Abstract
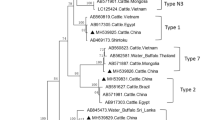
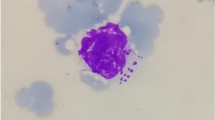
Theileria orientalis is known to be a group of benign cattle parasites with a cosmopolitan distribution, and has been classified into 11 genotypes through MPSP gene phylogenetic analysis. In China, T. orientalis is the most prevalent Theileria species, with several genotypes, but few fatal cases have been reported. In June 2020, dairy cattle in Zhangjiakou, Hebei Province, showed clinical symptoms of piroplasmosis, causing many animals to die. Blood smears and PCR detection results confirmed T. orientalis infection with a 66.7% positive rate of collected blood samples. The MPSP sequences analysis revealed parasite genotypes 1 (Chitose) and 2 (Ikeda). Aiming to isolate the pathogens, experimental animal was infected with T. orientalis via inoculation of the positive blood samples. The results has shown that only T. orientalis genotype 2 (Ikeda) was obtained that has confirmed by MPSP and 18S rRNA sequences analysis, indicating that the Ikeda type was predominant and responsible for the disease. Although many T. orientalis genotypes are present in China, the possibility of T. orientalis genotypes 1 and 2 infections in confined dairy cattle should be considered to avoid additional economic losses.



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
Code availability
Not applicable.
References
Chaisi ME, Collins NE, Oosthuizen MC (2014) Phylogeny of Theileria buffeli genotypes identified in the South African buffalo (Syncerus caffer) population. Vet Parasitol 204:87–95. https://doi.org/10.1016/j.vetpar.2014.06.001
Chansiri K, Kawazu S, Kamio T et al (1999) Molecular phylogenetic studies on Theileria parasites based on small subunit ribosomal RNA gene sequences. Vet Parasitol 83:99–105. https://doi.org/10.1016/S0304-4017(99)00052-7
Eamens GJ, Gonsalves JR, Jenkins C et al (2013) Theileria orientalis MPSP types in Australian cattle herds associated with outbreaks of clinical disease and their association with clinical pathology findings. Vet Parasitol 191:209–217. https://doi.org/10.1016/j.vetpar.2012.09.007
Fernández de Marco M, Brugman VA, Hernández-Triana LM et al (2016) Detection of Theileria orientalis in mosquito blood meals in the United Kingdom. Vet Parasitol 229:31–36. https://doi.org/10.1016/j.vetpar.2016.09.012
Forshaw D, Alex SM, Palmer DG et al (2020) Theileria orientalis Ikeda genotype infection associated with anaemia, abortion and death in beef cattle in Western Australia. Aust Vet J 98:290–297. https://doi.org/10.1111/avj.12937
Gebrekidan H, Gasser RB, Baneth G et al (2016) Molecular characterization of Theileria orientalis from cattle in Ethiopia. Ticks Tick Borne Dis. https://doi.org/10.1016/j.ttbdis.2016.03.005
George N, Bhandari V, Reddy DP, Sharma P (2015) Emergence of new genotype and diversity of Theileria orientalis parasites from bovines in India. Infection, Genet Evol 36:27–34. https://doi.org/10.1016/j.meegid.2015.08.033
Gubbels MJ, Hong Y, van der Weide M et al (2000) Molecular characterisation of the Theileria buffeli/orientalis group. Int J Parasitol 30:943–952
Gubbels M-J, Yin H, Bai Q et al (2002) The phylogenetic position of the Theileria buffeli group in relation to other Theileria species. Parasitol Res 88:S28–S32
He L, Feng H-H, Zhang W-J et al (2012) Occurrence of Theileria and Babesia species in water buffalo (Bubalus babalis, Linnaeus, 1758) in the Hubei province, South China. Vet Parasitol 186:490–496. https://doi.org/10.1016/j.vetpar.2011.11.021
Jeong W, Yoon SH, An DJ et al (2010) A molecular phylogeny of the benign Theileria parasites based on major piroplasm surface protein (MPSP) gene sequences. Parasitology 137:241–249. https://doi.org/10.1017/S0031182009991090
