Abstract
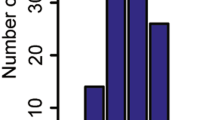
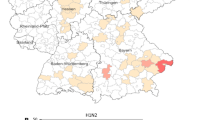
The pig is known as a “mixing vessel” for influenza A viruses. The co-circulation of multiple influenza A subtypes in pig populations can lead to novel reassortant strains. For this study, swine influenza surveillance was conducted from September 2011 to February 2014 on 46 swine farms in Thailand. In total, 78 swine influenza viruses were isolated from 2,821 nasal swabs, and 12 were selected for characterization by whole genome sequencing. Our results showed that the co-circulation of swine influenza subtypes H1N1, H3N2, and H1N2 in Thai swine farms was observable throughout the 3 years of surveillance. Furthermore, we repeatedly found reassortant viruses between endemic swine influenza viruses and pandemic H1N1 2009. This observation suggests that there is significant and rapid evolution of swine influenza viruses in swine. Thus, continuous surveillance is critical for monitoring novel reassortant influenza A viruses in Thai swine populations.




Similar content being viewed by others
References
S. Tong, X. Zhu, Y. Li, M. Shi, J. Zhang, M. Bourgeois, H. Yang, X. Chen, S. Recuenco, J. Gomez, L.M. Chen, A. Johnson, Y. Tao, C. Dreyfus, W. Yu, R. McBride, P.J. Carney, A.T. Gilbert, J. Chang, Z. Guo, C.T. Davis, J.C. Paulson, J. Stevens, C.E. Rupprecht, E.C. Holmes, I.A. Wilson, R.O. Donis, PLoS Pathog. 9, e1003657 (2013)
C.W. Olsen, Virus Res. 85, 199–210 (2002)
S. Kupradinun, P. Peanpijit, C. Bhodhikosoom, Y. Yoshioka, A. Endo, K. Nerome, Arch. Virol. 118, 9 (1991)
P. Kitikoon, D. Sreta, S.N. Na Ayudhya, M. Wongphatcharachai, J. Lapkuntod, D. Prakairungnamthip, N. Bunpapong, S. Suradhat, R. Thanawongnuwech, A. Amonsin, Virus Genes 43, 1–5 (2011)
N. Takemae, S. Parchariyanon, S. Damrongwatanapokin, Y. Uchida, R. Ruttanapumma, C. Watanabe, S. Yamaguchi, T. Saito, Influenza Other Respir. Viruses 2, 181–189 (2008)
P. Lekcharoensuk, J. Nanakorn, W. Wajjwalku, R. Webby, W. Chumsing, Vet. Microbiol. 145, 230–244 (2010)
D. Sreta, S. Tantawet, S.N. Na Ayudhya, A. Thontiravong, M. Wongphatcharachai, J. Lapkuntod, N. Bunpapong, R. Tuanudom, S. Suradhat, L. Vimolket, Y. Poovorawan, R. Thanawongnuwech, A. Amonsin, P. Kitikoon, Emerg. Infect. Dis. 16, 1587–1590 (2010)
E. Spackman, D.A. Senne, T.J. Myers, L.L. Bulaga, L.P. Garber, M.L. Perdue, K. Lohman, L.T. Daum, D.L. Suarez, J. Clin. Microbiol. 40, 3256–3260 (2002)
WHO, WHO Manual on Animal Influenza Diagnosis and Surveillance (WHO Global Influenza Programme, Geneva, 2002), pp. 15–66
E. Hoffmann, J. Stech, Y. Guan, R.G. Webster, D.R. Perez, Arch. Virol. 146, 2275–2289 (2001)
R.C. Edgar, BMC Bioinform. 5, 113 (2004)
A.J. Drummond, A. Rambaut, BMC Evol. Biol. 7, 214 (2007)
K. Tamura, J. Dudley, M. Nei, S. Kumar, Mol. Biol. Evol. 24, 1596–1599 (2007)
A.L. Vincent, S.L. Swenson, K.M. Lager, P.C. Gauger, C. Loiacono, Y. Zhang, Vet. Microbiol. 137, 51–59 (2009)
C.F. Arias, M. Escalera-Zamudio, L. Soto-Del Rio Mde, A.G. Cobian-Guemes, P. Isa, S. Lopez, Arch. Med. Res. 40, 643–654 (2009)
D. Kobasa, S. Kodihalli, M. Luo, M.R. Castrucci, I. Donatelli, Y. Suzuki, T. Suzuki, Y. Kawaoka, J. Virol. 73, 6743–6751 (1999)
G. Neumann, T. Noda, Y. Kawaoka, Nature 459, 931–939 (2009)
N. Charoenvisal, J. Keawcharoen, D. Sreta, S. Chaiyawong, N. Nonthabenjawan, S. Tantawet, S. Jittimanee, J. Arunorat, A. Amonsin, R. Thanawongnuwech, Virus Genes 47, 75–85 (2013)
Y. Hiromoto, S. Parchariyanon, N. Ketusing, P. Netrabukkana, T. Hayashi, T. Kobayashi, N. Takemae, T. Saito, Virus Res. 169, 175–181 (2012)
A. Pereda, A. Rimondi, J. Cappuccio, R. Sanguinetti, M. Angel, J. Ye, T. Sutton, M. Dibarbora, V. Olivera, M.I. Craig, M. Quiroga, M. Machuca, A. Ferrero, C. Perfumo, D.R. Perez, Influenza Other Respir. Viruses 5, 409–412 (2011)
M. Kobayashi, I. Takayama, T. Kageyama, H. Tsukagoshi, M. Saitoh, T. Ishioka, Y. Yokota, H. Kimura, M. Tashiro, K. Kozawa, Emerg. Infect. Dis. 19, 1972–1974 (2013)
A.L. Vincent, W. Ma, K.M. Lager, B.H. Janke, J.A. Richt, Adv. Virus Res. 72, 127–154 (2008)
B. Lina, M. Bouscambert, V. Enouf, D. Rousset, M. Valette, S. van der Werf, Eurosurveillance 16, 20039 (2011)
S. Lindstrom, S. Sugita, A. Endo, M. Ishida, P. Huang, S.H. Xi, K. Nerome, Arch. Virol. 141, 1349–1355 (1996)
Acknowledgments
This research was supported, in part, by the National Research University Project of the Office of Higher Education Commission (WCU005-HR57), the Ratchadapiseksomphot Endowment Fund of Chulalongkorn University (RES560530129-HR), and the National Research Council of Thailand. The authors would like to thank Chulalongkorn University for its financial support to the Center of Excellence for Emerging and Re-emerging Infectious Diseases in Animals and for a Dutsadi Phiphat scholarship to NN. We also would like to thank the Thailand Research Fund for its financial support to the TRF Senior Scholar to AA (RTA5780006).
Conflict of interest
The author(s) declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nonthabenjawan, N., Chanvatik, S., Chaiyawong, S. et al. Genetic diversity of swine influenza viruses in Thai swine farms, 2011–2014. Virus Genes 50, 221–230 (2015). https://doi.org/10.1007/s11262-014-1153-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11262-014-1153-x