Abstract
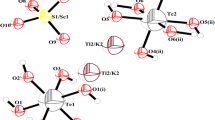
A new compound thallium ammonium sulfate selenate tellurate: Tl0.92(NH4)0.08(SO4)0.65(SeO4)0.35Te(OH)6 (TlNSSeTe) has been synthesized by slow evaporation at room temperature to specify its phase transition temperatures. X-ray diffractometry data revealed crystallization in the monoclinic system with a P21/c space group with these parameters: a = 12.336(2) Å, b = 7.2038(14) Å, c = 12.017(2) Å et β = 110.631(10)°, V = 999.4(3) Å3 and Z = 4. Three coexisting anions (TeO\( _{6}^{6 - } \), SO\( _{4}^{2 - } \) and SeO\( _{4}^{2 - } \)) are the main characteristics of these atomic arrangements in the unit cell, related by O–H…O and N–H…O bonds building up the crystal. The cations Tl+ and NH\( _{4}^{ + } \) lie at the same positions between these polyhedra. The new synthesized compound has been characterized by using the differential scanning calorimetry (DSC), thermal analysis [differential thermogravimetric analysis (TG), as well as thermodifference analysis (DTA)], FT-IR and Raman spectroscopy techniques. The DSC results demonstrated phase transitions at 384 K, 465 K and 472 K. DTA and TG proved that decomposition takes place in three steps. The vibrational spectroscopy study at room temperature unveiled not only the occurrence and the independence of anionic and cationic groups, but also the importance of the hydrogen bonds.









Similar content being viewed by others
References
J. Lin, K. Diefenbach, J.N. Cross, J.M. Babo, T.E. Albrecht-Schmitt, Inorg. Chem. 52, 13278 (2013)
J. Lin, K. Diefenbach, N. Kikugawa, R.E. Baumbach, T.E. Albrecht-Schmitt, Inorg. Chem. 53, 8555 (2014)
J. Lin, J.N. Cross, J. Diwu, N.A. Meredith, T.E. Albrecht-Schmitt, Inorg. Chem. 52, 4277 (2013)
J. Lin, J.N. Cross, J. Diwu, M.J. Polinski, E.M. Villa, T.E. Albrecht-Schmitt, Inorg. Chem. 51, 11949 (2012)
J. Lin, J.N. Cross, J. Diwu, E.M. Villa, T.E. Albrecht-Schmitt, Inorg. Chem. 51, 10083 (2012)
Y.Y. Tang, Z.Z. He, W.B. Guo, S.Y. Zhang, M. Yang, Inorg. Chem. 53, 5862 (2014)
J. Lin, P. Chai, K. Diefenbach, M. Shatruk, T.E. Albrecht-Schmitt, Chem. Mater. 26, 2187 (2014)
H. Litaiem, M. Dammak, T. Mhiri, A. Cousson, J. Alloys Compd. 396, 34 (2005)
L. Ktari, M. Dammak, A. Hadrich, A. Cousson, M. Nierlich, F. Romain, T. Mhiri, Solid State Sci. 6, 1393 (2004)
L. Ktari, M. Dammak, A. Madani, T. Mhiri, Solid State Ion. 145, 225 (2001)
A. Elferjani, S. Garcia-Granda, M. Dammak, J. Alloys Compd. 749, 448 (2018)
K. Ghorbel, H. Litaiem, L. Ktari, S. Garcia-Granda, M. Dammak, Chem. Res. Chin. Univ. 32, 902 (2016)
H. Khemakhem, Ferroelectrics 234, 47 (1999)
R. Zilber, A. Durif, M.T. Averbuch-Pouchot, Acta Cryst. B 36, 2743 (1980)
R. Zilber, A. Durif, M.T. Averbuch-Pouchot, Acta Cryst. B 37, 650 (1981)
R. Zilber, A. Durif, M.T. Averbuch-Pouchot, Acta Cryst. B 38, 1554 (1982)