Junlong L, Li Y, Liu A et al (2015) Development of a multiplex PCR assay for detection and discrimination of Theileria annulata and Theileria sergenti in cattle. Parasitol Res. https://doi.org/10.1007/s00436-015-4478-z
Kakati P, Sarmah PC, Ray D et al (2015) Emergence of oriental theileriosis in cattle and its transmission through Rhipicephalus (Boophilus) microplus in Assam, India. Vet World 8:1099–1104. https://doi.org/10.14202/vetworld.2015.1099-1104
Kakuda T, Shiki M, Kubota S et al (1998) Phylogeny of benign Theileria species from cattle in Thailand, China and the U.S.A. based on the major piroplasm surface protein and small subunit ribosomal RNA genes. Int J Parasitol 28:1261–1267. https://doi.org/10.1016/S0020-7519(98)00113-1
Kamau J, de Vos AJ, Playford M et al (2011) Emergence of new types of Theileria orientalis in Australian cattle and possible cause of theileriosis outbreaks. Parasit Vectors 4:22. https://doi.org/10.1186/1756-3305-4-22
Kawabuchi T, Tsuji M, Sado A et al (2005) Babesia microti-like parasites detected in feral raccoons (Procyon lotor) captured in Hokkaido, Japan. J Vet Med Sci 67:825–827
Kawazu S, Kamio T, Kakuda T et al (1999) Phylogenetic relationships of the benign Theileria species in cattle and Asian buffalo based on the major piroplasm surface protein (p33/34) gene sequences. Int J Parasitol 29:613–618. https://doi.org/10.1016/S0020-7519(99)00008-9
Kim SJ, Tsuji M, Kubota S et al (1998) Sequence analysis of the major piroplasm surface protein gene of benign bovine Theileria parasites in east Asia. Int J Parasitol 28:1219–1227
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Lawrence KE, Gedye K, McFadden AMJ et al (2016) An observational study of the vertical transmission of Theileria orientalis (Ikeda) in a New Zealand pastoral dairy herd. Vet Parasitol 218:59–65. https://doi.org/10.1016/j.vetpar.2016.01.003
Lawrence KE, Gedye K, Pomroy WE (2019) A longitudinal study of the effect of Theileria orientalis Ikeda type infection on three New Zealand dairy farms naturally infected at pasture. Vet Parasitol 276:108977. https://doi.org/10.1016/j.vetpar.2019.108977
Li L-H, Wang J-Z, Zhu D et al (2020) Detection of novel piroplasmid species and Babesia microti and Theileria orientalis genotypes in hard ticks from Tengchong County, Southwest China. Parasitol Res 119:1259–1269. https://doi.org/10.1007/s00436-020-06622-6
Liu AH, Guan GQ, Liu JL et al (2011) Polymorphism analysis of Chinese Theileria sergenti using allele-specific polymerase chain reaction of the major piroplasm surface protein gene. J Parasitol 97:116–121. https://doi.org/10.1645/GE-2444.1
Luo J, Lu W (1997) Cattle theileriosis in China. Trop Anim Health Prod 29:4S–7S
McFadden AMJ, Rawdon TG, Meyer J et al (2011) An outbreak of haemolytic anaemia associated with infection of Theileria orientalis in naive cattle. N Z Vet J 59:79–85. https://doi.org/10.1080/00480169.2011.552857
Mekata H, Minamino T, Mikurino Y et al (2018) Evaluation of the natural vertical transmission of Theileria orientalis. Vet Parasitol 263:1–4. https://doi.org/10.1016/j.vetpar.2018.09.017
Oakes VJ, Yabsley MJ, Schwartz D et al (2019) Theileria orientalis Ikeda Genotype in Cattle, Virginia, USA. Emerg Infect Dis 25:1653–1659. https://doi.org/10.3201/eid2509.190088
Perera PK, Gasser RB, Anderson GA et al (2013) Epidemiological survey following oriental theileriosis outbreaks in Victoria, Australia, on selected cattle farms. Vet Parasitol 197:509–521. https://doi.org/10.1016/j.vetpar.2013.06.023
