Abstract
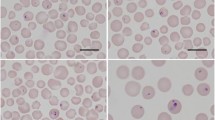
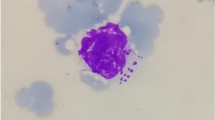
Theileria orientalis (T. orientalis) is a benign species of Theileria that is found all throughout the world, including Egypt. The purpose of this work was to determine the current status of T. orientalis infection in cattle, as well as to define T. orientalis genetic diversity using the major piroplasm surface protein (MPSP) genes. In 2020, blood samples from 500 asymptomatic cattle were collected, which live in four different governorates at northern Egypt and examined using PCR assay based on MPSP gene. Overall, 44 blood samples (8.8%, 44/500) were positive for T. orientalis, and the highest prevalence rate was observed in Kafer ElSheikh while it was relatively low in Gharbia governorate. Moreover, based on multivariable analysis, the risk of infection with T. orientalis infection was higher in mixed breed cattle and in animals of age group more than 3 years compared to other. Additionally, absence of acaricide application, infestation with ticks, and communal grazing system were identified as potential risk factors for T. orientalis infection. Phylogenetic analysis revealed that the isolates studied belonged to two MPSP genotypes, types 1 and 2. These findings reveal that the MPSP genotypes discovered in the present work displayed genetic variation that was linked to geographic area. Therefore, further research should be focused on developing an effective T. orientalis monitoring and preventive program.


Similar content being viewed by others
Data availability
All data analyzed during this study are included in this published article.
References
Aktas, M., Altay, K., and Dumanli, N., 2006. A molecular survey of bovine Theileria parasites among apparently healthy cattle and with a note on the distribution of ticks in eastern Turkey, Veterinary parasitology, 138, 179-185.
Alsubki, R.A., Albohairy, F.M., Attia, K.A., Kimiko, I., Selim, A., and Sayed-Ahmed, M.Z., 2022. Assessment of Seroprevalence and Associated Risk Factors for Anaplasmosis in Camelus dromedarius, Veterinary Sciences, 9, 57
Altangerel, K., Sivakumar, T., Inpankaew, T., Jittapalapong, S., Terkawi, M.A., Ueno, A., Xuan, X., Igarashi, I., and Yokoyama, N., 2011. Molecular prevalence of different genotypes of Theileria orientalis detected from cattle and water buffaloes in Thailand, Journal of Parasitology, 97, 1075-1079.
Amira, A.-H., Ahmed, L., Ahmed, J., Nijhof, A., and Clausen, P.-H., 2018. Epidemiological study on tropical theileriosis (Theileria annulata infection) in the Egyptian Oases with special reference to the molecular characterization of Theileria spp, Ticks and tick-borne diseases, 9, 1489-1493.
Bishop, R.P., Odongo, D., Ahmed, J., Mwamuye, M., Fry, L.M., Knowles, D.P., Nanteza, A., Lubega, G., Gwakisa, P., and Clausen, P.H., 2020. A review of recent research on Theileria parva: Implications for the infection and treatment vaccination method for control of East Coast fever, Transboundary and emerging diseases, 67, 56-67.
Brahma, J., Baishya, B.C., Phukan, A., Mahato, G., Deka, D.K., and Goswami, S., 2018. Prevalence of Theileria orientalis in crossbred cattle of Kamrup district of Assam, Int. J. Chem. Stud., 6, 1791-1794.
Eamens, G.J., Gonsalves, J.R., Jenkins, C., Collins, D., and Bailey, G., 2013. Theileria orientalis MPSP types in Australian cattle herds associated with outbreaks of clinical disease and their association with clinical pathology findings, Veterinary parasitology, 191, 209-217.
Elsify, A., Sivakumar, T., Nayel, M., Salama, A., Elkhtam, A., Rizk, M., Mosaab, O., Sultan, K., Elsayed, S., and Igarashi, I., 2015. An epidemiological survey of bovine Babesia and Theileria parasites in cattle, buffaloes, and sheep in Egypt, Parasitology international, 64, 79-85.
