Abstract
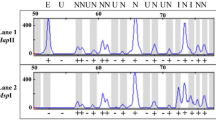
The best known and most thoroughly studied epigenetic phenomenon is DNA methylation, which plays an important role in regulating gene expression during plant regeneration and development. In this study, the methylation-sensitive amplified polymorphism (MSAP) technique was carried out to determine differences in methylation profiles between two forms of protocorm-like bodies (PLBs), continuously proliferating PLBs (cPLBs) and spontaneously-differenting PLBs (sdPLBs), derived from cultures of Cymbidium hybridium. A total of 72 selective primer combinations were used to assess the status of cytosine methylation of DNA in these tissues. Of 4,440 fragments obtained 911 fragments, each representing a recognition site cleaved by one or both of the isoschizomers (Hpa II and Msp I), were amplified and were significantly different between the two forms of PLBs. Frequency of total and full-methylation of cPLBs and sdPLBs were 26.7/12.2%, 24.1/11.1%, respectively. In addition, 14 types of MSAP patterns detected in the two forms of PLBs belonged to two classes, type I and II. Sequencing of 14 differentially methylated fragments and their subsequent blast search revealed that cytosine methylated 5′-CCGG-3′ sequences were equally distributed in the coding and non-coding regions. Southern blotting was conducted to verify the methylation polymorphism.




Similar content being viewed by others
Abbreviations
- 6-BA:
-
N6-benzylaminopurine
- NAA:
-
α-Naphthalene acetic acid
- MSAP:
-
Methylation-sensitive amplified polymorphism
- PLB:
-
Protocorm-like body
References
Ashikawa I (2001) Surveying CpG methylation at 5′-CCGG in the genomes of rice cultivars. Plant Mol Biol 45:31–39. doi:10.1023/A:1006457321781
Borg S, Brandstrup B, Jensen TJ, Poulsen C (1997) Identification of new protein species among 33 different small GTP-binding proteins encoded by cDNAs from Lotus japonicus, and expression of corresponding mRNAs in developing root nodules. Plant J 11:237–250. doi:10.1046/j.1365-313X.1997.11020237.x
Cervera MT, Ruiz-Garcia L, Martinez-Zapater JM (2002) Analysis of DNA methylation in Arabidopsis thaliana based on methylation-sensitive AFLP markers. Mol Genet Genomics 268:543–552. doi:10.1007/s00438-002-0772-4
Chan SW, Henderson IR, Jacobsen SE (2005) Gardening the genome: DNA methylation in Arabidopsis thaliana. Nat Rev Genet 6:351–360. doi:10.1038/nrg1601
Chang YJ, Jae YL, Joon K (2004) RPS3, a DNA repair endonuclease and ribosomal protein, is involved in apoptosis. FEBS Lett 560:81–85. doi:10.1016/S0014-5793(04)00074-2
Cubas P, Vincent C, Coen E (1999) An epigenetic mutation responsible for natural variation in floral symmetry. Nature 401:157–161. doi:10.1038/43657
Finnegan EJ, Peacock WJ, Dennis ES (1996) Reduced DNA methylation in Arabidopsis thaliana results in abnormal plant development. Proc Natl Acad Sci USA 93:8449–8454. doi:10.1073/pnas.93.16.8449
Finnegan EJ, Peacock WJ, Dennis ES (2000) DNA methylation, a key regulation of plant development and other processes. Curr Opin Genet Dev 10:217–223. doi:10.1016/S0959-437X(00)00061-7
Hong L, Deng XX (2005) Analysis of DNA Methylation in Navel Oranges Based on MSAP Marker. Sci Agric Sin 38:2301–2307 in Chinese
Honma T, Goto K (2001) Complexes of MADS-box proteins are sufficient to convert leaves into floral organs. Nature 409:525–529. doi:10.1038/35054083
Horvath E, Szalai G, Janda T (2003) Effect of vernalization and 5-azacytidine on the methylation level of DNA in wheat (Triticum aestivum L., cv. Martonvasar 15). Plant Sci 165:689–692. doi:10.1016/S0168-9452(03)00221-8
Jacobsen SE, Sakai H, Finnegan EJ (2000) Ectopic hypermethylation of flower-specific genes in Arabidopsis. Curr Biol 10:179–186. doi:10.1016/S0960-9822(00)00324-9
Janousek B, Matsunaga S, Kejnovsky E (2002) DNA methylation analysis of a male reproductive organ specific gene (MROS1) during pollen development. Genome 45:930–938. doi:10.1139/g02-052
