Abstract
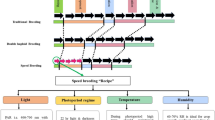
Horticultural crop improvement would benefit from a standardized, systematic, and statistically robust procedure for validating quantitative trait loci (QTLs) in germplasm relevant to breeding programs. Here, we describe and demonstrate a strategy for developing reference germplasm sets of perennial, clonally propagated crops, especially those with long juvenile periods. Germplasm is chosen to efficiently represent important members of larger pedigree-connected genepools. To facilitate validation of multiple QTLs, genome-wide representation of alleles is optimized for designated important breeding parents (IBPs) by estimating average allelic representation in relatives. The strategy and arising principles were demonstrated in a simulated germplasm set. Strong statistical power can be achieved with a carefully chosen germplasm set composed of IBPs, their numerous unselected progenies and close relatives, and all available founders and intermediate ancestors. Crop Reference Sets were developed in the marker-assisted breeding (MAB)-enabling “RosBREED” project as a base resource for QTL validation in US breeding germplasm of apple (Malus × domestica), peach (Prunus persica), and sweet cherry (Prunus avium) consisting of 467, 452, and 268 individuals, respectively. These sets adequately represent the most designated IBPs, have distinct advantages for QTL validation over other germplasm arrangements of equal size, and are recommended as a base resource for QTL validation by breeders of these US crops. The strategy described here can be used to develop efficient reference germplasm sets suiting other breeding genepools or to calculate the statistical power for QTL validation of germplasm sets already established.







Similar content being viewed by others
References
Aranzana M, Abbassi E-K, Howad W, Arús P (2010) Genetic variation, population structure and linkage disequilibrium in peach commercial varieties. BMC Genet 11:69
Bink M, Boer M, ter Braak C, Jansen J, Voorrips R, van de Weg W (2008) Bayesian analysis of complex traits in pedigreed plant populations. Euphytica 161:85–96
Bink MCAM, Jansen J, Madduri M, Voorrips RE, Durel C-E, Kouassi AB, Laurens F, Mathis F, Gessler C, Gobbin D, Rezzonico F, Patocchi A, Kellerhals M, Boudichevskaia A, Dunemann F, Peil A, Nowicka A, Lata B, Stankiewicz-Kosyl M, Jeziorek K, Pitera E, Soska A, Tomala K, Evans KM, Fernández-Fernández F, Guerra W, Korbin M, Keller S, Lewandowski M, Plocharski W, Rutkowski K, Zurawicz E, Costa F, Sansavini S, Tartarini S, Komjanc M, Mott D, Antofie A, Lateur M, Rondia A, Gianfranceschi L, van de Weg WE (2014) Bayesian QTL analyses using pedigreed families of an outcrossing species, with application to fruit firmness in apple. Theor Appl Genet 127:1073–1090
Brown SK, Terry DE (1997) The Cornell apple breeding program: past, present and future. Fruit Var J 51:199–204
Chagné D, Crowhurst RN, Troggio M, Davey MW, Gilmore B, Lawley C, Vanderzande S, Hellens RP, Kumar S, Cestaro A, Velasco R, Main D, Rees J, Iezzoni A, Mockler T, Wilhelm L, van de Weg E, Gardiner SE, Bassil N, Peace C (2012) Genome-wide SNP detection, validation, and development of an 8K SNP array for apple. PLoS ONE 7(2):e31745
Chambers A, Whitaker VM, Gibbs B, Plotto A, Folta KM (2012) Detection of the linalool-producing NES1 variant across diverse strawberry (Fragaria spp.) accessions. Plant Breed 131:437–443
Costa F, van de Weg WE, Stella S, Dondini L, Pratesi D, Musacchi S, Sansavini S (2008) Map position and functional allelic diversity of Md-Exp7, a new putative expansin gene associated with fruit softening in apple (Malus × domestica Borkh.) and pear (Pyrus communis). Tree Genet Genomes 4:575–586
Dunemann F, Ulrich D, Malysheva-Otto L, Weber WE, Longhi S, Velasco R, Costa F (2012) Functional allelic diversity of the apple alcohol acyl-transferase gene MdAAT1 associated with fruit ester volatile contents in apple cultivars. Mol Breed 29:609–625
Edge-Garza DA, Zhu Y, Peace CP (2010) Enabling marker-assisted seedling selection in the Washington Apple Breeding Program. Acta Hort 859:369–373
Evans K (2013) Apple breeding in the Pacific Northwest. Acta Horticult 976:75–78
