Abstract
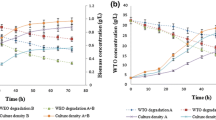
Kinetic studies and modeling of production parameters are essential for developing economical biosurfactant production processes. This study will provide a perspective on mechanistic reaction pathways to metabolize Waste Engine Oil (WEO). The results will provide relevant information on (i) WEO concentration above which growth inhibition occurs, (ii) chemical changes in WEO during biodegradation, and (iii) understanding of growth kinetics for the strain utilizing complex substrates. Laboratory scale experiments were conducted to study the kinetics and biodegradation potential of the strain Pseudomonas aeruginosa gi |KP 163922| over a range (0.5–8% (v/v)) of initial WEO concentration for 168 h. The kinetic models, such as Monod, Powell, Edward, Luong, and Haldane, were evaluated by fitting the experimental results in respective model equations. An unprecedented characterization of the substrate before and after degradation is presented, along with biosurfactant characterization. The secretion of biosurfactant during the growth, validated by surface tension reduction (72.07 ± 1.14 to 29.32 ± 1.08 mN/m), facilitated the biodegradation of WEO to less harmful components. The strain showed an increase in maximum specific growth rate (µmax) from 0.0185 to 0.1415 h−1 upto 49.92 mg/L WEO concentration. Maximum WEO degradation was found to be ~ 94% gravimetrically. The Luong model (adj. R2 = 0.97) adapted the experimental data using a non-linear regression method. Biochemical, 1H NMR, and FTIR analysis of the produced biosurfactant revealed a mixture of mono- and di- rhamnolipid. The degradation compounds in WEO were identified using FTIR, 1H NMR, and GC–MS analysis to deduce the mechanism.
Graphical abstract












Similar content being viewed by others
Data availability
No data was used for the research described in the article.
References
Abdel-Mawgoud AM, Lépine F, Déziel E (2014) A stereospecific pathway diverts β-oxidation intermediates to the biosynthesis of rhamnolipid biosurfactants. Chem Biol 21:156–164. https://doi.org/10.1016/j.chembiol.2013.11.010
Adebusoye SA, Ilori MO, Amund OO et al (2007) Microbial degradation of petroleum hydrocarbons in a polluted tropical stream. World J Microbiol Biotechnol 23:1149–1159. https://doi.org/10.1007/s11274-007-9345-3
Ahmad F, Jameel AT, Kamarudin MH, Mel M (2011) Study of growth kinetic and modeling of ethanol production by Saccharomyces cerevisae. African J Biotechnol 10:18842–18846. https://doi.org/10.5897/AJB11.2763
Ardestani F (2011) Investigation of the nutrient uptake and cell growth kinetics with monod and moser models for Penicillium brevicompactum ATCC 16024 in batch bioreactor. Iran J Energy Environ 2:117–121
Banat IM, Satpute SK, Cameotra SS et al (2014) Cost effective technologies and renewable substrates for biosurfactants’ production. Front Microbiol 5:1–19. https://doi.org/10.3389/fmicb.2014.00697
Bhattacharya M, Biswas D, Guchhait S (2018) Applied growth kinetic models for crude oil spill bioremediation in a batch scale bioreactor. J Environ Hazard 1(1):105–110.
Bhattacharya M, Biswas D, Sana S, Datta S (2014) Utilization of waste engine oil by Ochrobactrum pseudintermedium strain C1 that secretes an exopolysaccharide as a bioemulsifier. Biocatal Agric Biotechnol 3:167–176. https://doi.org/10.1016/j.bcab.2014.09.002
Bhattacharya M, Biswas D, Sana S, Datta S (2015) Biodegradation of waste lubricants by a newly isolated Ochrobactrum sp. C1. 3 Biotech 5:807–817. https://doi.org/10.1007/s13205-015-0282-9
Chong H, Li Q (2017) Microbial production of rhamnolipids: opportunities, challenges and strategies. Microb Cell Fact 16:1–12. https://doi.org/10.1186/s12934-017-0753-2
Das N, Chandran P (2011) Microbial degradation of petroleum hydrocarbon contaminants: an overview. Biotechnol Res Int 2011:1–13. https://doi.org/10.4061/2011/941810
Devaraj S, Sabapathy PC, Nehru L, Preethi K (2019) Bioprocess optimization and production of biosurfactant from an unexplored substrate: Parthenium hysterophorus. Biodegradation 30:325–334. https://doi.org/10.1007/s10532-019-09878-7
