Abstract
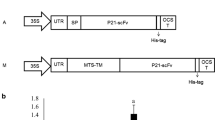
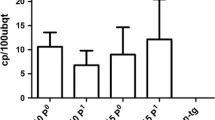
Citrus tristeza virus (CTV) causes one of the most destructive viral diseases of citrus worldwide. Generation of resistant citrus genotypes through genetic engineering could be a good alternative to control CTV. To study whether production of single-chain variable fragment (scFv) antibodies in citrus could interfere and immunomodulate CTV infection, transgenic Mexican lime plants expressing two different scFv constructs, separately and simultaneously, were generated. These constructs derived from the well-referenced monoclonal antibodies 3DF1 and 3CA5, specific against CTV p25 major coat protein, whose mixture is able to detect all CTV isolates characterized so far. ScFv accumulation levels were low and could be readily detected just in four transgenic lines. Twelve homogeneous and vigorous lines were propagated and CTV-challenged by graft inoculation with an aggressive CTV strain. A clear protective effect was observed in most transgenic lines, which showed resistance in up to 40–60% of propagations. Besides, both a delay in symptom appearance and attenuation of symptom intensity were observed in infected transgenic plants compared with control plants. This effect was more evident in lines carrying the 3DF1scFv transgene, being probably related to the biological functions of the epitope recognized by this antibody. This is the first report describing successful protection against a pathogen in woody transgenic plants by ectopic expression of scFv recombinant antibodies.





Similar content being viewed by others
References
Bajrovic K, Erdag B, Atalay EO, Cirakoclu B (2001) Full resistance to tobacco mosaic virus infection conferred by the transgenic expression of a recombinant antibody in tobacco. Biotechnol Biotechnol Equip 15:21–27
Ballester-Olmos JF, Pina JA, Carbonell EA, Moreno P, Hermoso de Mendoza A, Cambra M, Navarro L (1993) Biological diversity of Citrus tristeza virus (CTV) isolates in Spain. Plant Pathol 42:219–229
Bar-Joseph M, Marcus R, Lee RF (1989) The continuous challenge of Citrus tristeza virus control. Annu Rev Phytopathol 27:291–316
Bertolini E, Moreno A, Capote N, Olmos A, de Luis A, Vidal E, Pérez-Panadés J, Cambra M (2008) Quantitative detection of Citrus tristeza virus in plant tissues and single aphids by real-time RT–PCR. Eur J Plant Pathol 120:177–188
Bevan MW, Flavell RB, Chilton M-D (1983) A chimaeric antibiotic resistance gene as a selectable marker for plant cell transformation. Nature 304:184–187
Boonrod KJ, Galetzka D, Nagy PD, Conrad U, Krczal G (2004) Single-chain antibodies against a plant viral RNA-dependent RNA polymerase confer virus resistance. Nature Biotechnol 22:856–862
Bouaziz D, Ayadi M, Bidani A, Rouis S, Nouri-Ellouz O, Jellouli R, Drira N, Gargouri-Bouzid R (2009) A stable cytosolic expression of VH antibody fragment directed against PVY NIa protein in transgenic potato plant confers partial protection against the virus. Plant Sci 176:489–496
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brederode FT, Koperzwarthoff EC, Bol JF (1980) Complete nucleotide-sequence of Alfalfa mosaic virus RNA-4. Nucleic Acids Res 8:2213–2223
Cambra M, Garnsey SM, Permar TA, Henderson CT, Gumph D, Vela C (1990) Detection of Citrus tristeza virus (CTV) with a mixture of monoclonal antibodies. Phytopathology 80:S1034