Evans KM, Patocchi A, Rezzonico F, Mathis F, Durel CE, Fernández-Fernández F, Boudichevskaia A, Dunemann F, Stankiewicz-Kosyl M, Gianfranceschi L, Komjanc M, Lateur M, Madduri M, Noordijk Y, Van de Weg WE (2011) Genotyping of pedigreed apple breeding material with a genome covering set of SSRs: trueness to type of cultivars and their parentages. Mol Breed 28:535–547
Evans KM, Barritt BH, Konishi BS, Brutcher LJ, Ross CF (2012a) ‘WA 38’ apple. HortSci 47:1177–1179
Evans K, Luby J, Brown S, Clark M, Guan Y, Orcheski B, Schmitz C, Peace C, van de Weg E, Iezzoni A (2012b) Large-scale standardized phenotyping of apple in RosBREED. Acta Horticult 945:233–238
Folta K, Gardiner S (2009) Genetics and genomics of Rosaceae. Springer, New York
Frett T, Gasic K, Clark J, Byrne D, Gradiel T, Crisosto C (2012) Standardized phenotyping for fruit quality in peach [Prunus persica (L.) Batsch]. J Am Pom Soc 66:214–219
Haldar S, Haendiges S, Edge-Garza DA, Oraguzie NC, Olmstead J, Peace CP, Iezzoni A (2010) Applying genetic markers for self-compatibility in the WSU sweet cherry breeding program. Acta Horticult 859:375–380
Iezzoni A, Weebadde C, Luby J, Yue CY, van de Weg E, Fazio G, Main D, Peace CP, Bassil NV, McFerson J (2010) RosBREED: enabling marker-assisted breeding in Rosaceae. Acta Horticult 859:389–394
Keller-Przybyłkowicz LM, Korbin M (2009) Molecular screening of apple (Malus domestica) cultivars and breeding clones for their resistance to fire blight. J Fruit Ornam Plant Res 17:31–43
Khan MA, Durel CE, Duffy B, Drouet D, Kellerhals M, Gessler C, Patocchi A (2007) Development of molecular markers linked to the ‘Fiesta’ linkage group 7 major QTL for fire blight resistance and their application for marker-assisted selection. Genome 50:568–577
Le Roux P-M F, Christen D, Duffy B, Tartarini S, Dondini L, Yamamoto T, Nishitani C, Terakami S, Lespinasse Y, Kellerhals M, Patocchi A (2012) Redefinition of the map position and validation of a major quantitative trait locus for fire blight resistance of the pear cultivar ‘harrow Sweet’ (Pyrus communis L.). Plant Breed 131:656–664
Lenth RV (2001) Some practical guidelines for effective sample size determination. Am Stat 55:187–193
Lenth RV (2006–9) Java applets for power and sample size. University of Iowa. http://homepage.stat.uiowa.edu/~rlenth/power. Accessed 4 April 2013
Longhi S, Hamblin MT, Trainotti L, Peace CP, Velasco R, Costa F (2013) A candidate gene based approach validates Md-PG1 as the main responsible for a QTL impacting fruit texture in apple (Malus × domestica Borkh). BMC Plant Biol 13:37
Luby J, Fennell A (2005) Fruit breeding for the Northern Great Plains at the University of Minnesota and South Dakota State University. HortSci 41:25–26
Mariette S, Tavaud M, Arunyawat U, Capdeville G, Millan M, Salin F (2010) Population structure and genetic bottleneck in sweet cherry estimated with SSRs and the gametophytic self-incompatibility locus. BMC Genet 11:77
Meuwissen THE, Luo Z (1992) Computing inbreeding coefficients in large populations. Genet Sel Evol 24:305–313
Nybom H, Ahmadi-Afzadi M, Sehic J, Hertog M (2013) DNA marker-assisted evaluation of fruit firmness at harvest and post-harvest fruit softening in a diverse apple germplasm. Tree Genet Genomes 9:279–290
Oraguzie NC, Iwanami H, Soejima J, Harada T, Hall A (2004) Inheritance of the Md-ACS1 gene and its relationship to fruit softening in apple (Malus × domestica Borkh.). Theor Appl Genet 108:1526–1533
Oraguzie NC, Volz RK, Whitworth CJ, Bassett HCM, Hall AJ, Gardiner SE (2007) Influence of Md-ACS1 allelotype and harvest season within an apple germplasm collection on fruit softening during cold air storage. Postharvest Biol Technol 44:212–219
Peace C, Bassil N, Main D, Ficklin S, Rosyara UR, Stegmeir T, Sebolt A, Gilmore B, Lawley C, Mockler TC, Bryant DW, Wilhelm L, Iezzoni A (2012) Development and evaluation of a genome-wide 6K SNP array for diploid sweet cherry and tetraploid sour cherry. PLoS One 7(12):e48305
Rosyara UR, Sebolt AM, Peace C, Iezzoni AF (2013) Identification of the paternal parent for the sweet cherry (Prunus avium L.) cultivar ‘Bing’ and confirmation of descendants using SNP markers. J Am Soc Hort Sci (submitted)
USDA, National Agricultural Statistics Service (2013) Noncitrus Fruits and Nuts 2012 Preliminary Summary. http://usda01.library.cornell.edu/usda/current/NoncFruiNu/NoncFruiNu-01-25-2013.pdf. Accessed 13 July 2013