Fatima, M., Saeed, S., Shaikh, R.S., Ali, M., and Iqbal, F., 2015. A study on molecular detection of Theileria lestoquardi by PCR amplification in apparently healthy small ruminants from five districts of Southern Punjab, Pakistan Journal of Zoology, 47, 441.446..
Fuujisaki, K., 1992. A review of the taxonomy of Theileria sergenti/buffeli/orientalis group parasites in cattle, The Journal of Protozoology Research, 2, 87-96
Gul, N., Ayaz, S., Gul, I., Adnan, M., Shams, S., and Akbar, N., 2015. Tropical theileriosis and east coast fever in cattle: present, past and future perspective, International Journal of Current Microbiology and Applied Science, 4, 1000-1018.
Hailemariam, Z., Krücken, J., Baumann, M., Ahmed, J.S., Clausen, P.-H., and Nijhof, A.M., 2017. Molecular detection of tick-borne pathogens in cattle from Southwestern Ethiopia, PloS one, 12, e0188248.
Hall, T., Biosciences, I., and Carlsbad, C., 2011. BioEdit: an important software for molecular biology, GERF Bull Biosci, 2, 60-61.
Hassan, M.A., Liu, J., Sajid, M.S., Rashid, M., Mahmood, A., Abbas, Q., Guan, G., Yin, H., and Luo, J., 2018. Simultaneous detection of Theileria annulata and Theileria orientalis infections using recombinase polymerase amplification, Ticks and tick-borne diseases, 9, 1002-1005.
Jenkins, C., and Bogema, D.R., 2016. Factors associated with seroconversion to the major piroplasm surface protein of the bovine haemoparasite Theileria orientalis, Parasites & vectors, 9, 1-9.
Jeong, W., Yoon, S., An, D., Cho, S.-H., Lee, K.-K., and Kim, J.-Y., 2010. A molecular phylogeny of the benign Theileria parasites based on major piroplasm surface protein (MPSP) gene sequences, Parasitology, 137, 241-249.
Jirapattharasate, C., Moumouni, P.F.A., Cao, S., Iguchi, A., Liu, M., Wang, G., Zhou, M., Vudriko, P., Changbunjong, T., and Sungpradit, S., 2016. Molecular epidemiology of bovine Babesia spp. and Theileria orientalis parasites in beef cattle from northern and northeastern Thailand, Parasitology international, 65, 62-69.
Jongejan, F., and Uilenberg, G., 2004. The global importance of ticks, Parasitology, 129, S3-S14.
Kakuda, T., Shiki, M., Kubota, S., Sugimoto, C., Brown, W.C., Kosum, C., Nopporn, S., and Onuma, M., 1998. Phylogeny of benign Theileria species from cattle in Thailand, China and the USA based on the major piroplasm surface protein and small subunit ribosomal RNA genes, International journal for parasitology, 28, 1261-1267.
Kerario, I.I., Simuunza, M.C., Chenyambuga, S.W., Koski, M., Hwang, S.-G., and Muleya, W., 2017. Prevalence and risk factors associated with Theileria parva infection in cattle in three regions of Tanzania, Tropical animal health and production, 49, 1613-1621.
Khukhuu, A., Lan, D.T.B., Long, P.T., Ueno, A., Li, Y., Luo, Y., de MACEDO, A.C.C., Matsumoto, K., Inokuma, H., and Kawazu, S.-I., 2010. Molecular epidemiological survey of Theileria orientalis in Thua Thien Hue province, Vietnam, Journal of Veterinary Medical Science, 1012170417–1012170417.
Kumar, S., Stecher, G., and Tamura, K., 2016. MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets, Molecular biology and evolution, 33, 1870-1874.
l'Hostis, M., and Seegers, H., 2002. Tick-borne parasitic diseases in cattle: current knowledge and prospective risk analysis related to the ongoing evolution in French cattle farming systems, Veterinary Research, 33, 599-611.
Laisser, E., Chenyambuga, S., Karimuribo, E., Msalya, G., Kipanyula, M., Mwilawa, A., Mdegela, R., Kusiluka, L., and Gwakisa, P., 2016. Tick burden and acquisition of immunity to Theileria parva by Tarime cattle in comparison to Sukuma cattle under different tick control regimes in the Lake Zone of Tanzania, Journal of Veterinary Medicine and Animal Health, 8, 21-28.
