Abstract
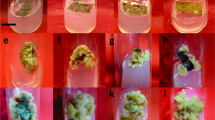
The present study reports an optimized protocol for high frequency in vitro plant propagation through direct and indirect organogenesis, phytochemical accumulation, molecular profiling and antioxidant evaluation for micropropagated Swertia minor, a promising alternative to industrially important Swertia chirayita. Moreover, the study also aimed at enhancing the production of antidiabetic and anti-obesity drug swertiamarin using an alternative technology of elicitated cell suspension cultures. Different types, concentrations and combination of cytokinins and auxins showed their effects during various in vitro growth stages. A combination of BAP (3.0 mg/l) and TDZ (1.0 mg/l) had a dominant role in promoting multiple shoot proliferation with production of an average of 19.1 ± 0.95 shoots/node in 85% response. MS medium added with IBA (2.0 mg/l) showed optimal response for in vitro rooting (9.2 ± 0.56 roots/shoot). In order to establish genetic stability, molecular marker-based profiling of micropropagated plants were done and 'monomorphic banding pattern were identical to the mother plants. 2,4-D (2.0 mg/l) supported the maximum callus induction and proliferation rate (95%). The wild-grown plants showed higher polyphenols content and antioxidant activities as compared to callus and in vitro derived plantlets. However, chitosan-treated (25 ppm) methanolic extract of cell biomass accumulated in cell suspension cultures produced higher contents of swertiamarin (1.45 mg/g DW) than salicylic acid and methyl jasmonate. The described protocol can be effectively used for the large-scale propagation, exploitation of active compounds and will serve as potential alternative to S. chirayita for fulfillment of over-growing industrial requirements.
Key message
The present investigation addressed in vitro regeneration, callus culture, somatic embryogenesis, molecular profiling, secondary metabolite production, cell suspension culture studies for first time in Swertia minor.





Similar content being viewed by others
Abbreviations
- ABTS:
-
2, 2-azino-bis (3-ethylbenzo-thiazoline-6-sulphonic acid)
- AEAC:
-
Ascorbic acid equivalent antioxidant capacity
- DMPD:
-
N, N-dimethyl-p-phenylenediamine
- DPPH:
-
1, 1- diphenyl-1-picryl hydrazyl
- DW:
-
Dry weight
- FRAP:
-
Ferric reducing antioxidant power
- FW:
-
Fresh weight
- g:
-
Gram
- MeJA:
-
Methyl jasmonate
- mg:
-
Milligram
- MS:
-
Murashige and Skoog’s medium
- PGR(s):
-
Plant growth regulator(s)
- RF:
-
Regeneration frequency
- RP-UFLC:
-
Reverse phase-ultra flow liquid chromatography
- RSA:
-
Radical scavenging activity
- SA:
-
Salicylic acid
References
Anonymous (1982) The Wealth of India: raw materials, vol. X. Publication and Information Directorate. CSIR, New Delhi, pp 78–81
Balaraju K, Agastian P, Ignacimuthu S (2009) Micropropagation of Swertia chirata Buch.-Hams. ex Wall.: a critically endangered medicinal herb. Acta Physiol Plant 31:487–494
Balaraju K, Saravanan S, Agastian P, Ignacimuthu S (2011) A rapid system for micropropagation of Swertia chirata Buch- Ham. ex Wall.: an endangered medicinal herb via direct somatic embryogenesis. Acta Physiol Plant 33:1123–1133
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: the FRAP assay. Anal Biochem 239:70–76
Brahmachari G, Mondal S, Gangopadhyay A, Gorai D, Mukhopadhyay B, Saha S, Brahmachari AK (2004) Swertia (Gentianaceae): chemical and pharmacological aspects. Chem Biodivers 1:1627–1651
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of free radical method to evaluate antioxidant activity. Lebensm Wiss Technol 28:25–30
Chavan JJ, Gaikwad NB, Dixit GB, Yadav SR, Bapat VA (2018) Biotechnological interventions for propagation, conservation and improvement of ‘Lantern Flowers’ (Ceropegia spp.). S Afr J Bot 114:192–216