Verde I, Bassil N, Scalabrin S, Gilmore B, Lawley CT, Gasic K, Micheletti D, Rosyara UR, Cattonaro F, Vendramin E, Main D, Aramini V, Blas AL, Mockler TC, Bryant DW, Wilhelm L, Troggio M, Sosinski B, Aranzana MJ, Arus P, Iezzoni A, Morgante M, Peace C (2012) Development and evaluation of a 9K SNP array for peach by internationally coordinated SNP detection and validation in breeding germplasm. PLoS ONE 7(4):e35668
Voorrips RE, Bink MCAM, van de Weg WE (2012) Pedimap: software for the visualization of genetic and phenotypic data in pedigrees. J Heredity 103:903–907
Zhu Y, Barritt BH (2008) Md-ACS1 and Md-ACO1 genotyping of apple (Malus × domestica Borkh.) breeding parents and suitability for marker-assisted selection. Tree Genet Genomes 4:555–562
Acknowledgments
We gratefully acknowledge RosBREED’s apple, peach, and sweet cherry “demonstration breeders” for making available vast troves of germplasm and for helping assemble pedigree information. Key germplasm was also kindly provided by interim curators of two National Plant Germplasm System collections, Gennaro Fazio of the Plant Genetic Resources Unit in Geneva, NY and Malli Aradhya of the National Clonal Germplasm Repository in Davis, CA. This work was funded by USDA’s National Institute of Food and Agriculture (NIFA)—specialty crop research initiative project, “RosBREED: Enabling marker-assisted breeding in Rosaceae” (2009-51181-05808) and was precipitated by grant number 2008-35300-04435 of the National Research Initiative of the USDA Cooperative State Research, Education and Extension Service (now NIFA).
Data archiving statement
A matrix of the AAR units provided by each individual included in the apple, peach, and sweet cherry Crop Reference Sets for each of the other individuals and ancestors of the sets was submitted to the Genome Database for Rosaceae (www.rosaceae.org).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Dirlewanger
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
Spreadsheet-based procedure to calculate average allelic representation of germplasm individuals, demonstrated with a simulated germplasm set (XLSX 324 kb)
Online Resource 2
The US Apple Crop Reference Set of 467 individuals, chosen to be representative of 57 important breeding parents (underlined), and their 70 known ancestors. Parentage according to pedigree records is indicated (“-” = unknown). Number of seedlings included for each family is indicated. For the cultivar McIntosh, two distinct types were included: ‘Marshall McIntosh’ and ‘Wijcik McIntosh’. AAR = average allelic representation of each individual achieved by the Set; Bal. = balanced, Uns. = unselected for typical breeding traits (PDF 768 kb)
Online Resource 3
The U.S. Peach Crop Reference Set of 452 individuals, chosen to be representative of 38 important breeding parents (underlined), and their 331 known ancestors. Parentage according to pedigree records is indicated (“-” = unknown). Number of seedlings included for each family is indicated. AAR = average allelic representation of each individual achieved by the Set; Bal. = balanced, Uns. = unselected for typical breeding traits (PDF 977 kb)
Online Resource 4
The U.S. Sweet Cherry Crop Reference Set of 268 individuals, chosen to be representative of 25 important breeding parents (underlined), and their 15 known ancestors. Parentage according to pedigree records is indicated (“-” = unknown). Number of seedlings included for each family is indicated. AAR = average allelic representation of each individual achieved by the Set; Bal. = balanced, Uns. = unselected for typical breeding traits (PDF 595 kb)
Online Resource 5
Guiding principles for establishing efficient germplasm sets for (discovery and) validation of existing alleles of a breeding program (PDF 83 kb)
Rights and permissions
About this article
Cite this article
Peace, C.P., Luby, J.J., van de Weg, W.E. et al. A strategy for developing representative germplasm sets for systematic QTL validation, demonstrated for apple, peach, and sweet cherry. Tree Genetics & Genomes 10, 1679–1694 (2014). https://doi.org/10.1007/s11295-014-0788-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11295-014-0788-z