genes VvGIN1 and VvGIN2 was enhanced during fruit set by GA application. Nevertheless, PAC treatment reduced VvGIN1 transcript accumulation after anthesis, but had no significant effect on the VvGIN2 transcript level at the same time point. Interestingly, the effect of GA and PAC application on expression of VvPIN1-like and VvPIN3-like (auxin transporters) was similar to that on VvGIN1 after blooming. In addition, the inducibility of the VvGIN1 and VvGIN2 promoters by GA was demonstrated using transient expression assays in tobacco leaves. These results indicated that enhanced VIN activity and carbohydrate import into grapevine fruitlets by GA application contribute to the increased fruit set through increasing sink strength and sugar signaling activities, possibly by regulating polar auxin transport.








Similar content being viewed by others
References
Albacete A, Canteronavarro E, Balibrea ME, Großkinsky DK, Smigocki AC, Roitsch T, Pérezalfocea F (2014) Hormonal and metabolic regulation of tomato fruit sink activity and yield under salinity. J Exp Bot 65:6081–6095
Antunes P, Paulo L, Barbosa AR, Anjos O (2012) Honey sugars analysis by ion chromatography method with integrated pulsed amperometric detection (IPAD). 7o Encontro Nacional de Cromatografia
Azuma T, Ueno S, Uchida N, Yasuda T (1997) Gibberellin-induced elongation and osmoregulation in internodes of floating rice. Physiol Plant 99:517–522
Balibrea Lara ME, Gonzalez Garcia MC, Fatima T, Ehness R, Lee T, Proels R, Tanner W, Roitsch T (2004) Extracellular invertase is an essential component of cytokinin-mediated delay of senescence. Plant Cell 16:1276–1287
Boyer JS, Mclaughlin JE (2007) Functional reversion to identify controlling genes in multigenic responses: analysis of floral abortion. J Exp Bot 58:267–277
Chan JJ, Hur YY, Jung SM, Noh JH, Do GR, Park SJ, Nam JC, Park KS, Hwang HS, Choi D (2014) Transcriptional changes of gibberellin oxidase genes in grapevines with or without gibberellin application during inflorescence development. J Plant Res 127:359–371
Coombe BG (1965) Increase in fruit set of Vitis vinifera by treatment with growth retardants. Nature 205:305–306
Dauelsberg P, Matus JT, Poupin MJ, Leiva-Ampuero A, Godoy F, Vega A, Arce-Johnson P (2011) Effect of pollination and fertilization on the expression of genes related to floral transition, hormone synthesis and berry development in grapevine. J Plant Physiol 168:1667–1674
Davies C, Robinson SP (1996) Sugar accumulation in grape berries. Cloning of two putative vacuolar invertase cDNAs and their expression in grapevine tissues. Plant Physiol 111:275–283
Dominguez F, Cejudo F (1999) Patterns of starchy endosperm acidification and protease gene expression in wheat grains following germination. Plant Physiol 119:81–87
Drews GN, Bowman JL, Meyerowitz EM (1991) Negative regulation of the Arabidopsis homeotic gene AGAMOUS by the APETALA2 product. Cell 65:991–1002
Fridman E, Liu YD (2004) Zooming in on a quantitative trait for tomato yield using interspecific introgressions. Science 305:1786–1789
Fuentes S, Ljung K, Sorefan K, Alvey E, Harberd NP, Ostergaard L (2012) Fruit growth in Arabidopsis occurs via DELLA-dependent and DELLA-independent gibberellin responses. Plant Cell 24:3982–3996. doi:10.1105/tpc.112.103192
Gubler F, Jacobsen JV (1992) Gibberellin-responsive elements in the promoter of a barley high-pl α-amylase gene. Plant Cell 4:1435–1441
Hayes MA, Feechan A, Dry IB (2010) Involvement of abscisic acid in the coordinated regulation of a stress-inducible hexose transporter (VvHT5) and a cell wall invertase in grapevine in response to biotrophic fungal infection. Plant Physiol 153:211–221
Hedden P, Graebe JE (1985) Inhibition of gibberellin biosynthesis by paclobutrazol in cell-free homogenates of Cucurbita maxima endosperm and Malus pumila embryos. J Plant Growth Regul 4:111–122
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database: 1999. Nucleic Acids Res 27:297–300
Hironori I, Miyako UT, Yutaka S, Motoyuki A, Makoto M (2002) The gibberellin signaling pathway is regulated by the appearance and disappearance of SLENDER RICE1 in nuclei. Plant Cell 14:57–70
Intrigliolo DS, Lakso AN (2009) Berry abscission is related to berry growth in Vitis labruscana ‘Concord’ and Vitis vinifera ‘Riesling’. Vitis 48:53–54
