Abstract
In this study, the anticancer properties of a series of synthesized indolyl-oxazoles drugs 1–6 and their interactions with DNA base pairs were investigated. The quantum molecular descriptors (chemical potential, hardness, and electrophilicity) for studied drugs and their complexes with DNA base pairs were calculated. The reaction enthalpies (BDE and IP) of HAT and SET-PT mechanism of anticancer action of drugs 1–6 were calculated. Results reveal that interactions of indolyl-oxazole drugs with DNA base pairs are energetically favorable and solvent increase the binding energies in comparison with gas phase. The binding energies of drugs 4–6 DNA base pairs complexes are more negative than corresponding values for drugs 1–3. The obtained binding energy and reaction enthalpy (BDE and IP) trends for drugs 1–6 confirm pervious experimental anticancer activity trends. IC50 scale has been used as a benchmark for measuring the anticancer activity. Based on theoretical and published experimental scales, drugs 4, 5, and 6 have higher anticancer activity among drugs 1–6. Finally, according to the obtained results, drugs 7–10 can consider as novel drugs with higher anticancer activity than drug 1. Results show that binding energies of drugs 7–10 with DNA base pairs were more negative than corresponding values for drugs 1–6. The BDE and IP values of drugs 4–10 were lower than corresponding values of drug 1. For drugs 1–10, results indicated that the SET-PT and HAT mechanisms represent the thermodynamically preferred mechanism in solvent and gas phase, respectively.


Similar content being viewed by others
Abbreviations
- DFT:
-
Density functional theory
- PW91:
-
Perdew–Wang 91
- COSMO:
-
Conductor like screening model
- B3LYP:
-
Becke, 3-parameter, Lee–Yang–Parr
- DNA:
-
Deoxyribonucleic acid
- WC:
-
Watson–Crick
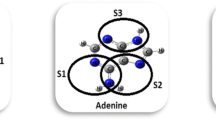
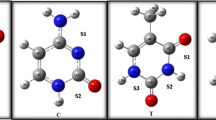
- A:
-
Adenine
- T:
-
Thymine
- G:
-
Guanine
- C:
-
Cytosine
- HB:
-
Hydrogen bond
- E b :
-
Binding energy
- BDE:
-
Bond dissociation enthalpy
- IP:
-
Ionization potential
- SET-PT:
-
Single electron transfer followed by proton transfer
- HAT:
-
Hydrogen atom transfer
- µ :
-
Chemical potential
- η :
-
Hardness
- ω :
-
Electrophilicity
- HOMO:
-
Highest occupied molecular orbital
- LUMO:
-
Lowest unoccupied molecular orbital
References
Wipf P (1995) Chem Rev 95:2115–2134
Lewis JR (1995) Nat Prod Res 12:135–163
Naik SR, Harindran J, Varde AB (2001) J Biotechnol 88:1–10
Kumar D, Sundaree S, Patel G, Rao VS (2008) Tetrahedron Lett 49:867–869
Nishida A, Fuwa M, Naruto S, Sugano Y, Saito H, Nakagawa M (2000) Tetrahedron Lett 41:4791–4794
Roy S, Haque S, Gribble GW (2006) Synthesis 3948–3954
Pettit GR, Knight JC, Herald DL, Davenport R, Pettit RK, Tucker BE, Schmidt JM (2002) J Nat Prod 65:1793–1797
Moody CJ, Roffey JRA, Stephens MA, Stratford IJ (1997) Anticancer Drugs 8(489–4):99
Glawischnig E (2007) Phytochemistry 68:401–406
Oka H, Yoshinari T, Murai T, Kawamura K, Satoh F, Funaishi K, Okura A, Suda H, Okanishi M, Shizuri Y (1991) J Antibiot 44:486–491
Garey D, Ramirez M, Gonzales S, Wertsching A, Tith S, Keefe K, Pen MR (1996) J Org Chem 61(4853–4):856
Matsunaga S, Fujita S, Sakata H, Fusetani N (1991) Tetrahedron 47:2999–3006
Iwasaki S, Kobayashi H, Furukawa J, Okuda S, Sato Z, Matsuda I, Noda T (1984) J Antibiot 37:354–362
Yoo SK (1992) Tetrahedron Lett 33:2159–2162
Connell R, Scavo F, Helquist P, Akermark B (1986) Tetrahedron Lett 27:5559–5562
Friedman BS, Sparks M, Adams R (1937) J Am Chem Soc 59:2262–2264