Lawrence, K., Gedye, K., McFadden, A., Pulford, D., and Pomroy, W., 2016. An observational study of the vertical transmission of Theileria orientalis (Ikeda) in a New Zealand pastoral dairy herd, Veterinary parasitology, 218, 59-65.
Liu, A., Guan, G., Liu, Z., Liu, J., Leblanc, N., Li, Y., Gao, J., Ma, M., Niu, Q., and Ren, Q., 2010. Detecting and differentiating Theileria sergenti and Theileria sinensis in cattle and yaks by PCR based on major piroplasm surface protein (MPSP), Experimental Parasitology, 126, 476-481.
McFadden, A., Rawdon, T., Meyer, J., Makin, J., Morley, C., Clough, R., Tham, K., Müllner, P., and Geysen, D., 2011. An outbreak of haemolytic anaemia associated with infection of Theileria orientalis in naive cattle, New Zealand veterinary journal, 59, 79-85
Mekata, H., Minamino, T., Mikurino, Y., Yamamoto, M., Yoshida, A., Nonaka, N., and Horii, Y., 2018. Evaluation of the natural vertical transmission of Theileria orientalis, Veterinary parasitology, 263, 1-4.
Mosabah, A.A., and Morsy, T.A., 2012. Tick paralysis: First zoonosis record in Egypt, Journal of the Egyptian Society of Parasitology, 240, 1-8.
Muhanguzi, D., Picozzi, K., Hatendorf, J., Thrusfield, M., Welburn, S.C., Kabasa, J.D., and Waiswa, C., 2014. Prevalence and spatial distribution of Theileria parva in cattle under crop-livestock farming systems in Tororo District, Eastern Uganda, Parasites & vectors, 7, 1-8.
Mukhebi, A., Perry, B.D., and Kruska, R., 1992. Estimated economics of theileriosis control in Africa, Preventive veterinary medicine, 12, 73-85
Nayel, M., El-Dakhly, K.M., Aboulaila, M., Elsify, A., Hassan, H., Ibrahim, E., Salama, A., and Yanai, T., 2012. The use of different diagnostic tools for Babesia and Theileria parasites in cattle in Menofia, Egypt, Parasitology research, 111, 1019-1024.
Nourollahi-Fard, S.R., Khalili, M., and Ghalekhani, N., 2015. Detection of Theileria annulata in blood samples of native cattle by PCR and smear method in Southeast of Iran, Journal of Parasitic Diseases, 39, 249-252.
Ola-Fadunsin, S.D., Sharma, R.S.K., Abdullah, D.A., Gimba, F.I., Jesse, F.F.A., and Sani, R.A., 2020. Molecular detection, prevalence and risk factors of Theileria orientalis infection among cattle in Peninsular Malaysia, Preventive veterinary medicine, 180, 105027.
Orkun, Ö., Deniz, A., and Güven, E., 2012. Survey of Theileria annulata and Theileria buffeli/orientalis Complex in Cattle in the Kirsehir Region Using Multiplex-PCR, Türkiye Parazitolojii Dergisi, 36, 9-11.
Ota, N., Mizuno, D., Kuboki, N., Igarashi, I., Nakamura, Y., Yamashina, H., Hanzaike, T., Fujii, K., Onoe, S., and Hata, H., 2009. Epidemiological survey of Theileria orientalis infection in grazing cattle in the eastern part of Hokkaido, Japan, Journal of Veterinary Medical Science, 71, 937-944.
Park, J., Han, Y.-J., Han, D.-G., Chae, J.-B., Chae, J.-S., Yu, D.-H., Lee, Y.-S., Park, B.-K., Kim, H.-C., and Choi, K.-S., 2017. Genetic characterization of Theileria orientalis from cattle in the Republic of Korea, Parasitology research, 116, 449-454.
Perera, P.K., Gasser, R.B., Firestone, S.M., Anderson, G.A., Malmo, J., Davis, G., Beggs, D.S., and Jabbar, A., 2014. Oriental theileriosis in dairy cows causes a significant milk production loss, Parasites & vectors, 7, 1-8
Pourhoseingholi, M.A., Vahedi, M., and Rahimzadeh, M., 2013. Sample size calculation in medical studies, Gastroenterology and Hepatology from bed to bench, 6, 14.