Dobler L, Vilela LF, Almeida RV, Neves BC (2016) Rhamnolipids in perspective : gene regulatory pathways, metabolic engineering, production and technological forecasting. N Biotechnol 33:123–135. https://doi.org/10.1016/j.nbt.2015.09.005
Dolman BM, Wang F, Winterburn JB (2019) Integrated production and separation of biosurfactants. Process Biochem 83:1–8. https://doi.org/10.1016/j.procbio.2019.05.002
Edwards VH (1970) The influence of high substrate concentrations on microbial kinetics. Biotechnol Bioeng 12:679–712. https://doi.org/10.1002/bit.260120504
Farias CBB, Almeida FCG, Silva IA et al (2021) Production of green surfactants: market prospects. Electron J Biotechnol 51:28–39. https://doi.org/10.1016/j.ejbt.2021.02.002
Fibriana F, Upaichit A, Cheirsilp B (2021) Low - cost production of cell—bound lipases by pure and co - culture of yeast and bacteria in palm oil mill effluent and the applications in bioremediation and biodiesel synthesis. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-021-02070-z
Förster E, Becker J, Dalitz F et al (2015) NMR Investigations on the aging of motor oils. Energy Fuels 29:7204–7212. https://doi.org/10.1021/acs.energyfuels.5b02164
Ganesh Kumar A, Vijayakumar L, Joshi G et al (2014) Biodegradation of complex hydrocarbons in spent engine oil by novel bacterial consortium isolated from deep sea sediment. Bioresour Technol 170:556–564. https://doi.org/10.1016/j.biortech.2014.08.008
Gaur S, Gupta S, Jain A (2021) Characterization and oil recovery application of biosurfactant produced during bioremediation of waste engine oil by strain Pseudomonas aeruginosa gi|KP 16392| isolated from Sambhar salt lake. Bioremediat J 25:308–325. https://doi.org/10.1080/10889868.2020.1871316
Gaur S, Sahani A, Chattopadhyay P et al (2022) Remediation of waste engine oil contaminated soil using rhamnolipid based detergent formulation. Mater Today Proc. https://doi.org/10.1016/j.matpr.2022.08.452
Ghazali FM, Rahman RNZA, Salleh AB, Basri M (2004) Biodegradation of hydrocarbons in soil by microbial consortium. Int Biodeterior Biodegradation 54:61–67. https://doi.org/10.1016/j.ibiod.2004.02.002
Gudina EJ, Rodrigues AI, Alves E et al (2015) Bioconversion of agro-industrial by-products in rhamnolipids toward applications in enhanced oil recovery and bioremediation. Bioresour Technol 177:87–93. https://doi.org/10.1016/j.biortech.2014.11.069
Gutie U, Sobero G (2018) Overproduction of rhamnolipids in Pseudomonas aeruginosa PA14 by redirection of the carbon flux from polyhydroxyalkanoate synthesis and overexpression of the rhlAB - R operon. Biotechnol Lett 8:1561–1566. https://doi.org/10.1007/s10529-018-2610-8
Haldane JBS (1930) Enzymes longmans. Green Co
Heryani H, Putra MD (2017) Kinetic study and modeling of biosurfactant production using Bacillus sp. Electron J Biotechnol 27:49–54. https://doi.org/10.1016/j.ejbt.2017.03.005
Heyd M, Kohnert A, Tan TH et al (2008) Development and trends of biosurfactant analysis and purification using rhamnolipids as an example. Anal Bioanal Chem 391:1579–1590. https://doi.org/10.1007/s00216-007-1828-4
Hua Z, Chen Y, Du G, Chen J (2004) Effects of biosurfactants produced by Candida antarctica on the biodegradation of petroleum compounds. World J Microbiol Biotechnol 20:25–29. https://doi.org/10.1023/B:WIBI.0000013287.11561.d4
Ibrahim HMM (2016) Biodegradation of used engine oil by novel strains of Ochrobactrum anthropi HM-1 and Citrobacter freundii HM-2 isolated from oil-contaminated soil. 3 Biotech 6:1–13. https://doi.org/10.1007/s13205-016-0540-5
Ibrahim HMM (2018) Characterization of biosurfactants produced by novel strains of Ochrobactrum anthropi HM-1 and Citrobacter freundii HM-2 from used engine oil-contaminated soil. Egypt J Pet 27:21–29. https://doi.org/10.1016/j.ejpe.2016.12.005
Ibrahim S, Shukor MY, Syed MA et al (2016) Characterisation and growth kinetics studies of caffeine-degrading bacterium Leifsonia sp. strain SIU. Ann Microbiol 66:289–298. https://doi.org/10.1007/s13213-015-1108-z
Jiang J, Zu Y, Li X et al (2020) Recent progress towards industrial rhamnolipids fermentation: process optimization and foam control. Bioresour Technol 298:122394. https://doi.org/10.1016/j.biortech.2019.122394
