Abstract
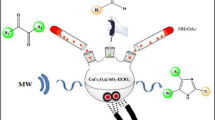
In this work, we report the synthesis and identification of ZnFe2O4@SiO2–SO3H as a novel and green nanomagnetic solid acid catalyst, which is containing the sulfuric acid catalytic sites on the surface of ZnFe2O4 magnetic nanoparticles as the catalytic support. The as-synthesized ZnFe2O4@SiO2–SO3H MNPs were characterized with accuracy by different physicochemical methods; including, X-ray powder diffraction, energy-dispersive spectroscopy, Scanning electron microscopy, Fourier transform infrared, X-ray elemental mapping, Thermogravimetric analysis, and Vibrating Sample Magnetometer analyses. The catalytic activity of this nanomagnetic material is considered for the synthesis of the diversely substituted tetrahydrobenzo[b]pyran and DHQZ (2,3-dihydroquinazolin-4(1H)-ones) in ethanol solvent, which affording excellent yields (86–97%). The heterogeneous nature of the catalyst was confirmed via the hot filtration experiment. Furthermore, the activity of the recycled nanocatalyst was examined for at least five cycles with a negligible loss of its activity (97–92%). Also, the amount of sulfamic acid function in the ZnFe2O4@SiO2–SO3H was calculated from back titration.














Similar content being viewed by others
References
A. Anders, A. Vakula, S. Tarapov, A. Belous, Telecommun. Radio Eng. (English Transl. Elektrosvyaz Radiotekhnika) 75, 1849 (2016)
S.M. Pormazar, A. Dalvand, Int. J. Environ. Anal. Chem. 00, 1 (2020)
E. Leal, J. Dantas, P.T.A. dos Santos, S.M. de C.M. Bicalho, R.H.G.A. Kiminami, M.R. da Silva, A. C.F. de M. Costa, Appl. Surf. Sci. 455, 635 (2018)
P. Koohi, A. Rahbar-kelishami, H. Shayesteh, Environ. Technol. Innov. 23, 101559 (2021)
D. Pilnaj, P. Kuráň, M. Št’astný, V. Pilařová, P. Janoš, M. Kormunda, J. Tokarský, Environ. Technol. Innov. 24, 101905 (2021)
Z. Na, R. Yao, Q. Yan, X. Wang, X. Sun, X. Wang, Electrochim. Acta. 367, 137464 (2021)
Z. Hossaini, N. Tabarsaei, S. Khandan, P. Valipour, M. Ghorchibeigi, Appl. Organomet. Chem. 35, e6114 (2021)
M. Rafieezadeh, A.H. Kianfar, Environ. Technol. Innov. 23, 101756 (2021)
H. Veisi, A. Zohrabi, S.A. Kamangar, B. Karmakar, S.G. Saremi, K. Varmira, M. Hamelian, J. Organomet. Chem. 951, 122005 (2021)
S. Tang, M. Zhao, D. Yuan, X. Li, Z. Wang, X. Zhang, T. Jiao, J. Ke, Chemosphere 268, 129315 (2021)
Q. Zhai, W. Zhai, B. Gao, Y. Shi, X. Cheng, Colloids Surf. A Physicochem. Eng. Asp. 625, 126871 (2021)
B.N. Sahu, S.C. Sahoo, N. Venkataramani, S. Prasad, R. Krishnan, J. Magn. Magn. Mater. 523, 167629 (2021)
T. Tamoradi, S.M. Mousavi, M. Mohammadi, New J. Chem. 44, 3012 (2020)
R. Elshypany, H. Selim, K. Zakaria, A.H. Moustafa, S.A. Sadeek, S.I. Sharaa, P. Raynaud, A.A. Nada, Environ. Technol. Innov. 23, 101710 (2021)