M. Dammak, H. Khemakhem, T. Mhiri, A.W. Kolsi, A. Daoud, J. Alloys Compd. 280, 107 (1998)
M. Dammak, H. Khemakhem, T. Mhiri, J. Phys. Chem. Solids 62, 2069 (2001)
L. Ktari, M. Dammak, A.W. Kolsi, A. Cousson, J. Alloys Compd. 476, 54 (2009)
M. Dammak, L. Ktari, A. Cousson, T. Mhiri, J. Solid State Chem. 178, 2109 (2005)
L. Ktari, M. Dammak, T. Mhiri, J.M. Savariault, J. Solid State Chem. 16, 1 (2000)
M. Abdelhedi, M. Dammak, A.W. Kolsi, A. Cousson, Anal. Sci. X-ray Struct. Anal. Online 24, 93 (2008)
M. Abdelhedi, L. Ktari, M. Dammak, A. Cousson, A.W. Kolsi, J. Alloys Compd. 460, 147 (2008)
M. Abdelhedi, M. Dammak, A. Cousson, A.W. Kolsi, J. Alloys Compd. 398, 55 (2005)
M. Dammak, A. Hadrich, T. Mhiri, J. Alloys Compd. 428, 8 (2007)
L. Ktari, M. Dammak, T. Mhiri, A.W. Kolsi, Phys. Procedia. 2, 729 (2009)
Nonius, in: B.V. Nonius (Ed.), Kappa CCD Sever Software, Delft, The Netherlands, 1999
APEX2 version 1. 0–8, Bruker AXS, Madison, WI, 2003
D.J. Watkin, C.K. Prout, J.R. Carruthers, P.W. Betteridge, R.I. Cooper, CRYSTALS Issue 11 (Chemical Crystallography Laboratory, Oxford, UK, 2001)
K. Brandenburg, M. Berndt, DIAMOND Version 2.1.b, Crystal Impact, Gb R, (Bonn, Germany 1999)
Y.Y. Tang, W.B. Guo, S.Y. Zhang, M. Yang, Z.Z. He, Cryst. Growth Des. 14, 5206 (2014)
H. Frikha, M. Abdelhedi, M. Dammak, S. Garcia-Granda, J. Saudi Chem. Soc. 21, 324 (2017)
K. Ghorbel, H. Litaiem, L. Ktari, S. Garcia-Granda, M. Dammak, J. Mol. Struct. 1079, 225 (2015)
A. Elferjani, M. Abdelhedi, M. Dammak, A.W. Kolsi, J. Appl. Phys. A. 122, 742 (2016)
K. Ghorbel, H. Litaiem, L. Ktari, S. Garcia-Granda, M. Dammak, Ionics 22, 251 (2016)
A. Novak, Hydrogen Bonding in Solids, vol. 18 (Spring, Berlin, 1974), p. 177
M. Djemel, M. Abdelhedi, L. Ktari, M. Dammak, J. Mol. Struct. 1033, 84 (2013)
J. Faby, J. Loub, L. Feltl, J. Therm. Anal. 24, 95 (1982)
R. Ayadi, J. Lhoste, I. Ledoux-Rak, T. Mhiri, M. Boujelbene, J. Saudi Chem. Soc. 21, 869 (2017)
Y.Y. Tang, M. Cui, W.B. Guo, S.Y. Zhang, M. Yang, Z.Z. He, Cryst. Growth Des. 15, 2742 (2015)
B. Yotnoi, A. Rujiwatra, M.L.P. Reddy, D. Sarma, S. Natarajan, Cryst. Growth Des. 11, 1347 (2011)
K. Jaouadi, N. Zouari, T. Mhiri, Phase Transit. 90, 143 (2017)
N. Nouiri, K. Jaouadi, T. Mhiri, N. Zouari, Ionics 22, 1611 (2016)
Acknowledgements
This work is supported by the Ministry of Higher Education and Research of Tunisia. Authors would like to thank Pr. Kamel Maaloul from the English Language Unit at the Faculty of Sciences of Sfax for accepting to proofread and polish the language of this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elferjani, A., Garcia-Granda, S. & Dammak, M. Structural, vibrational and thermal properties of a new inorganic compound: Tl0.92(NH4)0.08(SO4)0.65(SeO4)0.35Te(OH)6. Res Chem Intermed 45, 1357–1379 (2019). https://doi.org/10.1007/s11164-018-3683-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3683-7