Rizk, M.A., Salama, A., El-Sayed, S.A.-E.-S., Elsify, A., El-Ashkar, M., Ibrahim, H., Youssef, M., and El-Khodery, S., 2017. Animal level risk factors associated with Babesia and Theileria infections in cattle in Egypt, Acta parasitologica, 62, 796-804.
Said, M.B., Attia, K.A., Alsubki, R.A., Mohamed, A.A., Kimiko, I., and Selim, A., 2022. Molecular epidemiological survey, genetic characterization and phylogenetic analysis of Anaplasma ovis infecting sheep in Northern Egypt, Acta Tropica, 106370.
Selim, A., Abdelhady, A., and Alahadeb, J., 2020a. Prevalence and first molecular characterization of Ehrlichia canis in Egyptian dogs, Pak Vet J, 41–117–121.
Selim, A., Abdelrahman, A., Thiéry, R., and Sidi-Boumedine, K., 2019a. Molecular typing of Coxiella burnetii from sheep in Egypt, Comparative immunology, microbiology and infectious diseases, 67, 101353
Selim, A., Alanazi, A.D., Sazmand, A., and Otranto, D., 2021a. Seroprevalence and associated risk factors for vector-borne pathogens in dogs from Egypt, Parasites & vectors, 14, 1-11.
Selim, A., Almohammed, H., Abdelhady, A., Alouffi, A., and Alshammari, F.A., 2021b. Molecular detection and risk factors for Anaplasma platys infection in dogs from Egypt, Parasites & vectors, 14, 1-6.
Selim, A., Attia, K., Ramadan, E., Hafez, Y.M., and Salman, A., 2019b. Seroprevalence and molecular characterization of Brucella species in naturally infected cattle and sheep, Preventive veterinary medicine, 171, 104756.
Selim, A., Manaa, E., Abdelhady, A., Said, M.B., and Sazmand, A., 2021c. Serological and molecular surveys of Anaplasma spp. in Egyptian cattle reveal high A. marginale infection prevalence, Iranian Journal of Veterinary Research, 22, 288.
Selim, A., and Radwan, A., 2020. Seroprevalence and molecular characterization of West Nile Virus in Egypt, Comparative immunology, microbiology and infectious diseases, 71, 101473.
Selim, A.M., Senapati, S.K., Das, M., Mishra, C., Patra, R.C., and Panda, S.K., 2020b. Molecular, epidemiological and haematological evaluation in Theileria orientalis infected cattle from an endemic region in India, Animal biotechnology, 1–8.
Simuunza, M., Weir, W., Courcier, E., Tait, A., and Shiels, B., 2011. Epidemiological analysis of tick-borne diseases in Zambia, Veterinary parasitology, 175, 331-342.
Sivakumar, T., Hayashida, K., Sugimoto, C., and Yokoyama, N., 2014. Evolution and genetic diversity of Theileria, Infection, Genetics and Evolution, 27, 250-263.
Sivakumar, T., Yoshinari, T., Igarashi, I., Kothalawala, H., Abeyratne, S.A.E., Vimalakumar, S.C., Meewewa, A.S., Kuleswarakumar, K., Chandrasiri, A.D.N., and Yokoyama, N., 2013. Genetic diversity within Theileria orientalis parasites detected in Sri Lankan cattle, Ticks and tick-borne diseases, 4, 235-241.
Tayebwa, D.S., Vudriko, P., Tuvshintulga, B., Guswanto, A., Nugraha, A.B., Gantuya, S., Batiha, G.E.-S., Musinguzi, S.P., Komugisha, M., and Bbira, J.S., 2018. Molecular epidemiology of Babesia species, Theileria parva, and Anaplasma marginale infecting cattle and the tick control malpractices in Central and Eastern Uganda, Ticks and tick-borne diseases, 9, 1475-1483.
Taylor, M., 2016. Coop RL. Wall, RL, Veterinary parasitology 4th ed West Sussex (UK): John Wiley & Sons, 7–200.