M. Nikoorazm, A. Ghorbani-Choghamarani, M. Khanmoradi, Appl. Organomet. Chem. 30, 705 (2016)
Z. Moradi, A. Ghorbani-Choghamarani, Sci. Rep. 11, 23967 (2021)
B. Sharma, J.-S. Sung, A.A. Kadam, J.-H. Myung, Appl. Surf. Sci. 530, 147272 (2020)
L. Liu, G. Su, X. Liu, W. Dong, M. Niu, Q. Kuang, A. Tang, J. Xue, Environ. Technol. Innov. 22, 101436 (2021)
R. Pourhasan Kisomi, F. Shirini, M. Golshekan, Appl. Organomet. Chem. 35, 6212 (2021)
X. Huang, J. Xiao, Q. Yi, D. Li, C. Liu, Y. Liu, J. Environ. Manag. 308, 114613 (2022)
H. Aghavandi, A. Ghorbani-Choghamarani, J. Phys. Chem. Solids 170, 110952 (2022)
M. Malmir, M.M. Heravi, Z. Amiri, K. Kafshdarzadeh, Sci. Rep. 11, 1 (2021)
S. Chen, J. Yan, C. Wang, D. Lu, At. Spectrosc. 40, 199 (2019)
N. Muhammad, S. Nadeem, Eur. Phys. J. Plus. 132, 377 (2017)
H. Ren, C. Li, Z. Han, T. Li, X. Jin, R. Zhou, J. Chem. Technol. Biotechnol. 95, 3202 (2020)
E.A. Chavarriaga, A.A. Lopera, V. Franco, C.P. Bergmann, J. Alarcón, J. Magn. Magn. Mater. 497, 166054 (2020)
A. Ghorbani-Choghamarani, Z. Taherinia, M. Mohammadi, Environ. Technol. Innov. 24, 102050 (2021)
L.T.T. Nguyen, D.-V.N. Vo, L.T.H. Nguyen, A.T.T. Duong, H.Q. Nguyen, N.M. Chu, D.T.C. Nguyen, T. Van Tran, Environ. Technol. Innov. 25, 102130 (2022)
T. Li, Z. Wang, Z. Zhang, K. Feng, J. Liang, D. Wang, L. Zhou, Sep. Purif. Technol. 276, 119344 (2021)
I.V. Korolkov, A.V. Zibert, L.I. Lissovskaya, K. Ludzik, M. Anisovich, A.L. Kozlovskiy, A.E. Shumskaya, M. Vasilyeva, D.I. Shlimas, M. Jażdżewska, B. Marciniak, R. Kontek, D. Chudoba, M.V. Zdorovets, Int. J. Mol. Sci. 22, 8687 (2021)
A. Aschenaki, F. Ren, J. Liu, W. Zheng, Q. Song, W. Jia, J.J. Bao, Y. Li, RSC Adv. 11, 33692 (2021)
M.J. Khodabakhshi, H. Ahmad Panahi, E. Konoz, A. Feizbakhsh, S. Kimiagar, Polym. Technol. Mater. 60, 1247 (2021)
S.F. Hojati, N. MoeiniEghbali, S. Mohamadi, T. Ghorbani, Org. Prep. Proced. Int. 50, 408 (2018)
S. Chehab, Y. Merroun, T. Ghailane, R. Ghailane, S. Boukhris, B. Lakhrissi, A. Souizi, J. Iran. Chem. Soc. 18, 2665 (2021)
H. Faroughi Niya, N. Hazeri, M. Rezaie Kahkhaie, M.T. Maghsoodlou, Res. Chem. Intermed. 46, 1685 (2020)
S. Chehab, Y. Merroun, T. Ghailane, R. Ghailane, S. Boukhris, A. Souizi, Polycycl. Aromat. Compd. 1, 1 (2021)
V. Alagarsamy, K. Chitra, G. Saravanan, V.R. Solomon, M.T. Sulthana, B. Narendhar, Eur. J. Med. Chem. 151, 628 (2018)
Y. Merroun, S. Chehab, T. Ghailane, R. Ghailane, S. Boukhris, A. Hassikou, N. Habbadi, B. Lakhrissi, A. Souizi, Mediterr. J. Chem. 10, 553 (2020)
E. Ezzatzadeh, Z. Hossaini, Appl. Organomet. Chem. 34, 1 (2020)
P.M.S. Bedi, V. Kumar, M.P. Mahajan, Bioorg. Med. Chem. Lett. 14, 5211 (2004)
M. Asif, Int. J. Med. Chem. 2014, 1 (2014)
Y. Merroun, S. Chehab, T. Ghailane, S. Boukhris, R. Ghailane, N. Habbadi, A. Hassikou, B. Lakhrissi, A. Souizi, J. Turk. Chem. Soc. Sect. A Chem. 5, 303 (2018)
