Abstract
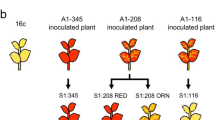
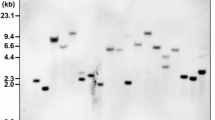
Eight transgenic grapevine lines transformed with the coat protein gene of Grapevine fanleaf virus (GFLV-CP) were analyzed for a correlation between transgene expression, siRNAs production and DNA methylation. Bisulphite genome sequencing was used for a comprehensive analysis of DNA methylation. Methylated cytosine residues of CpG and CpNpG sites were detected in the GFLV-CP transgene, in the T7 terminator and in the 35S promoter of three grapevines without transgene expression, but no detectable level of siRNAs was recorded in these lines. The detailed analysis of 8 lines revealed the complex arrangements of T-DNA and integrated binary vector sequences as crucial factors that influence transgene expression. After inoculation with GFLV, no change in the levels of cytosine methylation was observed, but transgenic and untransformed plants produced short siRNAs (21–22 nt) indicating that the grapevine plants responded to GFLV infection by activating a post-transcriptional gene silencing mechanism.





Similar content being viewed by others
References
An G, Ebert PR, Mitra A, Ha SB (1988) Binary vectors. In: Gelvin SB, Schilperoort RA (eds) Plant molecular biology manual. Kluwer, Dortrecht p A3/1-19
Andret-Link P, Laporte C, Valat L et al (2004) Grapevine fanleaf virus: still a major threat to the grapevine industry. J Plant Pathol 86:183–195
Aufsatz W, Mette MF, van der Winden J et al (2002) RNA-directed DNA methylation in Arabidopsis. Proc Natl Acad Sci USA 99:16499–16506. doi:10.1073/pnas.162371499
Baulcombe D (2004) RNA silencing in plants. Nature 431:356–363. doi:10.1038/nature02874
Cao XF, Aufsatz W, Zilberman D et al (2003) Role of the DRM and CMT3 Methyltransferases in RNA-directed DNA methylation. Curr Biol 13:2212–2217. doi:10.1016/j.cub.2003.11.052
Carra A, Gambino G, Schubert A (2007) A Cetyltrimethyl ammonium bromide -based method to extract low molecular weight RNA from polysaccharide-rich plant tissues. Anal Biochem 360:318–320. doi:10.1016/j.ab.2006.09.022
Chenna R, Sugawara H, Koike T et al (2003) Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res 31:3497–3500. doi:10.1093/nar/gkg500
Fladung M, Kumar S, Ahuja R (1997) Genetic transformation of Populus genotypes with different chimaeric gene constructs: transformation efficiency and molecular analysis. Transgenic Res 6:111–121. doi:10.1023/A:1018421620040
Fusaro AF, Matthew L, Smith NA et al (2006) RNA interference-inducing hairpin RNAs in plants act through the viral defence pathway. EMBO Rep 7:1168–1175. doi:10.1038/sj.embor.7400837
Gambino G, Gribaudo I, Leopold S et al (2005) Molecular characterization of grapevine plants transformed with GFLV resistance genes: I. Plant Cell Rep 24:655–662. doi:10.1007/s00299-005-0006-4
Gambino G, Perrone I, Gribaudo I (2008) A rapid and effective method for RNA extraction from different tissues of grapevine and other woody plants. Phytochem Anal 19:520–525. doi:10.1002/pca.1078
Gambino G, Chitarra W, Maghuly F, et al (2009) Characterization of T-DNA insertions in transgenic grapevines obtained by Agrobacterium-mediated transformation. Mol Breed. doi:10.1007/s11032-009-9293-8
Gölles R, da Câmara Machado A, Tsolova V et al (1998) Transformation of somatic embryos of Vitis sp. with different constructs containing nucleotide sequences from nepovirus coat protein genes. Acta Hortic 447:265–270
Gölles R, Moser M, Pühringer H et al (2000) Transgenic grapevines expressing coat protein gene sequences of Grapevine fanleaf virus, Arabis mosaic virus, Grapevine virus A and Grapevine virus B. Acta Hortic 528:305–311
Hamilton A, Voinnet O, Chappell L, Baulcombe D (2002) Two classes of short interfering RNA in RNA silencing. EMBO J 21:4671–4679. doi:10.1093/emboj/cdf464
Hannon GJ (2002) RNA interference. Nature 418:244–251. doi:10.1038/418244a
Haque AKMN, Yamaoka N, Nishiguchi M (2007) Cytosine methylation is associated with RNA silencing in silenced plants but not with systemic and transitive RNA silencing through grafting. Gene 396:321–331. doi:10.1016/j.gene.2007.04.003
Hetzl J, Foerster AM, Raidl G, Scheid OM (2007) CyMATE: a new tool for methylation analysis of plant genornic DNA after bisulphite sequencing. Plant J 51:526–536. doi:10.1111/j.1365-313X.2007.03152.x