Leutwiler LS, Hough-Evans BR, Meyerowitz EM (1984) The DNA of Arabidopsis thaliana. Mol Gen Genet 194:15–23. doi:10.1007/BF00383491
Li ML, Wang QM, Zhu ZJ (2002) Studies on the changes of DNA methylation level, GA content and protein in non-heading Chinese cabbage during vernalization. Acta Hortic Sin 29:353–357 in Chinese
Lim Y, Lee SM, Lee JY, Moon EP, Lee BJ, Kim J (2002) Complete genomic structure of human rpS3: identification of functional U15b snoRNA in the fifth intron. Gene 286:291–297. doi:10.1016/S0378-1119(02)00502-4
Liu B, Brubaker CL, Mergeai G, Cronn RC, Wendel JF (2001) Polyploid formation in cotton is not accompanied by rapid genomic changes. Genome 44:321–330. doi:10.1139/gen-44-3-321
Liu FP, Hong LP, Zheng Q (2007) Effects of 6-BA, 2, 4-D on PLB explants induction from Phalaenopsis. Acta Agr Jiangxi 8:69–71 in Chinese
Lu GY, Wu XM, Chen BY (2005) MSAP analysis of DNA methylation during and following Brassica rapa seed germination. Chin Sci Bull 24:2750–2756 in Chinese
Lu Y, Rong TZ, Cao MJ (2008) Analysis of DNA methylation in different maize tissues. J Genet Genomics 35:41–48. doi:10.1016/S1673-8527(08)60006-5
Luff B, Pawlowski L, Bender J (1999) An inverted repeat triggers cytosine methylation of identical sequences in Arabidopsis. Mol Cell 3:505–511. doi:10.1016/S1097-2765(00)80478-5
Lund G, Messing J, Viotti A (1995) Endosperm specific demethylation and activation of specific alleles of alpha-tubulin genes of Zea mays. Mol Gen Genet 246:716–722. doi:10.1007/BF00290717
Madlung A, Masuelli RW, Watson B, Reynolds SH, Davison J, Comai L (2002) Remodeling of DNA methylation and phenotypic and transcriptional changes in synthetic Arabidopsis allotetraploids. Plant Physiol 129:733–746. doi:10.1104/pp.003095
Martienssen RA, Colot V (2001) DNA methylation and epigenetic inheritance in plants and lamentous fungi. Science 293:1070–1074. doi:10.1126/science.293.5532.1070
McClelland M, Nelson M, Raschke E (1994) Effect of site-specific modification on restriction endonucleases and DNA modification methyltransferases. Nucleic Acids Res 22:3640–3659. doi:10.1093/nar/22.17.3640
Messeguer R, Ganal MW, Stevens JC, Tanksley SD (1991) Characterization of the level, target sites and inheritance of cytosine methylation in tomato nuclear DNA. Plant Mol Biol 16:753–770. doi:10.1007/BF00015069
Moore PB (1998) The three-dimensional structure of the ribosome and its components. Annu Rev Bioph Biom 27:35–38. doi:10.1146/annurev.biophys.27.1.35
Murray MG, Thompson WF (1980) Rapid isolation of high weight plant DNA. Nucleic Acids Res 8:4231–4233. doi:10.1093/nar/8.19.4321
Noyer JL, Causse S, Tomekpe K, Bouet A, Baurens FC (2005) A new image of plantain diversity assessed by SSR, AFLP and MSAP markers. Genetica 124:61–69. doi:10.1007/s10709-004-7319-z
Paszkowski J, Whitham SA (2001) Gene silencing and DNA methylation. Curr Opin Plant Biol 4:123–129. doi:10.1016/S1369-5266(00)00147-3
Portis E, Acquadro A, Comino C (2004) Analysis of DNA methylation during germination of peper (Capsicum annuum L) seeds using methylation-sensitive amplification polymorphism (MSAP). Plant Sci 166:169–178. doi:10.1016/j.plantsci.2003.09.004
ReynaLopez GE, Simpson J, RuizHerresa J (1997) Differences in DNA methylation patterns are detectable during the dimorphic transition of fungi by amplification of restriction polymorphisms. Mol Gen Genet 253:703–710. doi:10.1007/s004380050374
Riddle NC, Richards EJ (2002) The control of natural variation in cytosine methylation in Arabidopsis. Genetics 162:355–363
Rossi V, Motto M, Pellegrini L (1997) Analysis of the methylation pattern of the maize Opaque-2 (02) promoter and in vitro binding studies indicate that the 02 B-Zip protein and other endosperm factors can bind to methylated target sequences. J Biol Chem 272:13758–13771. doi:10.1074/jbc.272.21.13758
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Sha AH, Lin XH, Huang JB, Zhang DP (2005) Analysis of DNA methylation related to rice adult plant resistance to bacterial blight based on methylation-sensitive AFLP (MSAP) analysis. Mol Genet Genomics 273:484–490. doi:10.1007/s00438-005-1148-3
