Abstract
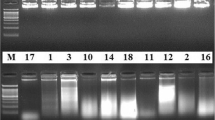
A reliable and reproducible protocol for in vitro regeneration has been developed for Alhagi maurorum, a rare and medicinally important plant of family fabaceae. MS medium with BAP (2.0 mg l−1) proved to be the best for shoot bud induction from nodal segments. The rate of shoot multiplication was found to be influenced by a number of factors, viz., media composition, plant growth regulator’s type and concentration, successive transfer of mother explant for different passage, culture vessels and gelling agents. Modified MS medium (modified having nitrates reduced to half) solidified with 0.14 % gelrite and containing BAP (0.5 mg l−1), IAA (0.1 mg l−1) and additives was found optimum for shoot multiplication which gave rise to maximum number of shoots (33.5 ± 3.43 per culture vessel). The in vitro regenerated shoots were rooted under both in vitro (on half strength MS salts with 1.0 mg l−1 IBA + 100 mg l−1 activated charcoal) as well as ex vitro (on sterile soilrite by treating shoot base with 250 mg l−1 each of IBA and NOA for 4 min in green house) conditions. Thereafter, the in vitro and ex vitro rooted plantlets were hardened under green house conditions with 70 and 90 % survival rate, respectively. Start codon targeted (SCoT), inter simple sequence repeats (ISSR) and random amplified polymorphic DNA (RAPD) markers were used to validate the genetic homogeneity of seven tissue cultured plantlets growing in green house condition with mother plant. The amplification products were monomorphic across all the seven micropropagated plants as well as mother plant produced by all SCoT, ISSR and RAPD primers applied. The monomorphic banding pattern in micropropagated plants and the mother plant confirms the genetic homogeneity of the in vitro raised plants and demonstrates the reliability of our in vitro propagation system for A. maurorum. To the best of our knowledge, this is the first report on micropropagation and genetic homogeneity assessment of A. maurorum, which can be applied for large scale multiplication of elite genotypes of A. maurorum.





Similar content being viewed by others
Abbreviations
- AC:
-
Activated charcoal
- BAP:
-
6-benzylaminopurine
- IAA:
-
Indole-3-acetic acid
- IBA:
-
Indole-3-butyric acid
- ISSR:
-
Inter simple sequence repeats
- Kn:
-
Kinetin
- MMS:
-
Modified Murashige and Skoog
- NAA:
-
Naphthalene acetic acid
- NOA:
-
Naphthoxy acetic acid
- PFD:
-
Photon flux density
- PGR:
-
Plant growth regulator
- RAPD:
-
Random amplified polymorphic DNA
- RH:
-
Relative humidity
- SCoT:
-
Start codon targeted
References
Akhani M, Ghorbanli M (1993) A contribution to the halophytic vegetation and flora of Iran. In: Lieth H, Al Masoom A (eds) Towards the rational use of high salinity tolerant plants 1:35–44
Benmahioul B, Dorion N, Kaid-Harche M, Daguin F (2012) Micropropagation and ex vitro rooting of Pistachio (Pistacia vera L.). Plant Cell Tiss Organ Cult 108:353–358
Carra A, Sajeva M, Abbate L, Siragusa M, Sottile F, Carimi F (2012) In vitro plant regeneration of caper (Capparis spinosa L.) from floral explants and genetic stability of regenerants. Plant Cell Tiss Organ Cult 109:373–381
Collard BCY, Mackill DJ (2009) Start codon targeted (SCoT) polymorphism: a simple, novel DNA marker technique for generating gene-targeted markers in plants. Plant Mol Biol Rep 27:86–93
Gupta AK, Harish, Rai MK, Phulwaria M, Shekhawat NS (2011) Isolation of genomic DNA suitable for community analysis from mature trees adapted to arid environment. Gene 487:156–159
Gupta AK, Harish, Rai MK, Phulwaria M, Agarwal T, Shekhawat NS (2014) In vitro propagation, encapsulation, and genetic fidelity analysis of Terminalia arjuna: a cardioprotective medicinal tree. Appl Biochem Biotechnol 173:1481–1494. doi:10.1007/s12010-014-0920-4
Hamed A, Perrone A, Mahalel U, Oleszek W, Stochmal A, Piacente S (2012) Oleanane glycosides from the roots of Alhagi maurorum. Phytochem Lett 5:782–787
Hameed M, Ashraf M, Al-Quriany F, Nawaz T, Ahmad MSA, Younis A, Naz N (2011) Medicinal flora of the Cholistan desert: a review. Pak J Bot 43:39–50
Harish, Gupta AK, Phulwaria M, Rai MK, Shekhawat NS (2014) Conservation genetics of endangered medicinal plant Commiphora wightii in Indian Thar Desert. Gene 535:266–272
Hassanein AM, Mazen AMA (2001) Adventitious bud formation in Alhagi graecorum. Plant Cell Tiss Organ Cult 65:31–35
Honari M, Askari H, Khosrowchahli M (2014) Use of desirability function method in optimization of regeneration and callus induction of Alhagi camelorum. Am J Plant Sci 5:268–274
Islam MT, Dembele DP, Keller ERJ (2005) Influence of explant, temperature and different culture vessels on in vitro culture for germplasm maintenance of four mint accessions. Plant Cell Tiss Organ Cult 81:123–130
Kaçar YA, Biçen B, Varol I, Mendi YY, Serçe S, Çetiner S (2010) Gelling agents and culture vessels affect in vitro multiplication of banana plantlets. Genet Mol Res 9:416–424