Chavan JJ, Gaikwad NB, Kshirsagar PR, Umdale SD, Bhat KV, Dixit GB, Yadav SR (2015) Highly efficient in vitro proliferation and genetic stability analysis of micropropagated Ceropegia evansii by RAPD and ISSR markers: a critically endangered plant of Western Ghats. Plant Biosyst 149:442–450
Chavan JJ, Gaikwad NB, Yadav SR (2013) High multiplication frequency and genetic stability analysis of Ceropegia panchganiensis, a threatened ornamental plant of Western Ghats: conservation implications. Sci Hortic 161:134–142
Chavan JJ, Nimbalkar MS, Gaikwad NB, Dixit GB, Yadav SR (2011) In vitro propagation of Ceropegia spiralis Wight - an endemic and rare potential ornamental plant of peninsular India. Proc Natl Acad Sci India Sect B Biol Sci 81:120–126
Chokkalingam U, Kumarasamy C, Mathan S, Athar A, Palathurai SM (2012) Antioxidant and structure–activity relationships of five tetra-oxygenated xanthones from Swertia minor. Knobl Nat Prod Res 26(13):1265–1270
Fogliano V, Verde V, Randazzo G, Ritieni A (1999) Method for measuring antioxidant activity and its application to monitoring the antioxidant capacity of wines. J Agric Food Chem 47(3):1035–1040
Jha TB, Dafadar A, Chaudhuri RK (2011) Somatic embryogenesis in Swertia chirata Buch. Ham. ex Wall. - a multipotent medicinal herb. Asian J Biotechnol 3:186–193
Joshi P, Dhawan V (2007) Assessment of genetic fidelity of micropropagated Swertia chirayita plantlets by ISSR marker assay. Biol Plantarum 51:22–26
Kaur P, Gupta RC, Dey A, Malik T, Pandey DK (2020) Optimization of salicylic acid and chitosan treatment for bitter secoiridoid and xanthone glycosides production in shoot cultures of Swertia paniculata using response surface methodology and artificial neural network. BMC Plant Biol 20:225
Kshirsagar P, Chavan J, Nimbalkar M, Yadav S, Dixit G, Gaikwad N (2015) Phytochemical composition, antioxidant activity and HPLC profiles of Swertia species from Western Ghats. Nat Prod Res 29:780–784
Kshirsagar P, More T, Arvindekar A, Gaikwad N (2014) Antioxidant, antihyperglycemic and antiglycation properties of some Swertia species from Western Ghats. Int J Pharm Pharm Sci 6:303–306
Kshirsagar P, Umdale S, Chavan J, Gaikwad N (2017) Molecular authentication of medicinal plant, Swertia chirayita and its adulterant species. Proc Natl Acad Sci India Sect B Biol Sci 87(1):101–107
Kshirsagar PR, Chavan JJ, Gaikwad NB, Pai SR, Bapat VA (2020) Metabolite profiling, antioxidant potential and RP - UFLC determination of bioactive molecules from eight Swertia species. Biocatal Agric Biotechnol 23:101479
Kshirsagar PR, Chavan JJ, Umdale SD, Nimbalkar MS, Dixit GB, Gaikwad NB (2015) Highly efficient in vitro regeneration, establishment of callus and cell suspension cultures and RAPD analysis of regenerants of Swertia lawii Burkill. Biotechnol Rep 6:79–84
Kshirsagar PR, Gaikwad NB, Pai SR, Bapat VA (2017) Optimization of extraction techniques and quantification of swertiamarin and mangiferin by using RP-UFLC method from eleven Swertia species. S Afr J Bot 108:81–89
Kshirsagar PR, Pai SR, Nimbalkar MS, Gaikwad NB (2016) RP-HPLC analysis of seco-irridoid glycoside Swertiamarin from different Swertia species. Nat Prod Res 30:865–868
Kshirsagar PR, Jagtap UB, Gaikwad NB, Bapat VA (2019) Ethnopharmacology, phytochemistry and pharmacology of medicinally potent genus Swertia: an update. S Afr J Bot 124:444–483
Kumar V, Chandra S (2013) Efficient regeneration and antioxidant activity of the endangered species Swertia chirayita. Int J Bio Sci 4(4):823–833
Kumar RR, Purohit VK, Prasad P, Nautiyal AR (2018) Efficient in vitro propagation protocol of Swertia chirayita (Eoxb. ex Fleming) Karsten: a critically endangered medicinal plant. Natl Acad Sci Lett 41:123–127