Shaked H, Kashkush K, Ozkan H, Feldman M, Levy AA (2001) Sequence elimination and cytosine methylation are rapid and reproducible responses of the genome to wide hybridization and allopolyploidy in wheat. Plant Cell 13:1749–1759
Sherman JD, Talbert LE (2002) Vernalization induced changes of the DNA methylation pattern in winter wheat. Genome 45:253–260. doi:10.1139/g01-147
Soppe WJ, Jacobsen SE, Alonso-Blanco C, Jackson JP, Kakutani T (2000) The late flowering phenotype of fwa mutants is caused by gain-of-function epigenetic alleles of a homeodomain gene. Mol Cell 6:791–802. doi:10.1016/S1097-2765(05)00090-0
Steward N, Kusano T, Sano H (2000) Expression of ZmMET1, a gene encoding a DNA methyltransferase from maize, is associated not only with DNA replication in actively proliferating cells but also with altered DNA methylation status in cold-stressed quiescent cells. Nucleic Acids Res 28(17):3250–3259. doi:10.1093/nar/28.17.3250
Tanaka H, Masuta C, Uehara K (1997) Morphological changes and hypomethy-lation of DNA in transgenic tobacco expressing antisense NA of the S-adenosyl-l-homocysteine hydrolase gene. Plant Mol Biol 35:981–986. doi:10.1023/A:1005896711321
Tariq M, Paszkowski J (2004) DNA and histone methylation in plants. Trends Genet 20:244–251. doi:10.1016/j.tig.2004.04.005
Theißen G, Saedler H (2001) Floral quarters. Nature 409:469–471. doi:10.1038/35054172
Twyman RM (1998) Advanced molecular biology: a concise reference. BIOS Scientific Publishers, Oxford
Ulian EC, Magill JM, Magill CW, Smith RH (1996) DNA methylation and expression of NPTII in transgenic petunians and progeny. Theor Appl Genet 92:976–981. doi:10.1007/BF00224037
Vanyshin BF (2006) DNA methylation in plants. CTMI 301:67–122
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M (1995) AFLP: a new technique for DNA fingerprinting. Nucleic Acids Res 23:4407–4414. doi:10.1093/nar/23.21.4407
Wassenegger M (2000) RNA-directed DNA methylation. Plant Mol Biol 43:203–220. doi:10.1023/A:1006479327881
Wool IG (1996) Extraribosomal functions of ribosomal proteins. Trends Biochem Sci 21:164–165
Xiong LZ, Xu CG, Maroof MAS (1999) Patterns of cytosine methylation in an elite rice hybrid and its parental lines, detected by a methylation sensitive amplification polymorphism technique. Mol Gen Genet 261:439–446. doi:10.1007/s004380050986
Xu ML, Li XQ, Korban SS (2000) AFLP-based detection of DNA methylation. Plant Mol Biol Rep 18:321–330. doi:10.1007/BF02825064
Xu W, Cho H, Kadam S, Banayo ME, Anderson S, Yates RJ, Emerson MB, Evans MR (2004) A methylation-mediator complex in hormone signaling. Genes Dev 18:144–156. doi:10.1101/gad.1141704
Yi ZB, Sun Y, Niu TT (2005) Patterns of DNA cytosine methylation between hybrids and their parents in sorghum genome. Acta Agronom Sin 9:1138–1143 in Chinese
Zhan ZG, Xu C, Zhang M (2002) The form construction of PLBs of orchids. Seed 5:36–37 in Chinese
Zhu GF, Chen ML, Luo ZW, Luo SQ, Lv FB, Wang BQ (2004) Induction and propagation of hybrid protocorm like-body of crosses between Cymbidium sinense and Cymbidium hybridium. Acta Hortic Sin 31:688–690 in Chinese
Zilberman D, Henikoff S (2007) Genome-wide analysis of DNA methylation patterns. Development 134:3959–3965. doi:10.1242/dev.001131
Acknowledgments
The authors are grateful for the generous support provided by profs. Chengbin Chen and Xiulan li, and critical comments on the manuscript by Doc. Renji Pei. The work was supported by the Natural Science Foundation of Tianjin (08JCYBJC04500) and the Key Projects in Tianjin Science & Technology Pillar Program (09ZCKFNC01600).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, X., Ma, Y., Chen, F. et al. Analysis of DNA methylation patterns of PLBs derived from Cymbidium hybridium based on MSAP. Plant Cell Tiss Organ Cult 98, 67–77 (2009). https://doi.org/10.1007/s11240-009-9539-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-009-9539-5