Zhao Z, Scarlato GR, Armstrong RW (1991) Tetrahedron Lett 32:1609–1612
Chudasama V, Wilden JD (2008) Chem Commun 32:3768–3770
Kumar D, Sundaree S, Johnson EO, Shah K (2009) Bioorg Med Chem Lett 19(4):492–494
Koser GF (2001) Aldrichimica Acta 34:89–90
Moriarty RM, Vaid RK, Koser GF (1990) Synlett 1:365–383
Tanaka K, Toda F (2000) Chem Rev 100:1025–1074
Kumar D, Kumar NM, Sundaree S, Johnson EO, Shah K (2010) Eur J Med Chem 45:1244–1249
Wright JS, Johnson ER, Dilabio GA (2001) J Am Chem Soc 123:1173–1183
Vafiadis AP, Bakalbassis EG (2005) Chem Phys 316:195–204
Musialik M, Litwinienko G (2005) Org Lett 7:4951–4954
Zhang HY, Ji HF (2003) J Mol Struct 663:167–174
Becke AD (1993) Chem Phys 98:5648–5662
Becke AD (1988) Phys Rev A 38:3098–3100
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Davidson ER, Feller D (1986) Chem Rev 86:681–696
Hehre WJ, Radom L, Schleyer PVR, Pople JA (1986) Ab initio molecular orbital theory. Wiley, New York
Perdew JP (1986) Phys Rev B 33:8822–8824
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899–926
Weinhold F, Carpenter J (1989) Springer, New York, pp 227–236
Klamt A, Jonas V, Burger T, Lohrenz JCW (1998) J Phys Chem A 102:5074–5085
Klamt A, Eckert F (2000) Fluid Phase Equilib 172:43–72
Mehler C, Klamt A, Peukert W (2002) AIChE J 48:1093–1099
Klamt A, Eckert F, Hornig M (2001) J Comput Aided Mol Des 15:355–365
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03. Gaussian Inc, Pittsburgh PA
Watson JD, Crick FHC (1953) Nature 171:737–738
Brenner S, Jacob F, Meselson M (1961) Nature 190:576–581
Guerra CF, Bickelhaupt FM, Snijders JG, Baerends EJ (1999) Chem A Eur J 5:3581–3594
Hobza P, Sponer J (1999) Chem Rev 99:3247–3276
Hobza P, Zahradnik R, Mueller-Dethlefs K (2006) Chem Commun 71:443–531
Shishkin OV, Sponer J, Hobza P (1999) J Mol Struct 477:15–21
Meng F, Liu C, Xu W (2003) Chem Phys Lett 373:72–78
Kawahara SI, Kobori A, Sekine M, Uchimaru T (2001) J Phys Chem A 105:10596–10601
Fonseca Guerra C, Bickelhaupt FM, Snijders JG, Baerends EJ (2000) J Am Chem Soc 122:4117–4128
Destexhe A, Smets J, Adamowicz L, Maes G (1994) J Phys Chem 98:1506–1514
Gould IR, Kollman PA (1994) J Am Chem Soc 116:2493–2499
Kyogoku Y, Lord RC, Rich A (1967) Proc Natl Acad Sci USA 57:250–257
Kyogoku Y, Lord RC, Rich A (1969) Biochem Biophys Acta 179:10–17
Iwahashi H, Kyogoku Y (1977) J Am Chem Soc 99:7761–7765
Newmark RA, Cantor CR (1968) J Am Chem Soc 90:5010–5017
Petersen SB, Led JJ (1981) J Am Chem Soc 103:5308–5313
Pullman B, Claverie P, Caillet J (1996) Proc Natl Acad Sci USA 55:904–912
Kudritskaya ZG, Danilov VI (1976) J Theor Biol 59:303–318
Hobza P, Sandorfy C (1987) J Am Chem Soc 109:1302–1307
Aida M (1988) J Comput Chem 9:362–368
Danilov VI, Tolokh IS, Poltev VI, Malenkov GG (1984) FEBS Lett 167:245–248
Hobza P, Sponer J, Polasek M (1995) J Am Chem Soc 117:792–798
Sponer J, Hobza P (2000) J Phys Chem A 104:4592–4597
Gilli G, Bellucci F, Ferretti V, Bertolasi V (1989) J Am Chem Soc 111:1023–1028
Guerra CF, Bickelhaupt FM, Snijders JG, Baerends EJ (1999) Chem Eur J 5:3581–3594
Meyer M, Suhnel J (1997) J Biomol Struct Dyn 15:619–624
Alkorta I, Elguero J (1998) Chem Soc Rev 27:163–170
Emsley J (1980) Chem Soc Rev 9:91–124
Kaplan IG (1986) Theory of molecular interactions. Elsevier, Amsterdam
Desiraju GR (2002) Acc Chem Res 35:565–573
Chattaraj PK, Sengupta S (1996) J Phys Chem 100:16126–16130
Aoiz FJ, Friedrich B, Herrero VJ, Sáez Rábanos V, Verdasco JE (1998) Chem Phys Lett 289:132–140
Karlberg GS, Rossmeisl J, Norskov JK (2007) Phys Chem Chem Phys 9:5158–5161
