Abstract
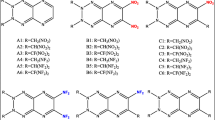
A number of new energetic compounds were synthesized based on a combination of pyrazole, furazan, and 1,2,4-oxadiazole. Density, temperature of decomposition, and mechanical sensitivity of the compounds obtained were experimentally determined. Their detonation parameters were calculated.
Similar content being viewed by others
References
J. P. Agrawal, D. L. Hodgson, Organic Chemistry of Explosives, John Wiley & Sons, 2007, pp. 332–334.
T. M. Klapotke, Chemistry of High-Energy Materials, 2nd ed., Walter de Gruyter, Berlin, 2012.
S. G. Zlotin, A. M. Churakov, M. P. Egorov, L. L. Fershtat, M. S. Klenov, I. V. Kuchurov, N. N. Makhova, G. A. Smirnov, Yu. V. Tomilov, V. A. Tartakovsky, Mendeleev Commun., 2021, 31, 731; DOI: https://doi.org/10.1016/j.mencom.2021.11.001.
A. B. Sheremetev, Chem. Heterocycl. Compd., 2017, 53, 629.
M. Benz, T. M. Klapötke, J. Stierstorfer, Z. Anorg. Allg. Chem., 2020, 646, 1380; DOI: https://doi.org/10.1002/zaac.202000123.
J. Cai, Ch. Xie, J. Xiong, J. Zhang, P. Yin, S. Pang, Chem. Eng. J., 2022, 433, 134480; DOI: https://doi.org/10.1016/j.cej.2021.134480.
T. Yan, Ch. Yang, J. Ma, G. Cheng, H. Yang, Chem. Eng. J., 2022, 428, 131400; DOI: https://doi.org/10.1016/j.cej.2021.131400.
T. Yan, G. Cheng, H. Yang, New J. Chem., 2020, 44, 6643; DOI: https://doi.org/10.1039/D0NJ00518E.
M. Xu, G. Cheng, H. Xiong, B. Wang, X. Ju, H. Yang, New J. Chem., 2019, 43, 11157; DOI: https://doi.org/10.1039/C9NJ01445D.
A. A. Konnov, I. M. Dubrovin, M. S. Klenov, O. V. Anikin, A. M. Churakov, Yu. A. Strelenko, A. N. Pivkina, V. A. Tartakovsky, Russ. Chem. Bull., 2021, 70, 2189; DOI: https://doi.org/10.1007/s11172-021-3331-z.
X. Zheng, T. Yan, L. Qian, H. Yang, G. Cheng, Def. Technol., 2022; DOI: https://doi.org/10.1016/j.dt.2022.03.003.
T. E. Khoranyan, T. K. Shkineva, I. A. Vatsadze, A. Kh. Shakhnes, N. V. Muravyev, A. B. Sheremetev, I. L. Dalinger, Chem. Heterocycl. Compd., 2022, 58, 37; DOI: https://doi.org/10.1007/s10593-022-03054-1.
L. L. Fershtat, N. N. Makhova, ChemPlusChem., 2019, 85, 13; DOI: https://doi.org/10.1002/cplu.201900542.
S. G. Zlotin, I. L. Dalinger, N. N. Makhova, V. A. Tartakovsky, Russ. Chem. Rev., 2020, 89, 1; DOI: https://doi.org/10.1070/RCR4908.
I. L. Dalinger, A. V. Kormanov, K. Yu. Suponitsky, N. V. Muravyev, A. B. Sheremetev, Chem. — Asian J., 2018, 13, 1165; DOI: https://doi.org/10.1002/asia.201800214.
I. L. Dalinger, K. Yu. Suponitsky, A. N. Pivkina, A. B. Sheremetev, Propellants, Explos., Pyrotech., 2016, 41, 789; DOI: https://doi.org/10.1002/prep.201600050.
I. L. Dalinger, K. Yu. Suponitsky, T. K. Shkineva, D. B. Lempert, A. B. Sheremetev, J. Mater. Chem. A, 2018, 6, 14780; DOI: https://doi.org/10.1039/C8TA05179H.
I. L. Dalinger, T. K. Shkineva, I. A. Vatsadze, A. V. Kormanov, A. M. Kozeev, K. Yu. Suponitsky, A. N. Pivkina, A. B. Sheremetev, FirePhysChem, 2021, 1, 83; DOI: https://doi.org/10.1016/j.fpc.2021.04.005.
I. L. Dalinger, A. Kh. Shakhnes, K. A. Monogarov, K. Yu. Suponitsky, A. B. Sheremetev, Mendeleev Commun., 2015, 25, 429; DOI: https://doi.org/10.1016/j.mencom.2015.11.010.
I. L. Dalinger, I. A. Vatsadze, T. K. Shkineva, A. V. Kormanov, M. I. Struchkova, K. Yu. Suponitsky, A. A. Bragin, K. A. Monogarov, V. P. Sinditskii, A. B. Sheremetev, Chem. — Asian J., 2015, 10, 1987; DOI: https://doi.org/10.1002/asia.201500533.
T. K. Shkineva, I. A. Vatsadze, T. E. Khoranyan, D. L. Lipilin, A. N. Pivkina, I. L. Dalinger, Chem. Heterocycl. Compd., 2021, 57, 828; DOI: https://doi.org/10.1007/s10593-021-02987-3.