Kang JY, Choi HI, Im MY, Kim SY (2002) Arabidopsis basic leucine zipper proteins that mediate stress-responsive abscisic acid signaling. Plant Cell 14:343–357
Lecourieux F, Kappel C, Lecourieux D, Serrano A, Torres E, Arcejohnson P, Delrot S (2014) An update on sugar transport and signalling in grapevine. J Exp Bot 65:821–832
Liu HF, Wu BH, Fan PG, Li SH, Li LS (2006) Sugar and acid concentrations in 98 grape cultivars analyzed by principal component analysis. Journal of the Science of Food & Agriculture 86:1526–1536
Liu YH, Offler CE, Ruan YL (2016) Cell wall invertase promotes fruit set under heat stress by suppressing ROS-independent cell death. Plant Physiol 172
Lu L, Liang J, Zhu X, Xiao K, Li T, Hu J (2016) Auxin- and cytokinin-induced berries set in grapevine partly rely on enhanced gibberellin biosynthesis. Tree Genet Genomes 12:1–12
Miller GAIL (1959) Use of dinitrosalicylic acid reagent for detection of reducing sugars. Anal Chem 31
Mishra BS, Manjul S, Priyanka A, Ashverya L (2009) Glucose and auxin signaling interaction in controlling Arabidopsis thaliana seedlings root growth and development. PLoS One 4:e4502–e4502
Mitsuhashi W, Sasaki S, Kanazawa A, Yang Y, Kamiya Y, Toyomasu T (2004) Differential expression of acid invertase genes during seed germination in Arabidopsis thaliana. Biosci Biotechnol Biochem 68:602–608
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4325
Nathalie K, Carlos A, Francisca G, Serge D, Patricio A-J (2014) Differential behavior within a grapevine cluster: decreased ethylene-related gene expression dependent on auxin transport is correlated with low abscission of first developed berries. PLoS One 9:e111258–e111258
Nonis A, Ruperti B, Pierasco A, Canaguier A, Adam-Blondon AF, Gaspero GD, Vizzotto G (2008) Neutral invertases in grapevine and comparative analysis with Arabidopsis, poplar and rice. Planta 229:129–142
Pérez FJ, Gómez M (2000) Possible role of soluble invertase in the gibberellic acid berry-sizing effect in Sultana grape. Plant Growth Regul 30:111–116
Proels RK, Gonzalez MC, Roitsch T (2006) Gibberellin-dependent induction of tomato extracellular invertase Lin7 is required for pollen development. Funct Plant Biol 33:547–554
Rabot A, Portemer V, Péron T, Mortreau E, Leduc N, Hamama L, Coutos-Thévenot P, Atanassova R, Sakr S, Le GJ (2014) Interplay of sugar, light and gibberellins in expression of Rosa hybrida vacuolar invertase 1 regulation. Plant Cell Physiol 55:1734–1748
Riou-Khamlichi C, Menges M, Healy JM, Murray JA (2000) Sugar control of the plant cell cycle: differential regulation of Arabidopsis D-type cyclin gene expression. Molecular & Cellular Biology 20:4513–4521
Roitsch T, Gonzalez MC (2004) Function and regulation of plant invertases: sweet sensations. Trends Plant Sci 9:606–613
Ruan Y-L, Patrick JW, Bouzayen M, Osorio S, Fernie AR (2012) Molecular regulation of seed and fruit set. Trends Plant Sci 17:656–665. doi:10.1016/j.tplants.2012.06.005
Ruffner HP, Hurlimann M, Skrivan R (1995) Soluble invertase from grape berries: purification, deglycosylation and antibody specificity. Plant Physiol Biochem 33:25–31
Serrani JC, Fos M, Atarés A, García-Martínez JL (2007) Effect of gibberellin and auxin on parthenocarpic fruit growth induction in the cv Micro-Tom of tomato. J Plant Growth Regul 26:211–221
Serrani JC, Ruiz-Rivero O, Fos M, García-Martínez JL (2008) Auxin-induced fruit-set in tomato is mediated in part by gibberellins. Plant J 56:922–934
Skeffington AW, Graf A, Duxbury Z, Gruissem W, Smith AM (2014) Glucan, water dikinase exerts little control over starch degradation in Arabidopsis leaves at night. Plant Physiol 165:866–879
Skriver K, Olsen FL, Rogers JC, Mundy J (1991) Cis-acting DNA elements responsive to gibberellin and its antagonist abscisic acid. Proc Natl Acad Sci U S A 88:7266–7270
Sturm A (1999) Invertases. Primary structures, functions, and roles in plant development and sucrose partitioning. Plant Physiol 121:1–8
Sutoh K, Yamauchi D (2003) Two cis-acting elements necessary and sufficient for gibberellin-upregulated proteinase expression in rice seeds. Plant J 34:635–645(611)
Tymowska-Lalanne Z, Kreis M (1998) The plant invertases: physiology, biochemistry and molecular biology. Adv Bot Res 28:71–117