Cambra M, Asensio M, Gorris MT, Pérez E, Camarasa E, García JA, Moya JJ, López-Abella D, Vela C, Sanz A (1994) Detection of Plum pox potyvirus using monoclonal antibodies to structural and non-structural proteins. EPPO Bull 24:569–577
Chiu WL, Niwa Y, Zeng W, Hirano T, Kobayashi H, Sheen J (1996) Engineered GFP as a vital reporter in plants. Current Biol 6:325–330
Costa AS, Müller GW (1980) Tristeza control by cross protection: a US-Brazil cooperative success. Plant Dis 64:538–541
De Jaeger G, Buys E, Eeckhout D, De Wilde C, Jacobs A, Kapila J, Angenon G, Van Montagu M, Gerats T, Depicker A (1999) High level accumulation of single-chain variable fragments in the cytosol of transgenic Petunia hybrida. Eur J Biochem 259:426–434
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: version II. Plant Mol Biol Rep 4:19–21
Domínguez A, Guerri J, Cambra M, Navarro L, Moreno P, Peña L (2000) Efficient production of transgenic citrus plants expressing the coat protein gene of citrus tristeza virus. Plant Cell Rep 19:427–433
Domínguez A, Fagoaga C, Navarro L, Moreno P, Peña L (2002a) Constitutive expression of untranslatable versions of the p25 coat protein gene in Mexican lime (Citrus aurantifolia (Christm.) Swing.) transgenic plants does not confer resistance to Citrus tristeza virus (CTV). In: Durán-Vila N, Milne RG, da Graça JV (eds) Proceedings of the XV conference of the international organization of citrus virologists, University of California, Riverside, USA, pp 341–344. http://www.ivia.es/iocv/
Domínguez A, de Mendoza AH, Guerri J, Cambra M, Navarro L, Moreno P, Peña L (2002b) Pathogen-derived resistance to Citrus tristeza virus (CTV) in transgenic Mexican lime (Citrus aurantifolia (Christ.) Swing.) plants expressing its p25 coat protein gene. Mol Breed 10:1–10
EPPO (2004) Citrus tristeza closterovirus. Diagnostic protocols for regulated pests. European and Mediterranean Plant Protection Organization. Bulletin OEPP/EPPO bulletin 34: 239–246. http://www.eppo.org/QUARANTINE/virus/Citrus_tristeza/pm7-31(1)%20CTV000%20web.pdf
Esteban O, García JA, Gorris MT, Domínguez E, Cambra M (2003) Generation and characterisation of functional recombinant antibody fragments against RNA replicase NIb from Plum pox virus. Biochem Biophys Res Commun 301:167–175
Fagoaga C, López C, de Mendoza AH, Moreno P, Navarro L, Flores R, Peña L (2006) Post-transcriptional gene silencing of the p23 silencing suppressor of Citrus tristeza virus confers resistance to the virus in transgenic Mexican lime. Plant Mol Biol 60:153–165
Fecker LE, Kaufmann A, Commandeur U, Commandeur J, Köenig R, Burgermeister W (1996) Expression of single-chain antibody fragments (scFv) specific for Beet necrotic yellow vein virus coat protein or 25 kDa protein in Escherichia coli and Nicotiana benthamiana. Plant Mol Biol 32:979–986
Fecker LF, Köenig R, Obermeier C (1997) Nicotiana benthamiana plants expressing beet necrotic yellow vein virus (BNYVV) coat protein-specific scFv are partially protected against the establishment of the virus in the early stages of infection and its pathogenic effects in the late stages of infection. Arch Virol 142:1857–1863
Fischer R, Emans N, Schillberg S (2001) Achieving plant disease resistance by antibody expression. Can J Plant Pathol 23:236–245
Folimonov A, Folimonova S, Bar-Joseph M, Dawson WO (2007) A stable RNA-virus based vector for citrus trees. Virology 368:205–216
Galeffi P, Giunta G, Guida S, Cantale C (2002) Engineering of a single variable fragment antibody specific for the Citrus tristeza virus and its expression in Escherichia coli and Nicotiana tabacum. Eur J Plant Pathol 108:479–483
