Abstract
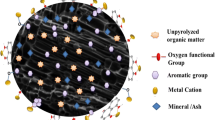
This paper reports on the effect of activating agents such as the impregnation ratio of phosphoric acid (1:1–1:5) at constant activation temperature on the performance of porous activated carbon from waste residues (maize tassel). The variation in the impregnation ratio of the produced activated carbon (AC) from 1:1 to 1:5 enabled the preparation of a high surface area (1,263 m2/g) and a large pore volume (1.592 cm3/g) of AC produced from maize tassel (MT) using a convectional chemical activating agent (phosphoric acid). Impregnation ratios (IR) of the precursors were varied between 1:1 and 1:5 in which it was found that the ratio of 1:4 was optimal based on the high surface area, while 1:5 has the optimal pore volume value for the produced activated carbon.













Similar content being viewed by others
References
Ahmadpour, A., & Do, D. D. (1996). The preparation of active carbons from coal by chemical and physical activation. Carbon, 34, 471–479.
Ahmadpour, A., & Do, D. D. (1997). The preparation of activated carbon from macadamia nutshell by chemical activation. Carbon, 35, 1723–1732.
Aksu, Z. J., & Yener, A. (2001). Comparative adsorption/biosorption study of monochlorinated phenols onto various sorbents. Waste Manag, 21, 695–702.
Bansal, R. C., & Goyal, M. (2005). Activated carbon adsorption. Boca Raton: Taylor and Francis.
Bansal, R. C., Donnet, J. B., & Stoeekli, H. F. (1988). Active carbon. New York: Marcel Dekker.
Baquero, M. C., Giraldo, L., Moreno, J. C., Sua´rez-Garcı´a, F., Martı´nez-Alons, A., & Tasco´n, J. M. D. (2003). Activated carbons by pyrolysis of coffee bean husks in presence of phosphoric acid. Journal of Analytical Applied Pyrolysis, 70(2), 779–784.
Boonamnuavitaya, V., Sae-ung, S., & Tanthapanichakoon, W. (2003). Preparation of activated carbons from coffee residue for the adsorption of formaldehyde. Separation Purification Technology, 42, 159–168.
Bota, A., Laszlo, K., Nagy, L. G., & Schlimper, H. (1997). Active carbon from apricot pits. Magyar Kemiac Folyoirat, 103(9), 470–479.
Daud, W. M. A., & Ali, W. S. W. (2004). Comparison on pore development of activated carbon produced from palm shell and coconut shell. Bioresour Technol, 93, 63–69.
Demiral, H., Demiral, L., Karabacakoglu, B., & Tumsek, F. (2011). Production of activated carbon from olive bagasse by physical activation chem. Engineering Research Design, 89, 206–213.
Diao, Y., Walawender, W. P., & Fan, L. T. (2002). Activated carbons prepared from phosphoric acid activation of grain sorghum. Bioresour Technol, 81, 45–52.
Elmorsi, T. M. (2011). Equilibrium isotherms and kinetic studies of removal of methylene blue dye by adsorption onto miswak leaves as a natural adsorbent. Journal of Environmental Protection, 2, 817–827.
G'omez-Serrano, V., Cuerda-Correa, E. M., Fern´andez-Gonz´ales, M. C., Alexandre-Franco, M. F., & Mac´ıas-Garc´ıa, A. (2005). Preparation of activated carbons from chestnut wood by phosphoric acid-chemical activation. Study of microporosity and fractal dimension. Material Letters, 59, 846–853.
Gaspard, S., Altenor, S., Dawson, E. A., Barnes, P. A., & Quensanga, A. (2007). Activated carbon from vetiver roots: gas and liquid adsorption studies. Journal Hazardous Material, 144, 73–81.
Guo, Y., & Rockstraw, D. A. (2006). Physical and chemical properties of carbons synthesized from xylan, cellulose, and Kraft lignin by H3PO4 activation. Carbon, 44, 1464–1475.
Hameed, B. H., & Rahman, A. A. (2008). Removal of phenol from aqueous solutions by adsorption onto activated carbon prepared from biomass material. Journal Hazardous Material, 160, 576–581.
Ho, Y. S., & McKay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry, 34(5), 451–465.
Ho, Y. S., & Ofomaja, A. E. (2006). Pseudo-second-order model for lead ion sorption from aqueous solutions onto palm kernel fiber. Journal of hazardous materials, 129(1–3), 137–142.
Hsu, L. Y., & Teng, H. (2000). Influence of different chemical reagents on the preparation of activated carbons from bituminous coal. Fuel Process Technology, 64, 155–166.
Hu, Z., & Srinivasan, M. P. (1999). Preparation of high-surface-area activated carbons from coconut shell. Microporous Mesoporous Material, 27, 11–18.
Hu, Z., Srinivasan, M. P., & Ni, Y. (2001). Novel activation process for preparing highly microporous and mesoporous activated carbons. Carbon, 39, 877–886.
Jagtoyen, M., & Derbyshire, F. (1998). Activated carbons from yellow poplar and white oak by H3PO4 activation. Carbon, 36, 1085–1097.
Jankowska, H., Swiatkowski, A., Choma, J. (1991). Active Carbon. Chichester, West Sussex: Ellis Horwood publisher.
Jaramillo, J., Modesto Alvarez, P., Gόmez-Serrano, V. (2010). Oxidation of activated carbon by dry and wet methods surface chemistry and textural modifications. Fuel Processing Technology, 91(11), 1768–1775.
Laine, J., Calafat, A., & Labady, M. (1989). Preparation and characterization of activated carbons from coconut shell impregnated with phosphoric acid. Carbon, 27, 191–195.