Parthasarathi R, Subramanian V, Chattaraj PK (2003) Chem Phys Lett 382:48–56
Kramer KH, Bernstein RB (1964) J Chem Phys 40:200–203
Brooks PR, Jones ME (1966) J Chem Phys 45:3449–3450
Chermette H (1999) J Comput Chem 20:129–154
Parr RG, Yang W (1989) Density-functional theory of atoms and molecules. Oxford University Press, Oxford
Geerlings P, Proft FD, Langenaeker W (2003) Chem Rev 103:1793–1873
Roy RK, Saha S (2010) Prog Chem Sect C 106:118–162
Parr RG, Donnelly RA, Levy M, Palke WE (1978) J Chem Phys 68:3801–3808
Parr RG, Pearson RG (1983) J Am Chem Soc 105:7512–7516
Parr RG, Szentpaly LV, Liu S (1999) J Am Chem Soc 121:1922–1924
Chattaraj PK, Sarkar U, Roy DR (2006) Chem Rev 106:2065–2091
Kanvah S, Schuster GB (2010) Supplementary material (ESI) for organic and biomolecular chemistry. The Royal Society of Chemistry
Kanvah S, Schuster GB (2006) Pure Appl Chem 78:2297–2304
Kanvah S, Schuster GB (2005) Nucleic Acids Res 33:5133–5138
Parthasarathi R, Subramanian V, Roy DR, Chattaraj PK (2004) Bioorg Med Chem 12:5533–5543
Mineura K, Fushimi S, Itoh Y, Kowada M (1987) Cell Biol Int Rep 11:797–802
Bizarro MM, Cabral BJC, dos Santos RMB, Simons JAM (1999) Pure Appl Chem 71:1249–1256
Parker VD (1992) J Am Chem Soc 114:7458–7462
Wilhelm E, Battino R, Wilcock RJ (1977) Chem Rev 77:219–262
Rimarcik J, Lukes V, Klein E, Ilcin M (2010) J Mol Struct (Theochem) 952:25–30
Brinck T, Haeberline M, Jonsson M (1997) J Am Chem Soc 119:4239–4244
Tomasi J, Persico M (1997) Chem Rev 94:2027–2094
Barone V, Cossi M, Tomasi J (1997) Chem Phys 107:3210–3221
Najafi M, Nazarparvar E, Haghighi Mood K, Zahedi M, Klein E (2011) Comput Theor Chem 965:114–122
Najafi M, Zahedi M, Klein E (2011) Comput Theor Chem 978:16–28
Najafi M, Haghighi Mood K, Zahedi M, Klein E (2011) Comput Theor Chem 969:1–12
Tomasi J, Mennucci B, Cammi R (2005) Chem Rev 105:2999–3093
Cramer CJ, Truhlar DG (1999) Chem Rev 99:2161–2200
Boes ES, Livotto PR, Stassen H (2006) Chem Phys 33:142–158
Klein E, Lukes V (2006) J Mol Struct: Theochem 767:43–50
Klein E, Lukes V, Cibulkova Z, Polovkova J (2006) J Mol Struct 758:149–159
Najafi M, Farmanzadeh D, Zahedi M, Klein E (2013) Acta Chim Slov 60:43–55
Sadasivam K, Kumaresan R (2011) Comput Theor Chem 963:227–235
Calliste CA, Kozlowski D, Duroux JL, Champavier Y, Chulia AJ, Trouillas P (2010) Food Chem 118:489–496
Fifen JJ, Nsangou M, Dhaouadi Z, Motapon O, Jaidane N (2011) Comput Theor Chem 966:232–243
Trouillas P, Marsal P, Svobodova A, lova JV, Gazak R, Hrbac J, Sedmera P, Kren V, Lazzaroni R, Duroux JL, Walterova D (2008) J Phys Chem A 112:1054–1063
Stepanic V, Troselj KG, Lucic B, Markovic Z, Amic D (2013) Food Chem 141:1562–1570
Leopoldini M, Marino T, Russo N, Toscano M (2004) J Phys Chem A 108:4916–4922
Nam PC, Nguyen MT, Chandra AK (2006) J Phys Chem A 110:10904–10911
Mohajeri A, Asemani SS (2009) J Mol Struct 930:15–20
Klein E, Lukes V (2006) Chem Phys 330:515–525
Zhao F, Liu ZQ, Wu D (2008) Chem Phys Lipids 151:77–84
Bizarro MM, Cabral BJC, dos Santos RMB, Simons JAM (1999) Pure Appl Chem 71:1249–1256
Acknowledgments
We gratefully acknowledge the support of the University of Mazandaran for research facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farmanzadeh, D., Najafi, M. Theoretical study of anticancer properties of indolyl-oxazole drugs and their interactions with DNA base pairs in gas phase and solvent. Struct Chem 26, 831–844 (2015). https://doi.org/10.1007/s11224-014-0546-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-014-0546-8