Vignault C, Vachaud M, Cakir B, Glissant D, Dédaldéchamp F, Büttner M, Atanassova R, Fleurat-Lessard P, Lemoine R, Delrot S (2005) VvHT1 encodes a monosaccharide transporter expressed in the conducting complex of the grape berry phloem. J Exp Bot 56:1409–1418
Vivian-Smith A, Koltunow AM (1999) Genetic analysis of growth-regulator-induced parthenocarpy in Arabidopsis. Plant Physiol 121:437–452
Wang L, Ruan YL (2013) Regulation of cell division and expansion by sugar and auxin signaling. Front Plant Sci 4:163–163
Weaver RJ (1958) Effect of gibberellic acid on fruit set and berry enlargement in seedless grapes of Vitis vinifera. Nature 181:851–852
Weigel D, Glazebrook J (2006) Transformation of agrobacterium using the freeze-thaw method. Cold Spring Harbor Protocols 2006
William P, John P, Yong-Ling R (2015) Tomato ovary-to-fruit transition is characterized by a spatial shift of mRNAs for cell wall invertase and its inhibitor with the encoded proteins localized to sieve elements. Mol Plant 8:315–328
Wu LL, Mitchell JP, Cohn NS, Kaufman PB (1993) Gibberellin (GA3) enhances cell wall invertase activity and mRNA levels in elongating dwarf pea (Pisum sativum) shoots. Int J Plant Sci 154:280–289
Ye J, Di-An N, Yong-Ling R (2009) Posttranslational elevation of cell wall invertase activity by silencing its inhibitor in tomato delays leaf senescence and increases seed weight and fruit hexose level. Plant Cell 21:2072–2089
Yuan TT, Xu HH, Zhang KX, Guo TT, Lu YT (2013) Glucose inhibits root meristem growth via ABA INSENSITIVE 5, which represses PIN1 accumulation and auxin activity in Arabidopsis. Plant Cell Environ 37:1338–1350
Zanor MI, Osorio S, Nunes-Nesi A, Carrari F, Lohse M, Usadel B, Kühn C, Bleiss W, Giavalisco P, Willmitzer L (2009) RNA interference of LIN5 in tomato confirms its role in controlling Brix content, uncovers the influence of sugars on the levels of fruit hormones, and demonstrates the importance of sucrose cleavage for normal fruit development and fertility. Plant Physiol 150:1204–1218
Zhang C, Yoshida A (2005) 13C-photosynthate accumulation in Japanese pear fruit during the period of rapid fruit growth is limited by the sink strength of fruit rather than by the transport capacity of the pedicel. J Exp Bot 56:2713–2719
Zhang XY, Wang XL, Wang XF, Xia GH, Pan QH, Fan RC, Wu FQ, Yu XC, Zhang DP (2006) A shift of phloem unloading from symplasmic to apoplasmic pathway is involved in developmental onset of ripening in grape berry. Plant Physiol 142:220–232
Zhang C, Tanabe K, Tamura F, Itai A, Yoshida M (2007) Roles of gibberellins in increasing sink demand in Japanese pear fruit during rapid fruit growth. Plant Growth Regul 52:161–172
Ziliotto F, Corso M, Rizzini FM, Rasori A, Botton A, Bonghi C (2012) Grape berry ripening delay induced by a pre-véraison NAA treatment is paralleled by a shift in the expression pattern of auxin- and ethylene-related genes. BMC Plant Biol 12:55–60
Acknowledgments
The National Natural Science Foundation of China (31471842) supported this research.
Data Archiving Statement
The sequences of the VvCWIN, VvVINs, VvNIs, and VvHT1 reported here are available in GenBank (http://www.ncbi.nlm.nih.gov/): VvCWIN (GenBank accession no. NM_001281279); VvGIN1 (GenBank accession no. XM_002265498); VvGIN2 (GenBank accession no. XM_002272773); VvNI1 (GenBank accession no. AM930846); VvNI2 (GenBank accession no. AM930848); VvNI3 (GenBank accession no. AM930850); VvNI4 (GenBank accession no. XM_003632216.2); VvNI5 (GenBank accession no. XM_010660432); VvNI6 (GenBank accession no. XM_002275612); VvNI7 (GenBank accession no. XM_002271883); VvNI8 (GenBank accession no. XM_002264250); VvNI9 (GenBank accession no. XM_002269598); VvHT1 (GenBank accession no. NM_001281111).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Troggio
Electronic supplementary material
ESM 1
(DOCX 742 kb)
Rights and permissions
About this article
Cite this article
Lu, L., Liang, J., Chang, X. et al. Enhanced vacuolar invertase activity and capability for carbohydrate import in GA-treated inflorescence correlate with increased fruit set in grapevine. Tree Genetics & Genomes 13, 21 (2017). https://doi.org/10.1007/s11295-017-1109-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11295-017-1109-0