Laghari AH, Memon S, Nelofar A, Khan KM, Yasmin A, Syed MN, Aman A (2010) A new flavanenol with urease-inhibition activity isolated from roots of manna plant camelthorn (Alhagi maurorum). J Mol Struct 965:65–67
Laghari AH, Memona S, Nelofarb A, Khan KM (2011) Alhagi maurorum: a convenient source of lupeol. Ind Crop Prod 34:1141–1145
Lakshmanan V, Venkataramareddy SR, Neelwarne B (2007) Molecular analysis of genetic stability in long-term micropropagated shoots of banana using RAPD and ISSR markers. Electron J Biotechnol 10:106–113
Larkin P, Scowcroft W (1981) Somaclonal variation—a novel source of variability from cell culture for plant improvement. Theor Appl Genet 60:197–214
Letham DS, Palni LMS (1983) The biosynthesis and metabolism of cytokinins. Ann Rev Plant Physiol 34:163–197
Marashdah MS, AL-Hazimi HM (2010) Pharmacological activity of ethanolic extract of Alhagi maurorum roots. Arab J Chem 3:39–42
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nestakova M, Havrlentová M, Faragó J (2000) Effect of gelling agents on in vitro multiplication of two ornamental plants. Biologia 55:409–411
Patel AK, Phulwaria M, Rai MK, Gupta AK, Shekhawat S, Shekhawat NS (2014) In vitro propagation and ex vitro rooting of Caralluma edulis (Edgew.) Benth. & Hook. f.: an endemic and endangered edible plant species of the Thar Desert. Sci Horti 165:175–180
Phulwaria M, Rai MK, Harish, Gupta AK, Ram K, Shekhawat NS (2012) An improved micropropagation of Terminalia bellirica from nodal explants of mature tree. Acta Physiol Plant 34:299–305
Rai MK, Asthana P, Jaiswal VS, Jaiswal U (2010) Biotechnological advances in guava (Psidium guajava L.): recent developments and prospects for further research. Trees Struct Funct 24:1–12
Rai MK, Phulwaria M, Harish, Gupta AK, Shekhawat NS, Jaiswal U (2012) Genetic homogeneity of guava plants derived from somatic embryogenesis using SSR and ISSR markers. Plant Cell Tiss Organ Cult 111:259–264
Ram K, Shekhawat NS (2012) Micropropagation of commercially cultivated henna (Lawsonia inermis) using nodal explants. Physiol Mol Biol Plants 17:281–289
Rathore NS, Rai MK, Phulwaria M, Rathore N, Shekhawat NS (2014) Genetic stability in micropropagated Cleome gynandra revealed by SCoT analysis. Acta Physiol Plant 36:555–559
Rawat JM, Rawat B, Agnihotri RK, Chandra A, Nautiyal S (2013) In vitro propagation, genetic and secondary metabolite analysis of Aconitum violaceum Jacq.: a threatened medicinal herb. Acta Physiol Plant 35:2589–2599
Samejo MQ, Memon S, Bhanger MI, Khan KM (2012) Chemical composition of essential oils from Alhagi maurorum. Chem Nat Compd 48:5
Shaker E, Mahmoud H, Mnaa S (2010) Anti-inflammatory and anti-ulcer activity of the extract from Alhagi maurorum (camelthorn). Food Chem Toxicol 48:2785–2790
Shekhawat NS, Mughal MH, Johri BM, Srivastava PS (1998) Indian contribution to plant tissue and organ culture. In: Srivastava PS (ed) Plant tissue culture and molecular biology: application and prospects. Narosa Publishing House, New Delhi, pp 751–811
Shekhawat NS, Phulwaria M, Harish, Rai MK, Kataria V, Shekhawat S, Gupta AK, Rathore NS, Vyas M, Rathore N, Vibha JB, Choudhary SK, Patel AK, Lodha D, Modi R (2012) Bioresearches of fragile ecosystem/desert. Proceed Nat Acad Sci India Sect B Biol Sci 82:319–334
Singh SK, Rai MK, Asthana P, Sahoo L (2009) An improved micropropagation of Spilanthes acmella through thin cell layer culture. Acta Physiol Plant 31:693–698
Thiyagarajan M, Venkatachalam P (2012) Large scale in vitro propagation of Stevia rebaudiana (bert) for commercial application: pharmaceutically important and antidiabetic medicinal herb. Ind Crop Prod 37:111–117
Thomas TD (2008) The role of activated charcoal in plant tissue culture. Biotechnol Adv 26:618–631
Tripathi M, Kumari N (2010) Micropropagation of a tropical fruit tree Spondias mangifera Willd. through direct organogenesis. Acta Physiol Plant 32:1011–1015
Yan H, Liang C, Yang L, Li Y (2010) In vitro and ex vitro rooting of Siratia grosvenorii—a traditional medicinal plant. Acta Physiol Plant 32:115–120
Yokawa K, Kagenishi T, Baluska F (2013) Root photomorphogenesis in laboratory-maintained Arabidopsis seedlings. Trends Plant Sci 18:117–119
You Y, Nguyen-Hai N, Yong K, Ki-Hwan B, Byung-Zun A (2003) Antiangiogenic activity of lupeol from Bombax ceiba. Phytother Res 17:341–344
Acknowledgments
We are thankful to Council of Scientific and Industrial Research (CSIR), New Delhi for financial assistance in the form of Junior Research Fellowship (JRF) to authors T.A. and Senior Research Fellowship (SRF) to A.K.P., respectively. We are also thankful to Mr. Ramesh Dagla for his assistance in collecting the plant material.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agarwal, T., Gupta, A.K., Patel, A.K. et al. Micropropagation and validation of genetic homogeneity of Alhagi maurorum using SCoT, ISSR and RAPD markers. Plant Cell Tiss Organ Cult 120, 313–323 (2015). https://doi.org/10.1007/s11240-014-0608-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-014-0608-z