Gargouri-Bouzid R, Jaoua L, Rouis S, Saidi MN, Bouaziz D, Ellouz R (2006) PVY-resistant transgenic potato plants expressing an anti-NIa protein scFv antibody. Mol Biotechnol 33:133–140
Garnsey SM, Cambra M (1991) Enzyme-linked immunosorbent assay (ELISA) for citrus pathogens. In: Roistacher CN (ed) Graft-transmisible diseases of citrus: handbook for detection and diagnosis. Food and Agriculture Organization of the United Nations-FAO, Rome, pp 193–216
Ghorbel R, Juárez J, Navarro L, Peña L (1999) Green fluorescent protein as a screenable marker to increase the efficiency of generating transgenic woody fruit plants. Theor App Genet 99:350–358
Guilley H, Dudley RK, Jonard G, Balazs E, Richards KE (1982) Transcription of Cauliflower mosaic virus DNA - detection of promoter sequences, and characterization of transcripts. Cell 30:763–773
Hilf ME, Karasev AV, Pappu HR, Gumpf DJ, Niblett CL, Garnsey SM (1995) Characterization of citrus tristeza virus subgenomic RNAs in infected tissue. Virology 208:576–582
Karasev AV, Boyko VP, Gowda S, Nikolaeva OV, Hilf ME, Koonin EV, Niblett CL, Cline K, Gumpf DJ, Lee RF, Garnsey SM, Lewandowski DJ, Dawson WO (1995) Complete sequence of the Citrus tristeza virus-RNA genome. Virology 208:511–520
Kerschbaumer RJ, Himmler G (1999) Dedicated expression vectors for the production of diagnostic reagents. In: Harper K, Ziegler A (eds) Recombinant antibodies-application in plant science and plant pathology. Taylor & Francis, London, pp 57–81
Kollerová E, Glasa M, Šubr ZW (2008) Western blotting analysis of the Plum pox virus capsid protein. J Plant Pathol 90(1 Suppl):S119–S122
Krueger RR, Navarro L (2007) Citrus germplasm resources. In: Khan IA (ed) Citrus genetics, breeding and biotechnology. USDA-ARS National Clonal Germplasm Repository for Citrus & Dates, Riverside, CA, pp 45–140
Lu R, Folimonov A, Shintaku M, Li WX, Falk BW, Dawson WO, Ding SW (2004) Three distinct suppressors of RNA silencing encoded by a 20-kb viral RNA genome. Proc Nat Acad Sci USA 101:15742–15747
Moreno P, Ambrós S, Albiach-Martí MR, Guerri J, Peña L (2008) Citrus tristeza virus: a pathogen that changed the course of the citrus industry. Mol Plant Pathol 9:251–268
Nickel H, Kawchuk L, Twyman RM, Zimmermann S, Junghans H, Winter S, Fischer R, Prufer D (2008) Plantibody-mediated inhibition of the Potato leafroll virus P1 protein reduces virus accumulation. Virus Res 136:140–145
Nölke G, Cobanov P, Uhde-Holzem K, Reustle G, Fischer R, Schillberg S (2009) Grapevine fanleaf virus (GFLV)-specific antibodies confer GFLV and Arabis mosaic virus (ArMV) resistance in Nicotiana benthamiana. Mol Plant Pathol 10:41–49
Owen M, Gandecha A, Cockburn B, Whitelam G (1992) Synthesis of a functional antiphytochrome single-chain-Fv protein in transgenic tobacco. Biotechnology 10:790–794
Pappu HR, Pappu SS, Kano T, Koizumi M, Cambra M, Moreno P, Su HJ, Garnsey SM, Lee RF, Niblett CL (1995) Mutagenic analysis and localization of a highly conserved epitope near the amino-terminal end of the citrus tristeza closterovirus capsid protein. Phytopathology 85:1311–1315
Peña L, Cervera M, Juárez J, Navarro A, Pina JA, Navarro L (1997) Genetic transformation of lime (Citrus aurantifolia Swing.): factors affecting transformation and regeneration. Plant Cell Rep 16:731–737
Prins M, Lohuis D, Schots A, Goldbach R (2005) Phage display-selected single-chain antibodies confer high levels of resistance against Tomato spotted wilt virus. J Gen Virol 86:2107–2113
