Abstract
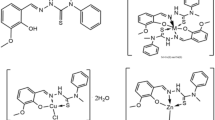
An interesting series of cobalt(II) complexes of the new ligands: 4[N-(benzalidene)amino]antipyrinethiosemicarbazone (BAAPTS), 4[N-(2′-hydroxy-benzalidene)amino]antipyrinethiosemicarbazone (HBAAPTS) and 4[N-(2′-hydroxy-1′-naphthalidene)amino]antipyrinethiosemicarbazone (HNAAPTS) were synthesized by reaction with Co(II) salts in ethanol. The general stoichiometry of the complexes was found to be [CoX2(H2O)(L)] and [Co(L)2](ClO4)2, where X = Cl, NO3, NCS or CH3COO and L = BAAPTS, HBAAPTS or HNAAPTS. The complexes were characterized by elemental analysis, molar conductivity measurement, molecular weight determination, magnetic moments at room temperature, infrared and electronic spectra. All the thiosemicarbazones behave as neutral tridentate (N, N, S) donor ligands. The conductivity measurements in PhNO2 solution indicated that the chloro, nitrato, thiocyanato and acetate complexes are essentially non-electrolytes, while the perchlorate complexes are 1:2 electrolytes. Thermogravimetric studies were performed for some representative complexes and the decomposition mechanism proposed. Antibacterial and antifungal properties of the ligands and their cobalt(II) complexes have also been examined and it has been observed that the complexes are more potent bactericides than the ligand.
Similar content being viewed by others
References
Scovill J.P., Klayman D.L. and Franchino C.F. (1982). J. Med. Chem. 25: 1261
Ali M.A., Chowdhary D.A. and Nazimuddin M. (1984). Polyhedron 3: 595
Hossain M.E., Alam M.N., Begum J., Ali M.A., Nazimuddin M., Smith F.E. and Hynes R.C. (1996). Inorg. Chim. Acta. 249: 207
Bindu P., Kurup M.R.P. and Satyakeerthy T.R. (1999). Polyhedron 18: 321
Agarwal R.K. and Prakash B. (2005). Trans. Metal Chem. 30: 696
Agarwal R.K., Prasad S. and Gahlot N. (2004). Turk. J. Chem. 28: 691
Agarwal R.K. and Prasad S. (2005). Turkish J. Chem. 29: 289
Agarwal R.K. and Prasad S. (2005). Bioinorg. Chem. Appl. 3: 271
Agarwal R.K., Bhushan B. and Singh G.B. (1993). J. Inst. Chem. (India) 65: 131
Welcher F.J. (1965). The Analytical Uses of Ethylenediamine Tetraacetic Acid. D. Van Nostrand Co., Inc., NY
Vogel A.I. (1978). A Text Book of Inorganic Quantitative Analysis. Green, London
Kurz E., Kober G. and Berl M. (1958). Anal. Chem. 30: 1983
Methods for Evaluating Plant Fungicides, Nematicides and Bactericides, Annon American Phytopathological Society, Minnisota, USA, 1978, p. 141.
B. N. Figgis and R. S. Nyholm, J. Chem. Soc., 338 (1959)
Kato M., Jonassen H.B. and Fanning J.C. (1964). Chem. Rev. 64: 99
Yamada S. (1966). Coord. Chem. Rev. 1: 415
Radhakrishnan P.S. and Indrasenan P. (1990). J. Ind. Chem.Soc. 67: 243
Lane S.T., Yamagauchi A. and James A.R. (1958). J. Am. Chem. Soc. 80: 527
Rana V.B. (1975). J. Inorg. Nucl. Chem. 37: 1826
P. W. Sadler, J. Chem. Soc., 957 (1961)
Singh B. and Mishra H. (1986). J. Ind. Chem. Soc. 63: 692, 1069
Aravindakshan K.K. (1987). Indian J. Chem. 26A: 241
Krishnamurthy S.K. and Soundararajan S. (1969). Can. J. Chem. 47: 995
Burmeister J.L. (1968). Coord. Chem. Revs. 3: 225
Hester R.W. and Grossman W.L. (1966). Inorg. Chem. 5: 1308
Lever A.B.P., Mantiovani E. and Ramaswamy B.S. (1971). Can. J. Chem. 49: 1957
A. B. P. Lever and B. S. Ramaswamy, J. Chem. Soc., 5042 (1963).
Reedijk J., Driessen W.L. and Groeneveld W.L. (1969). Recl. Trav. Chim. 88: 1095
Freeman F.S. and Carroll B. (1958). J. Phys. Chem. 62: 394
Coats A.W. and Redfern J.P. (1964). Nature 201: 68
Horowitz H.H. and Metzger G. (1958). Anal. Chem. 35: 1454
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Prasad, S., Agarwal, R.K. Cobalt(II) complexes of various thiosemicarbazones of 4-aminoantipyrine: syntheses, spectral, thermal and antimicrobial studies. Transition Met Chem 32, 143–149 (2007). https://doi.org/10.1007/s11243-006-0119-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-006-0119-9