Q. Wang, Y. Shao, M. Lu, Cryst. Growth Des., 2019, 19, 839; DOI: https://doi.org/10.1021/acs.cgd.8b01404.
D. A. Gulyaev, M. S. Klenov, A. M. Churakov, Y. A. Strelenko, I. V. Fedyanin, D. B. Lempert, E. K. Kosareva, T. S. Kon’kova, Y. N. Matyushin, V. A. Tartakovsky, RSC Adv., 2021, 11, 24013; DOI: https://doi.org/10.1039/D1RA03919A.
R. F. W. Bader, Atoms in Molecules. A Quantum Theory, Clarendon Press, Oxford, 1990.
T. A. Keith, AIMAll, Version 14.11.23, TK Gristmill Software, USA (KS); http://aim.tkgristmill.com.
E. Espinosa, I. Alkorta, I. Rozas, J. Elguero, E. Molins, Chem. Phys. Lett., 2001, 336, 457; DOI: https://doi.org/10.1016/S0009-2614(01)00178-6.
E. Espinosa, E. Molins, C. Lecomte, Chem. Phys. Lett., 1998, 285, 170; DOI: https://doi.org/10.1016/S0009-2614(98)00036-0.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery, K. N. Jr. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, J. A. Pople, Gaussian 03, Revision E.01, Gaussian, Inc., Wallingford, 2004.
A. B. Sheremetev, N. S. Aleksandrova, S. S. Semyakin, K. Yu. Suponitsky, D. B. Lempert, Chem. — Asian J., 2019, 14, 4255; DOI: https://doi.org/10.1002/asia.201901280.
N. V. Palysaeva, A. G. Gladyshkin, I. A. Vatsadze, K. Yu. Suponitsky, D. E. Dmitriev, A. B. Sheremetev, Org. Chem. Front., 2019, 6, 249; DOI: https://doi.org/10.1039/C8QO01173G.
K. Yu. Suponitsky, A. F. Smol’yakov, I. V. Ananyev, A. V. Khakhalev, A. A. Gidaspov, A. B. Sheremetev, ChemistrySelect, 2020, 5, 14543; DOI: https://doi.org/10.1002/slct.202004020.
Y. V. Nelyubina, I. V. Glukhov, M. Yu. Antipin, K. A. Lyssenko, Chem. Commun., 2010, 46, 3469; DOI: https://doi.org/10.1039/B927429D.
A. A. Gidaspov, V. A. Zalomlenkov, V. V. Bakharev, V. E. Parfenov, E. V. Yurtaev, M. I. Struchkova, N. V. Palysaeva, K. Yu. Suponitsky, D. B. Lempert, A. B. Sheremetev, RSC Adv., 2016, 6, 34921; DOI: https://doi.org/10.1039/C6RA05826D.
I. V. Fedyanin, A. I. Samigullina, I. A. Krutov, E. L. Gavrilova, D. V. Zakharychev, Crystals, 2021, 11, 1126; DOI: https://doi.org/10.3390/cryst11091126.
N. V. Muravyev, D. B. Meerov, K. A. Monogarov, I. N. Melnikov, E. K. Kosareva, L. L. Fershtat, A. B. Sheremetev, I. L. Dalinger, I. V. Fomenkov, A. N. Pivkina, Chem. Eng. J., 2021, 421, 129804, DOI: https://doi.org/10.1016/j.cej.2021.129804.
M. S. Westwell, M. S. Searle, D. L. Wales, D. H. Williams, J. Am. Chem. Soc., 1995, 117, 5013; DOI: https://doi.org/10.1021/ja00123a001.
N. M. Baraboshkin, A.-M. Stratulat, T. S. Pivina, Russ. Chem. Bull., 2021, 70, 1893; DOI: https://doi.org/10.1007/s11172-021-3293-1.
N. V. Muravyev, D. Wozniak, D. Piercey, J. Mater. Chem. A, 2022; DOI: https://doi.org/10.1039/D2TA01339H.
H. Li, L. Zhang, N. Petrutik, K. Wang, Q. Ma, D. Shem-Tov, F. Zhao, M. Gozin, ACS Cent. Sci., 2020, 6, 54; DOI: https://doi.org/10.1021/acscentsci.9b01096.
R. Bu, Y. Xiong, X. Wei, H. Li, Ch. Zhang, Cryst. Growth Des., 2019, 19, 5981; DOI: https://doi.org/10.1021/acs.cgd.9b00853.
Ph. F. Pagoria, M. Zhang, N. B. Zuckerman, A. J. DeHope, D. A. Parrish, Chem. Heterocycl. Compd., 2017, 53, 760; DOI: https://doi.org/10.1007/s10593-017-2122-9.
K. Cernovská, M. Kemter, H. C. Gallmeier, P. Rzepecki, T. Schrader, B. König, Org. Biomol. Chem., 2004, 2, 1603; DOI: https://doi.org/10.1039/B401968G.
T. Semeraro, C. Mugnaini, F. Manetti, S. Pasquini, F. Corelli, Tetrahedron, 2008, 64, 11249; DOI: https://doi.org/10.1016/j.tet.2008.09.030.
APEX2 and SAINT, Bruker AXS Inc., USA (WT), 2014.
G. M. Sheldrick, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the Academician of the Russian Academy of Sciences V. A. Tartakovsky on the occasion of his 90th birthday.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Published in Russian in Izyestiya Akademii Nauk. Seriya Khimicheskaya, No. 8, pp. 1750–1759, August, 2022.
Rights and permissions
About this article
Cite this article
Khoranyan, T.E., Serushkina, O.V., Vatsadze, I.A. et al. 3-(4-R-3-Furazanyl)-5-nitropyrazolyl-1,2,4-oxadiazoles as a new class of energy rich ensembles. Russ Chem Bull 71, 1750–1759 (2022). https://doi.org/10.1007/s11172-022-3586-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3586-z