Kumar V, Singh SK, Bandopadhyay R, Sharma MM, Chandra S (2014) In vitro organogenesis secondary metabolite production and heavy metal analysis in Swertia chirayita. Cent Eur J Biol 9:686–698
Kumar V, Van Staden J (2016) A review of Swertia chirayita (Gentianaceae) as a traditional medicinal plant. Front Pharmacol 6:308
Ludwig-Müller J (2000) Indole-3-butyric acid in plant growth and development. Plant Growth Regul 32(2):219–230
Mahendran G, Narmatha Bai V (2014) Micropropagation, antioxidant properties and phytochemical assessment of Swertia corymbosa (Griseb.) Wight ex C. B. Clarke: a medicinal plant. Acta Physiol Plant 36:589–603
Mahendran G, Narmatha Bai V (2017) Plant regeneration through direct somatic embryogenesis, antioxidant properties, and metabolite profiles of Swertia corymbosa (Griseb.) Wight ex C.B. Clarke. Plant Biosyst 151(1):39–49
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays for tobacco tissue cultures. Physiol Plant 15:473–497
Pant M, Bisht P, Gusain MP (2012) In vitro propagation through root-derived callus culture of Swertia chirata Buch. -Ham. ex Wall. Afr J Biotechnol 11(29):7408–7416
Pliankong P, Suksa-Ard P, Wannakrairoj S (2018) Chitosan elicitation for enhancing of vincristine and vinblastine accumulation in cell culture of Catharanthus roseus (L.) G. Don. J Agric Sci 10(12):287–293
Roy P, Abdulsalam FI, Pandey DK, Bhattacharjee A, Eruvaram NR, Malik T (2015) Evaluation of antioxidant, antibacterial, and antidiabetic potential of two traditional medicinal plants of India: Swertia cordata and Swertia chirayita. Pharmacogn Res 7(1):S57–S62
Saha N, Dutta Gupta S (2018) Promotion of shoot regeneration of Swertia chirata by biosynthesized silver nanoparticles and their involvement in ethylene interceptions and activation of antioxidant activity. Plant Cell Tiss Organ Cult 134:289–300
Scartezzini P, Speroni E (2000) Review on some plants of Indian traditional medicine with anti-oxidative activity. J Ethnopharmacol 71:23–43
Sharma V, Belwal N, Kamal B, Dobriyal AK, Jadon VS (2016) Assessment of genetic fidelity of in vitro raised plants in Swertia chirayita through, ISSR RAPD analysis and peroxidase profiling during organogenesis. Braz Arch Biotechnol. https://doi.org/10.1590/1678-4324-2016160389
Subramanya MD, Pai SR, Upadhya V, Ankad GM, Bhagwat SS, Hegde HV (2015) Total polyphenolic contents and in vitro antioxidant properties of eight Sida species from Western Ghats, India. J Ayurveda Integr Med 6:24–28
Acknowledgements
First author gratefully acknowledges the financial support by UGC New Delhi India for awarding Dr. D. S. Kothari PDF (No.F.42/2006 (BSR)/BL/1415/0461). Authors extend their gratitude towards the Head, Department of Biotechnology, Shivaji University, Kolhapur and the Principal, Yashavantrao Chavan Institute of Science (Autonomous), Satara for providing necessary laboratory facilities.
Author information
Authors and Affiliations
Contributions
PRK, JJC, NBG designed the experiment, performed tissue culture experiments, contributed to writing and corrected manuscript. PRK, AM and SS performed phytochemical experiments, collected and analyzed data, wrote the manuscript. JJC, NBG and VAB helped in experimental accomplishment, corrected manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated By Amita Bhattacharya.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kshirsagar, P.R., Mohite, A., Suryawanshi, S. et al. Plant regeneration through direct and indirect organogenesis, phyto-molecular profiles, antioxidant properties and swertiamarin production in elicitated cell suspension cultures of Swertia minor (Griseb.) Knobl. Plant Cell Tiss Organ Cult 144, 383–396 (2021). https://doi.org/10.1007/s11240-020-01962-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-020-01962-8