Prins M, Laimer M, Noris E, Schubert J, Wassenegger M, Tepfer M (2008) Strategies for antiviral resistance in transgenic plants. Mol Plant Pathol 9:73–83
Roistacher CN (1991) Techniques for biological detection of specific citrus graft-transmissible diseases. In: Roistacher CN (ed) Graft-transmissible diseases of citrus. Handbook for detection and diagnosis. Food and Agriculture Organisation of the United Nations-FAO, Rome, pp 13–156
Satyanarayana T, Gowda S, Mawassi M, Albiach-Marti MR, Ayllón MA, Robertson C, Garnsey SM, Dawson WO (2000) Closterovirus encoded HSP70 homolog and p61 in addition to both coat proteins function in efficient virion assembly. Virology 278:253–265
Schillberg S, Zimmermann S, Findlay K, Fischer R (2000) Plasma membrane display of anti-viral single chain Fv fragments confers resistance to tobacco mosaic virus. Mol Breed 6:317–326
Tavladoraki P, Benvenuto E, Trinca S, Demartinis D, Cattaneo A, Galeffi P (1993) Transgenic plants expressing a functional single-chain Fv-antibody are specifically protected from virus attack. Nature 366:469–472
Terrada E, Kerschbaumer RJ, Giunta G, Galeffi P, Himmler G, Cambra M (2000) Fully “Recombinant enzyme-linked immunosorbent assays” using genetically engineered single-chain antibody fusion proteins for detection of Citrus tristeza virus. Phytopathology 90:1337–1344
Vela C, Cambra M, Cortés E, Moreno P, Miguet JG, Pérez de San Román C, Sanz A (1986) Production and characterization of monoclonal antibodies specific for Citrus tristeza virus and their use for diagnosis. J Gen Virol 67:91–96
Villani ME, Roggero P, Bitti O, Benvenuto E, Franconi R (2005) Immunomodulation of cucumber mosaic virus infection by intrabodies selected in vitro from a stable single-framework phage display library. Plant Mol Biol 58:305–316
Voss A, Niersbach M, Hain R, Hj Hirsch, Yc Liao, Kreuzaler F, Fischer R (1995) Reduced virus infectivity in Nicotiana tabacum secreting a TMV-specific full-size antibody. Mol Breed 1:39–50
Xiao XW, Chu PWG, Frenkel MJ, Tabe LM, Shukla DD, Hanna PJ, Higgins TJV, Muller WJ, Ward CW (2000) Antibody-mediated improved resistance to ClYVV and PVY infections in transgenic tobacco plants expressing a single-chain variable region antibody. Mol Breed 6:421–431
Zhang MY, Zimmermann S, Fischer R, Schillberg S (2008) Generation and evaluation of movement protein-specific single-chain antibodies for delaying symptoms of Tomato spotted wilt virus infection in tobacco. Plant Pathol 57:854–860
Zimmermann S, Schillberg S, Liao YC, Fisher R (1998) Intracellular expression of TMV-specific single-chain Fv fragments leads to improved virus resistance in Nicotiana tabacum. Mol Breed 4:369–379
Acknowledgments
We thank J.A. Pina and J. Piquer for their technical assistance in growing citrus plants in greenhouses, and P. Serra for his help with western blot analysis. This research was supported by grants Prometeo/2008/121 from the Generalitat Valenciana and AGL2009-08052, RTA2005-00190 and AGL2009-07531 from the Ministerio de Ciencia e Innovación. M. Cervera is recipient of a “Ramón y Cajal” contract. O. Esteban and M. Gil were recipients of PhD fellowships from IVIA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cervera, M., Esteban, O., Gil, M. et al. Transgenic expression in citrus of single-chain antibody fragments specific to Citrus tristeza virus confers virus resistance. Transgenic Res 19, 1001–1015 (2010). https://doi.org/10.1007/s11248-010-9378-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-010-9378-5