Johnson P, Trybala A, Starov V, Pinfield VJ (2021) Effect of synthetic surfactants on the environment and the potential for substitution by biosurfactants. Adv Colloid Interface Sci. https://doi.org/10.1016/j.cis.2020.102340
Kashif A, Rehman R, Fuwad A et al (2022) Current advances in the classification, production, properties and applications of microbial biosurfactants—a critical review. Adv Colloid Interface Sci 306:102718. https://doi.org/10.1016/j.cis.2022.102718
Kumar AP, Janardhan A, Viswanath B et al (2016) Evaluation of orange peel for biosurfactant production by Bacillus licheniformis and their ability to degrade naphthalene and crude oil. 3 Biotech 6:43. https://doi.org/10.1007/s13205-015-0362-x
Lan G, Fan Q, Liu Y et al (2015) Rhamnolipid production from waste cooking oil using Pseudomonas. Biochem Eng J 101:44–54. https://doi.org/10.1016/j.bej.2015.05.001
Luong JHT (1987) Generalization of monod kinetics for analysis of growth data with substrate inhibition. Biotechnol Bioeng 29:242–248. https://doi.org/10.1002/bit.260290215
Maletić S, Dalmacija B, Rončević S et al (2009) Degradation kinetics of an aged hydrocarbon-contaminated soil. Water Air Soil Pollut 202:149–159. https://doi.org/10.1007/s11270-008-9965-8
Market Research Report (2021) Fortune business insights. https://www.fortunebusinessinsights.com/surfactants-market-102385
Medić AB, Karadžić IM (2022) Pseudomonas in environmental bioremediation of hydrocarbons and phenolic compounds- key catabolic degradation enzymes and new analytical platforms for comprehensive investigation. World J Microbiol Biotechnol 38:1–28. https://doi.org/10.1007/s11274-022-03349-7
Mishra S, Chauhan P, Gupta S et al (2017) CO2 sequestration potential of halo-tolerant bacterium Pseudomonas aeruginosa SSL-4 and its application for recovery of fatty alcohols. Process Saf Environ Prot 111:582–591. https://doi.org/10.1016/j.psep.2017.08.013
Monod J (1949) The growth of bacterial cultures. Ann Rev Microbiol 3:371–394. https://doi.org/10.1146/annurev.mi.03.100149.002103
Muloiwa M, Nyende-Byakika S, Dinka M (2020) Comparison of unstructured kinetic bacterial growth models. South African J Chem Eng 33:141–150. https://doi.org/10.1016/j.sajce.2020.07.006
Nitschke M, Costa SGVAO, Contiero J (2005) Rhamnolipid surfactants : an update on the general aspects of these remarkable biomolecules. Biotechnol Prog 21:1593–1600
Nur Asshifa MN, Zambry NS, Salwa MS, Yahya ARM (2017) The influence of agitation on oil substrate dispersion and oxygen transfer in pseudomonas aeruginosa USM-AR2 fermentation producing rhamnolipid in a stirred tank bioreactor. 3 Biotech. https://doi.org/10.1007/s13205-017-0828-0
Pasternak G, Askitosari TD, Rosenbaum MA (2020) Biosurfactants and synthetic surfactants in bioelectrochemical systems: a mini-review. Front Microbiol 11:1–9. https://doi.org/10.3389/fmicb.2020.00358
Pathania AS, Jana AK (2020) Utilization of waste frying oil for rhamnolipid production by indigenous Pseudomonas aeruginosa: Improvement through co-substrate optimization. J Environ Chem Eng 8:104304. https://doi.org/10.1016/j.jece.2020.104304
Patowary K, Saikia RR, Kalita MC, Deka S (2015) Degradation of polyaromatic hydrocarbons employing biosurfactant-producing Bacillus pumilus KS2. Ann Microbiol 65:225–234. https://doi.org/10.1007/s13213-014-0854-7
Patowary K, Patowary R, Kalita MC, Deka S (2017) Characterization of biosurfactant produced during degradation of hydrocarbons using crude oil as sole source of carbon. Front Microbiol 8:1–14. https://doi.org/10.3389/fmicb.2017.00279
Pavia DL, Lampman GM, Kriz GS, Vyvyan JA (2015) Introduction to spectroscopy. Cengage Learning, Boston
Powell EO (1967) The growth rate of microorganisms as a function of substrate concentration. Microb Physiol Contin Cult 3
Saharan BS, Sahu RK, Sharma D (2011) A review on biosurfactants : fermentation, current developments and perspectives. Genet Eng Biotechnol J 29:1–39
Salehi FM, Morina A, Neville A (2017) The effect of soot and diesel contamination on wear and friction of engine oil pump. Tribol Int 115:285–296. https://doi.org/10.1016/j.triboint.2017.05.041
Sana S, Datta S, Biswas D, Bhattacharya M (2017) Production kinetics of Rhamnolipid using fish fat: a step towards environmental hazard control of sewage. Environ Technol Innov 8:299–308. https://doi.org/10.1016/j.eti.2017.07.004
