Abstract
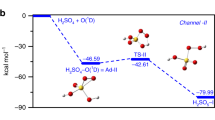
The reaction pathways of acid rain formation from reaction of sulfur dioxide vapor and water vapor on the singlet potential energy surface have been investigated theoretically. The calculated results show that the reactants are initially associated with the adduct SO2–H2O through a barrier less process. Subsequently, via a variety of transformations of isomer SO2–H2O, three kinds of products H2SO3, SO3 + H2, and H2O2 + 3SO are obtained. The cleavage and formation of the chemical bonds in the reaction pathways have been discussed using the structural parameters. Also, by means of the transition states and their connected intermediates or products at the CCSD(T)//B3LYP level, mechanism of H2O + SO2 reaction on the singlet potential energy surface are plotted. The calculation results show that the most suitable reaction pathways are the formation of H2SO3. Finally, the rate constants have been calculated only for these suitable pathways by the RRKM and TST theories at temperature range of 250–2500 K.




Similar content being viewed by others
References
Voegele AF, Loerting T, Tautermann CS, Hallbrucker A, Mayer E, Liedl KR (2004) Icarus 169:242
Li X, Meng L, Zheng S (2007) J Mol Struct 847:52
Krotkov NA, Carn SA, Krueger AJ, Bhartia PK, Yang K (2006) IEEE Trans Geosci Remote Sens 44:1259
Goodarzi M, Vahedpour M, Nazari F (2010) J Mol Struct 945:45
Goodarzi M, Vahedpour M, Nazari F (2010) J Mol Struct 944:110
Becke AD (1992) J Chem Phys 97:9173
Frisch MJ, Head-Gordon M, Pople JA (1990) Chem Phys Lett 166:281
Gonzalez C, Schlegel HB (1989) J Chem Phys 90:2154
Gonzalez C, Schlegel HB (1990) J Phys Chem 94:5523
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03, Revision B.03. Gaussian, Inc., Pittsburgh, PA
Biegler-Knìng F (2000) AIM2000 ver. 1.0. University of Applied Science, Bielefeld, Germany
Frisch MJ, Head-Gordon M, Pople JA (1990) Chem Phys Lett 166:275
Bader RFW (1990) Atoms in molecules—a quantum theory. Oxford University Press, Oxford
Bader RFW (1991) Chem Rev 91:893
Robinson PJ, Holbrook KA (1972) Unimolecular reactions. Wiley Inter Science, New York
Forst W (1973) Theory of unimolecular reactions. Academic Press, New York
Eyring H (1935) J Chem Phys 3:1079
Evans MG, Polyani M (1935) Trans Faraday Soc 31:875
Miyoshi A (2010) Steady-state unimolecular master-equation solver (SSUMES). University of Tokyo, Tokyo
Miyoshi A (2010) Gaussian post processor (GPOP). University of Tokyo, Tokyo
Acknowledgment
This project was supported by the Zanjan University and the authors would like to thank for financial supporting.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vahedpour, M., Zolfaghari, F. Mechanistic study on the atmospheric formation of acid rain base on the sulfur dioxide. Struct Chem 22, 1331–1338 (2011). https://doi.org/10.1007/s11224-011-9823-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-011-9823-y