Lua, A. C., & Yang, T. (2004). Effect of activation temperature on the textural and chemical properties of potassium hydroxide activated carbon prepared from pistachio-nut shell. Journal of Colloid Interface Science, 274, 594–601.
Moreno-Castillo, C. (2004). Adsorption of organic molecules from aqueous solutions on carbon materials. Carbon, 42, 83–94.
Moreno-Castillo, C., Rivera-Utrilla, J., Lopez-Ramon, M. V., & Crresco-Marin, F. (1995). Adsorption of some substituted phenols on activated carbons from a bituminous coal. Carbon, 33, 845–851.
Mozammel, H. M., Masahiro, O., & Bhattacharya, S. C. (2002). Activated charcoal from coconut shell using ZnCl2 activation. Biomass Bioenergy, 22, 397–400.
Namane, A., Mekarzia, A., Benrachedi, K., Bensemra, N., & Hellal, A. (2005). Determination of the adsorption capacity of activated carbon made from coffee grounds by chemical activation with ZnCl2 and H3PO4. Journal of Hazardous Material, 119, 189–194.
Olorundare, O. F., Krause, R. W. M., Okonkwo, J. O., & Mamba, B. B. (2012). Potential application of activated carbon from maize tassel for the removal of heavy metals in water. Journal of Physics Chemistry of the Earth, 50–52, 104–110.
Parra, J. B., Sousa, J. C., Pis, J. J., Pajares, J. A., & Bansal, R. C. (1995). Effect of gasification on the porous characteristics of activated carbons from semi anthracite. Carbon, 33, 801–807.
Prahas, D., Kartika, Y., Indraswati, N., & Ismadji, S. (2008). Activated carbon from jackfruit peel waste by H3PO4 chemical activation: pore structure and surface chemistry characterization. Chem Eng J, 140, 32–42.
Puziy, A. M., Poddubnaya, O. I., Mart´ınez-Alonso, A., Su´arez-Garc´ıa, F., & Tasc´on, J. M. D. (2005). Surface chemistry of phosphorus-containing carbons of lignocellulosic origin. Carbon, 43, 2857–2868.
Rodriguez-Reinoso, F., & Molina-Sabio, M. (1992). Activated carbons from lignocellulosic materials by chemical and/or physical activation: an overview. Carbon, 30, 1111–1118.
Ruland, W., & Smarsly, B. (2002). X-ray scattering of non-graphitic carbon: an improved method of evaluation. Journal of Applied Crystallography, 35, 624–633.
Sekar, M., Sakthi, V., & Rengaraj, S. (2004). Kinetics and equilibrium adsorption study of lead (II) onto activated carbon prepared from coconut shell. Journal of Colloid Interface Science, 279, 307–313.
Srinivasakannan, C., & Bakar, M. Z. A. (2004). Production of activated carbon from rubber wood sawdust. Biomass Bioenergy, 27, 89–96.
Srivastava, V. C., Swamy, M. M., Mall, I. D., Prasad, B., & Mishra, I. M. (2006). Adsorptive removal of phenol by bagasse fly ash and activated carbon: equilibrium, kinetics and thermodynamics. Colloids. Surface A: Physicochemical Engineering Aspects, 272, 89–104.
Sudaryanto, Y., Hartono, S. B., Irawaty, W., Hindarso, H., & Ismadji, S. (2006). High surface area activated carbon prepared from cassava peel by chemical activation. Bioresour Technol, 97, 734–739.
Teng, H., Yeh, T. S., & Hsu, L. H. (1998). Preparation of activated carbon from bituminous coal with phosphoric acid activation. Carbon, 36, 1387–1395.
Timur, S., Kantarli, I. C., Ikizoglu, E., & Yanik, J. (2006). Preparation of activated carbons from oreganum stalks by chemical activation. Energy Fuels, 20, 2636–2641.
Vernersson, T., Bonelli, P. R., Cerrella, E. G., & Cukierman, A. L. (2001). Arundo donax cane as a precursor for activated carbons preparation by phosphoric acid activation. Bioresour Technol, 83, 95–104.
Wang, T., Tan, S., & Liang, C. (2009). Preparation and characterization of activated carbon from wood via microwave-induced ZnCl2 activation. Carbon, 47, 1880–1883.
Wang, L., Guo, Y., Zou, B., Rong, C., Ma, X., Qu, Y., Li, Y., & Wang, Z. (2011). High surface area porous carbons prepared from hydrochars by phosphoric acid activation. Bioresour Technol, 102, 1947–1950.
Zvinowanda, C. M., Okonkwo, O. J., Agyei, N. M., & Shabalala, P. N. (2008a). Preparation and characterization of biosorbents made from maize tassel. Canadian Journal Pure Applied Science, 2(3), 483–488.
Zvinowanda, C. M., Okonkwo, J. O., Mpangela, V., Phaleng, J., Shabalala, P. N., Dennis, T., Forbes, P., Agyei, N. A., & Ozoemena, K. I. (2008b). Biosorption of toxic metals: the potential use of maize tassel for the removal of Pb (II) from aqueous solutions. Fresenius Environmental Bulletin, 17(7a), 814–818.
Acknowledgments
The authors would like to acknowledge the University of Johannesburg for funding this research project.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Olorundare, O.F., Msagati, T.A.M., Krause, R.W.M. et al. Activated Carbon from Lignocellulosic Waste Residues: Effect of Activating Agent on Porosity Characteristics and Use as Adsorbents for Organic Species. Water Air Soil Pollut 225, 1876 (2014). https://doi.org/10.1007/s11270-014-1876-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-014-1876-2