Perera PK, Gasser RB, Jabbar A (2015) Assessment of sequence variability in a p23 gene region within and among three genotypes of the Theileria orientalis complex from south-eastern Australia. Ticks Tick Borne Dis 6:123–128. https://doi.org/10.1016/j.ttbdis.2014.10.006
Proctor A, Ball M, Freeman P et al (2016) Prevalence of Theileria orientalis types in beef cattle herds on the North Coast of New South Wales. Aust Vet J 94:117–120. https://doi.org/10.1111/avj.12415
Pulford DJ, McFadden A, Hamilton JS, Donald J (2016) Investigation of the index case herd and identification of the genotypes of Theileria orientalis associated with outbreaks of bovine anaemia in New Zealand in 2012. N Z Vet J 64:21–28. https://doi.org/10.1080/00480169.2015.1090355
Qin G, Li Y, Liu J et al (2016) Molecular detection and characterization of Theileria infection in cattle and yaks from Tibet Plateau Region, China. Parasitol Res 115:2647–2652. https://doi.org/10.1007/s00436-016-5011-8
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sivakumar T, Hayashida K, Sugimoto C, Yokoyama N (2014) Evolution and genetic diversity of Theileria. Infection Genet Evol 27:250–263. https://doi.org/10.1016/j.meegid.2014.07.013
Tamura K, Stecher G, Peterson D et al (2013) MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/mst197
Tanaka M, Onoe S, Matsuba T et al (1993) Detection of Theileria sergenti infection in cattle by polymerase chain reaction amplification of parasite-specific DNA. J Clin Microbiol 31:2565–2569
Uilenberg G (2011) Theileria sergenti. Vet Parasitol 175:386. https://doi.org/10.1016/j.vetpar.2010.09.029
Uilenberg G, Perié NM, Spanjer AA, Franssen FF (1985) Theileria orientalis, a cosmopolitan blood parasite of cattle: demonstration of the schizont stage. Res Vet Sci 38:352–360
Vinodkumar K, Shyma V, Justin DK et al (2016) Fatal Theileria orientalis N2 genotype infection among Asian water buffaloes (Bubalus bubalis) in a commercial dairy farm in Kerala, India. Parasitology 143:69–74. https://doi.org/10.1017/S0031182015001468
Wang J, Yang J, Liu J et al (2018a) Molecular detection and genetic diversity of Theileria orientalis in cattle in China. Parasitol Res 117:3689–3694. https://doi.org/10.1007/s00436-018-6023-3
Wang J, Zhang J, Zhu Z et al (2018b) Molecular phylogenetic studies on clinical bovine piroplasmosis caused by benign Theileria in Shaanxi Province, China. J Vet Sci 19:846–849. https://doi.org/10.4142/jvs.2018.19.6.846
Watts JG, Playford MC, Hickey KL (2015) Theileria orientalis: a review. N Z Vet J 1–7. https://doi.org/10.1080/00480169.2015.1064792
Zakimi S, Kim J-Y, Oshiro M et al (2006) Genetic diversity of benign Theileria parasites of cattle in the Okinawa Prefecture. J Vet Med Sci 68:1335–1338
Funding
This study was financially supported by National Natural Science Foundation of China (31972706), ASTIP, CARS-37, and Jiangsu Co-innovation Center for Prevention and Control of Important Animal Infectious Diseases and Zoonoses, State Key Laboratory of Veterinary Etiological Biology Project.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Junlong Liu, Zhi Li, Aihong Liu, and Jinming Wang. The first draft of the manuscript was written by Junlong Liu. Guiquan Guan, Jianxun Luo and Hong Yin corrected the manuscript.
Corresponding author
Ethics declarations
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Abdul Jabbar
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, J., Li, Z., Liu, A. et al. Identification and isolation of pathogenic Theileria orientalis Ikeda genotype from confined dairy cattle, in Hebei, China. Parasitol Res 121, 395–402 (2022). https://doi.org/10.1007/s00436-021-07401-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-021-07401-7