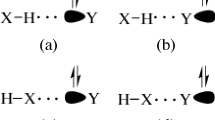Abstract
The hydrogen-bonding ability of five-membered heteroaromatic molecules containing one chalcogen and two heteroatoms with nitrogen in addition to chalcogen, respectively, have been analyzed using density functional and molecular orbital methods through adduct formation with water. The stabilization energies for all the adducts are established at B3LYP/6-31+G* and MP2/6-31+G* levels after correcting for the basis set superposition error by using the counterpoise method and also corrected for zero-point vibrational energies. A natural bond orbital analysis at B3LYP/6-31+G* level and natural energy decomposition analysis at HF/6-31+G* using MP2/6-31+G* geometries have been carried out to understand the nature of hydrogen-bonding interaction in monohydrated heterocyclic adducts. Nucleus-independent chemical shift have been evaluated to understand the correlation between hydrogen bond formation and aromaticity.



Similar content being viewed by others
References
Solimannejad M, Massahi S, Scheiner S (2009) J. Mol Struct (Theochem) 913:50
Oliveira BG, RCMUde Araújo, Ramos MN (2009) J. Mol Struct (Theochem) 908:79
Bandyopadhyay I, Lee HM, Kim KS (2005) J Phys Chem A 109:1720
Sobczyk L, Grabwski SJ, Krygowski TM (2005) Chem Rev 105:3513
Parthasarathi R, Subramanian V, Sathyamurthy N (2006) J Phys Chem A 110:3349
Melandri S, Sanz ME, Caminati W, Favero PG, Kisiel Z (1998) J Am Chem Soc 120:11504
Kun Y, LingLing LÜ, YanZhi L (2008) Chin Sci Bull 53:1315
Schwöbel J, Ebert R, Kühne R, Schüürmann G (2009) J Comput Chem 30:1454
Lopes PEM, Lamoureux G, Mackerell AD (2009) J Comput Chem 30:1821
Steiner T (2002) Angew Chem Int Ed 41:48
Zhao Y, Tishchenko O, Truhlar DG (2005) J Phys Chem B 109:19046
Schleyer PVR (2005) Chem Rev 105:3433
Raimondi M, Calderoni G, Famulari A, Raimondi L, Cozzi F (2003) J Phys Chem A 107:772
Mohan N, Vijayalakshmi KP, Koga N, Suresh CH (2010) J Comput Chem 31:2874
Imai YN, Inoue Y, Nakanishi I, Kitaura K (2009) J Comput Chem 30:2267
Samanta U, Chakrabarti P, Chandrasekhar J (1998) J Phys Chem A 102:8964
Huang D-M, Wang Y-B (2004) J Phys Chem A 108:11375
Kaur D, Khanna S (2011) Comput Theor Chem 963:71
Li S, Cooper VR, Thonhauser T, Puzder A, Langreth DC (2008) J Phys Chem A 112:9031
Carles S, Lecomte F, Schermann JP, Desfrancois C (2004) J Phys Chem A 104:10662
Sarkhel S, Rich A, Egli M (2003) J Am Chem Soc 125:8998
Hammed S, Akhtar T (2011) Curr Org Chem 15:694
Lipshutz BH (1986) Chem Rev 86:795
Marino G (1971) Adv Heterocycl Chem 13:235
Streitwieser A (1994) Organische Chemie. VCH, Basel
Modelli A, Burrow PD (2004) J Phys Chem A 108:5721
Hiraoka K, Takimoto H, Yamabe S (1987) J Am Chem Soc 109:7346
Frisch MJ, Trucks GW, Schlegal HB, Scuseria GE, Robb MA, Cheeseman JR, Zarkrzewski VG, Montgomery JA, Stratman RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennuci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Raghvachari K, Foresman JB, Oritz JV, Baboul AG, Stefanov BB, Liu G, Liashenko A, Piskorz P, Kamaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (2001) Guassian Inc., Pittsburgh
Hehre WJ, Radom L, Schleyer PVR, Pople JA (1986) Ab initio molecular orbital theory. Wiley, New York
Foresman JB, Frisch E (1996) Exploring chemistry with electronic structure methods. Guassian Inc, Pittsburgh
Boys SF, Bernardi F (1970) Mol Phys 19:553
Merrick JP, Moran D, Radom L (2007) J Phys Chem A 111:11683
Foster JP, Weinhold F (1980) J Am Chem Soc 102:7211
Reed AE, Weinhold F, Curtiss LA (1988) Chem Rev 88:899
Chen Z, Wannere CS, Corminboeuf C, Puchta R, Schleyer PvR (2005) Chem Rev 105:3842
Schleyer PvR, Maerker C, Dransfeld A, Jiao H, Hommes NjvE (1996) J Am Chem Soc 118:6317
Glendening ED, Streitwieser A (1994) J Chem Phys 100:2900
Glendening ED (1996) J Am Chem Soc 118:2473
Schenter GK, Glendening ED (1996) J Phys Chem 100:17152
Glendening ED, Badenhoop JK, Reed AE, Carpenter JE, Bohmann JA, Morales CM, Weinhold F (2001) Theoretical Chemistry Institute. University of Wisconsin, Madison
Weinhold F, Landis CR (2001) Chem Educ Res Pract 2:91
Schmidt MW, Baldridge KK, Boatz JA, Elbert ST, Gordon MS, Jensen JH, Koseki S, Matsunaga N, Nguyen KA, Su SJ, Windus TL, Dupuis M, Montgomery JA (1993) J Comput Chem 14:1347
Desiraju GR (2002) Acc Chem Res 35:565
Hideaki U, Morokuma K (1977) J Am Chem Soc 99:1316
Kaur D, Khanna S, Kaur RP (2010) J Mol Struct (Theochem) 949:14
Böhm H-J, Klebe G, Brode S, Hesse U (1996) Chem Eur J 2:1509
Nobeli I, Price SL, Lommerse JPM, Taylor R (1997) J Comput Chem 18:2060
Acknowledgments
The authors are highly thankful to UGC for financial assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kaur, D., Khanna, S. Theoretical study on the hydrogen bonding of five-membered heteroaromatics with water. Struct Chem 23, 755–764 (2012). https://doi.org/10.1007/s11224-011-9917-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-011-9917-6