N. Malecki, P. Carato, B. Rigo, J. Goossens, R. Houssin, C. Bailly, J. Hénichart, Bioorg. Med. Chem. 12, 641 (2004)
F. Khanmohammadi-Sarabi, A. Ghorbani-Choghamarani, H. Aghavandi, M.A. Zolfigol, Appl. Organomet. Chem. n/a, (n.d.)
A. Nouri Parouch, N. Koukabi, E. Abdous, Res. Chem. Intermed. 46, 3295 (2020)
P. Zhang, I. Lo, D. O’Connor, S. Pehkonen, H. Cheng, D. Hou, J. Colloid Interface Sci. 508, 39 (2017)
A.S. Varpe, M.D. Deshpande, J. Sol-Gel Sci. Technol. 96, 718 (2020)
A. Alizadeh, G. Abdi, M.M. Khodaei, M. Ashokkumar, J. Amirian, RSC Adv. 7, 14876 (2017)
F. Zhang, B. Shen, W. Jiang, H. Yuan, H. Zhou, J. Nanopart. Res. 21, 269 (2019)
N. Ali, F. Ali, A. Said, T. Begum, M. Bilal, A. Rab, Z.A. Sheikh, H.M.N. Iqbal, I. Ahmad, J. Inorg. Organomet. Polym. Mater. 30, 5063 (2020)
A. Maleki, M. Niksefat, J. Rahimi, Z. Hajizadeh, BMC Chem. 13, 1 (2019)
L. Khazdooz, A. Zarei, A.R. Hajipour, N. Sheikhan, Iran. J. Catal. 1, 1 (2011)
I. Elsayed, M. Mashaly, F. Eltaweel, M.A. Jackson, E.B. Hassan, Fuel 221, 407 (2018)
S.S. Pourpanah, S.M. Habibi-Khorassani, M. Shahraki, Cuihua Xuebao/Chin. J. Catal. 36, 757 (2015)
A. Davoodnia, S. Allameh, S. Fazli, N. Tavakoli-Hoseini, Chem. Pap. 65, 714 (2011)
M. Koolivand, M. Nikoorazm, A. Ghorbani-Choghamarani, R. Azadbakht, B. Tahmasbi, Sci. Rep. 11, 24475 (2021)
A. Ghorbani-Choghamarani, H. Aghavandi, M. Mohammadi, J. Porous Mater. 28, 1167 (2021)
A. Khandebharad, S. Sarda, C. Gill, M. Soni, B. Agrawal, Res. Chem. Intermed. 42, 5779 (2016)
M. Bakherad, A. Keivanloo, E. Moradian, A.H. Amin, R. Doosti, M. Armaghan, J. Iran. Chem. Soc. 15, 2811 (2018)
A. Saha, S. Payra, A. Asatkar, A.R. Patel, S. Banerjee, Curr. Organocatal. 6, 177 (2018)
G. Kumar, N.K. Mogha, D.T. Masram, A.C.S. Appl, Nano Mater. 4, 2682 (2021)
A. Dutta, K. Damarla, A. Kumar, P.J. Saikia, D. Sarma, Tetrahedron Lett. 61, 151587 (2020)
M.A. Erfan, B. Akhlaghinia, S.S.E. Ghodsinia, ChemistrySelect 5, 2306 (2020)
T. Tamoradi, M. Ghadermazi, A. Ghorbani-Choghamarani, Catal. Lett. 148, 857 (2018)
Acknowledgements
The authors thank the research facilities of Bu-Ali Sina University, Hamedan, Iran, for financial support of this research project.
Author information
Authors and Affiliations
Contributions
AG-C: Supervision, Conceptualization, Resources, Writing—review and editing. HA: Methodology, Conceptualization, Writing—original draft.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aghavandi, H., Ghorbani-Choghamarani, A. Preparation and application of ZnFe2O4@SiO2–SO3H, as a novel heterogeneous acidic magnetic nanocatalyst for the synthesis of tetrahydrobenzo[b]pyran and 2,3-dihydroquinazolin-4(1H)-one derivative. Res Chem Intermed 49, 441–467 (2023). https://doi.org/10.1007/s11164-022-04890-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-022-04890-8