Hily JM, Scorza R, Webb K, Ravelonandro M (2005) Accumulation of the long class of siRNA is associated with resistance to Plum pox virus in a transgenic woody perennial plum tree. Mol Plant Microbe Interact 18:794–799. doi:10.1094/MPMI-18-0794
Kishimoto N, Sakai H, Jackson J et al (2001) Site specificity of the Arabidopsis METI DNA methyltransferase demonstrated through hypermethylation of the superman locus. Plant Mol Biol 46:171–183. doi:10.1023/A:1010636222327
Kumar S, Fladung M (2001) Gene stability in transgenic aspen (Populus). II. Molecular characterization of variable expression of transgene in wild and hybrid aspen. Planta 213:731–740. doi:10.1007/s004250100535
Maghuly F, Machado A, Leopold S et al (2007) Long-term stability of marker gene expression in Prunus subhirtella: a model fruit tree species. J Biotechnol 127:310–321. doi:10.1016/j.jbiotec.2006.06.016
Martinez de Alba AE, Flores R, Hernandez C (2002) Two chloroplastic viroids induce the accumulation of small RNAs associated with posttranscriptional gene silencing. J Virol 76:13094–13096. doi:10.1128/JVI.76.24.13094-13096.2002
McCabe MS, Schepers F, van der Arend A et al (1999) Increased stable inheritance of herbicide resistance in transgenic lettuce carrying a petE promoter-bar gene compared with a CaMV 35S-bar gene. Theor Appl Genet 99:587–592. doi:10.1007/s001220051272
Melquist S, Bender J (2004) An internal rearrangement in an Arabidopsis inverted repeat locus impairs DNA methylation triggered by the locus. Genetics 16:437–448. doi:10.1534/genetics.166.1.437
Mette MF, Aufsatz W, van der Winden J et al (2000) Transcriptional silencing and promoter methylation triggered by double-stranded RNA. EMBO J 19:5194–5201. doi:10.1093/emboj/19.19.5194
Mishiba K, Nishihara M, Nakatsuka T et al (2005) Consistent transcriptional silencing of 35S-driven transgenes in gentian. Plant J 44:541–556. doi:10.1111/j.1365-313X.2005.02556.x
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. doi:10.1111/j.1399-3054.1962.tb08052.x
Rozen S, Skaletsky HJ (2000) Primer3 on the WWW for general users and for biologist programmers. In: Krawetz S, Misener S (eds) Bioinformatics methods and protocols: methods in molecular biology. Humana Press, Totowa, pp 365–386
Selker EU (1999) Gene silencing: repeats that count. Cell 97:157–160. doi:10.1016/S0092-8674(00)80725-4
Sonoda S, Mori M, Nishiguchi M (1999) Homology-dependent virus resistance in transgenic plants with the coat protein gene of Sweet potato feathery mottle potyvirus: target specificity and transgene methylation. Phytopathology 89:385–391. doi:10.1094/PHYTO.1999.89.5.385
Szittya G, Molnár A, Silhavy D et al (2002) Short defective interfering RNAs of tombusviruses are not targeted but trigger post-transcriptional gene silencing against their helper virus. Plant Cell 14:359–372. doi:10.1105/tpc.010366
Thomas MR, Matsumoto S, Cain P, Scott NS (1993) Repetitive DNA of grapevine: classes present and sequences suitable for cultivar identification. Theor Appl Genet 86:173–180
Vaucheret H (2006) Post-transcriptional small RNA pathways in plants: mechanisms and regulations. Genes Dev 20:759–771. doi:10.1101/gad.1410506
Vigne E, Komar V, Fuchs M (2004) Field safety assessment of recombination in transgenic grapevines expressing the coat protein gene of Grapevine fanleaf virus. Transgenic Res 13:165–179. doi:10.1023/B:TRAG.0000026075.79097.c9
Voinnet O (2005) Induction and suppression of RNA silencing: insights from viral infections. Nat Rev Genet 6:206–220. doi:10.1038/nrg1555
Xie Z, Fan B, Chen C, Chen Z (2001) An important role of an inducible RNA-dependent RNA polymerase in plant antiviral defense. Proc Natl Acad Sci USA 98:6516. doi:10.1073/pnas.111440998
Acknowledgments
We thank Federico Ghilino for excellent support in the grafting, Danila Cuozzo and Tiziano Strano for their technical assistance in micropropagation of transgenic grapevines and in greenhouse management.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gambino, G., Perrone, I., Carra, A. et al. Transgene silencing in grapevines transformed with GFLV resistance genes: analysis of variable expression of transgene, siRNAs production and cytosine methylation. Transgenic Res 19, 17–27 (2010). https://doi.org/10.1007/s11248-009-9289-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-009-9289-5