Thompson, J.D., Higgins, D.G., and Gibson, T.J., 1994. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice, Nucleic acids research, 22, 4673-4680.
Thrusfield, M., 2018. Veterinary epidemiology, (John Wiley & Sons).
Tuli, A., Singla, L.D., Sharma, A., Bal, M.S., Filia, G., and Kaur, P., 2015. Molecular epidemiology, risk factors and hematochemical alterations induced by Theileria annulata in bovines of Punjab (India), Acta parasitologica, 60, 378-390.
Uilenberg, G., 1995. International collaborative research: significance of tick-borne hemoparasitic diseases to world animal health, Veterinary parasitology, 57, 19-41.
Ullah, R., Shams, S., Khan, M.A., Ayaz, S., Akbar, N.u., Din, Q.u., Khan, A., Leon, R., and Zeb, J., 2021. Epidemiology and molecular characterization of Theileria annulata in cattle from central Khyber Pakhtunkhwa, Pakistan, PloS one, 16, e0249417.
Watts, J., Playford, M., and Hickey, K., 2016. Theileria orientalis: a review, New Zealand veterinary journal, 64, 3-9.
Yokoyama, N., Sivakumar, T., Ota, N., Igarashi, I., Nakamura, Y., Yamashina, H., Matsui, S., Fukumoto, N., Hata, H., and Kondo, S., 2012. Genetic diversity of Theileria orientalis in tick vectors detected in Hokkaido and Okinawa, Japan, Infection, Genetics and Evolution, 12, 1669-1675.
Yokoyama, N., Ueno, A., Mizuno, D., Kuboki, N., Khukhuu, A., Igarashi, I., Miyahara, T., Shiraishi, T., Kudo, R., and Oshiro, M., 2010. Genotypic diversity of Theileria orientalis detected from cattle grazing in Kumamoto and Okinawa prefectures of Japan, Journal of Veterinary Medical Science, 1010180362–1010180362.
Zakimi, S., Kim, J.-Y., Oshiro, M., Hayashida, K., Fujisaki, K., and Sugimoto, C., 2006. Genetic diversity of benign Theileria parasites of cattle in the Okinawa Prefecture, Journal of Veterinary Medical Science, 68, 1335-1338.
Zhou, M., Cao, S., Sevinc, F., Sevinc, M., Ceylan, O., Moumouni, P.F.A., Jirapattharasate, C., Liu, M., Wang, G., and Iguchi, A., 2016. Molecular detection and genetic identification of Babesia bigemina, Theileria annulata, Theileria orientalis and Anaplasma marginale in Turkey, Ticks and tick-borne diseases, 7, 126-134.
Zhou, Z., Li, K., Sun, Y., Shi, J., Li, H., Chen, Y., Yang, H., Li, X., Wu, B., and Li, X., 2019. Molecular epidemiology and risk factors of Anaplasma spp., Babesia spp. and Theileria spp. infection in cattle in Chongqing, China, PloS one, 14, e0215585.
Acknowledgements
The authors extend their appreciation to Princess Nourah Bint Abdulrahman University Researchers Supporting Project Number (PNURSP2022R20), Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia.
Funding
This research was funded by Princess Nourah Bint Abdulrahman University Researchers Supporting Project Number (PNURSP2022R20), Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Conceptualization, A.S., S.S. M.A., F.A., and K.A.; methodology, A.S. and K.A.; formal analysis, A.S.; investigation, A.S., S.S., and K.A.; resources, A.S., S.S. M.A., F.A., and K.A.; data curation, A.S. and S.S.; writing—original draft preparation A.S., S.S. M.A. F.A., and K.A.; writing—review and editing, A.S.. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study was carried out in accordance with the Declaration of Benha University and was approved by the Ethics Committee of Benha University’s Faculty of Veterinary Medicine (BUFVTM).
Consent to participate
All authors participated in this manuscript.
Consent for publication
All authors have consented to the submission of the manuscript in this journal.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Selim, A., Attia, K., AlKahtani, M.D.F. et al. Molecular epidemiology and genetic characterization of Theileria orientalis in cattle. Trop Anim Health Prod 54, 178 (2022). https://doi.org/10.1007/s11250-022-03176-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-022-03176-w