Sánchez C (2022) A review of the role of biosurfactants in the biodegradation of hydrophobic organopollutants: production, mode of action, biosynthesis and applications. World J Microbiol Biotechnol 38:1–24. https://doi.org/10.1007/s11274-022-03401-6
Santhoshkumar A, Ramanathan A (2020) Recycling of waste engine oil through pyrolysis process for the production of diesel like fuel and its uses in diesel engine. Energy 197:117240. https://doi.org/10.1016/j.energy.2020.117240
Santos DKF, Rufino RD, Luna JM et al (2016) Biosurfactants: Multifunctional biomolecules of the 21st century. Int J Mol Sci 17:1–31. https://doi.org/10.3390/ijms17030401
Saravanan P, Pakshirajan K, Saha P (2008) Growth kinetics of an indigenous mixed microbial consortium during phenol degradation in a batch reactor. Bioresour Technol 99:205–209. https://doi.org/10.1016/j.biortech.2006.11.045
Sarubbo LA, da Silva MGC, Durval IJB et al (2022) Biosurfactants: Production, properties, applications, trends, and general perspectives. Biochem Eng J 181:108377. https://doi.org/10.1016/j.bej.2022.108377
Sheikh Z, Pawar S, Rathod VK (2019) Enhancement of rhamnolipid production through ultrasound application and response surface methodology. Process Biochem 85:29–34. https://doi.org/10.1016/j.procbio.2019.06.023
Silva SNRL, Farias CBB, Rufino RD et al (2010) Glycerol as substrate for the production of biosurfactant by Pseudomonas aeruginosa UCP0992. Colloids Surf B Biointerfaces 79:174–183. https://doi.org/10.1016/j.colsurfb.2010.03.050
da SoaresSilva RCF, Almeida DG, Meira HM et al (2017) Production and characterization of a new biosurfactant from Pseudomonas cepacia grown in low-cost fermentative medium and its application in the oil industry. Biocatal Agric Biotechnol 12:206–215. https://doi.org/10.1016/j.bcab.2017.09.004
Tan YN, Li Q (2018) Microbial production of rhamnolipids using sugars as carbon sources. Microb Cell Fact. https://doi.org/10.1186/s12934-018-0938-3
Twigg MS, Baccile N, Banat IM et al (2021) Microbial biosurfactant research: time to improve the rigour in the reporting of synthesis, functional characterization and process development. Microb Biotechnol 14:147–170. https://doi.org/10.1111/1751-7915.13704
Vijayalakshmi V, Senthilkumar P, Mophin-Kani K et al (2018) Bio-degradation of Bisphenol A by Pseudomonas aeruginosa PAb1 isolated from effluent of thermal paper industry: Kinetic modeling and process optimization. J Radiat Res Appl Sci 11:56–65. https://doi.org/10.1016/j.jrras.2017.08.003
Walter V, Syldatk C, Hausmann R (2010) Screening concepts for the isolation of biosurfactant producing microorganisms. Biosurfactants. https://doi.org/10.1007/978-1-4419-5979-9_1
Zajšek K, Goršek A (2010) Modelling of batch kefir fermentation kinetics for ethanol production by mixed natural microflora. Food Bioprod Process 88:55–60. https://doi.org/10.1016/j.fbp.2009.09.002
Zhao F, Zhou J, Han S et al (2016) Medium factors on anaerobic production of rhamnolipids by Pseudomonas aeruginosa SG and a simplifying medium for in situ microbial enhanced oil recovery applications. World J Microbiol Biotechnol 32:1–11. https://doi.org/10.1007/s11274-016-2020-9
Acknowledgements
The authors express their immense gratitude towards the Chemical Engineering Department, BITS Pilani, for providing the required infrastructure, equipment, and chemicals to complete the experimental work successfully. The authors thank the Central NMR facility, BITS Pilani, for helping with the advanced characterizations.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
SG: conceived, designed, performed research, analysed data, and wrote and read the paper. SG and AJ: conceived, designed, read, and approved the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study did not require ethics approval.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gaur, S., Gupta, S. & Jain, A. Production, characterization, and kinetic modeling of biosurfactant synthesis by Pseudomonas aeruginosa gi |KP 163922|: a mechanism perspective. World J Microbiol Biotechnol 39, 178 (2023). https://doi.org/10.1007/s11274-023-03623-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-023-03623-2