Abstract
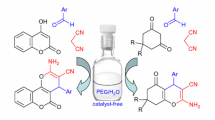
Tartaric acid is a highly effective and efficient catalyst for the one-pot synthesis of highly substituted piperidines through a combination of 1,3-dicarbonyl compounds, aromatic aldehydes, and various amines in methanol at room temperature. The advantages of this protocol are good yields, short reaction time, mild reaction conditions, no need for column chromatography, easy access, simple work-up procedure, and a cheap and biodegradable catalyst.
Graphical Abstract




Similar content being viewed by others
References
L.W. Xu, C.G. Xia, L. Li, J. Org. Chem. 69, 8482 (2004)
A. Domling, Chem. Rev. 106, 17 (2006)
F. Liéby-Muller, C. Simon, T. Constantieux, J. Rodriguez, QSAR Comb. Sci. 25, 432 (2006)
G.V.M. Sharma, K.L. Reddy, P.S. Lakshmi, P.R. Krishn, Synthesis 1, 55 (2006)
L.M. Wang, J. Sheng, L. Zhang, J.W. Han, Z.Y. Fan, H. Tian, C.T. Qian, Tetrahedron 61, 1539 (2005)
S. Benetti, R. Romagnoli, C. De Risi, G. Spalluto, V. Zanirato, Chem. Rev. 95, 1065 (1995)
P. Langer, Chem. Eur. J. 7, 3858 (2001)
P. Langer, Synthesis. 2002, 441 (2002)
C. Simon, T. Constantieux, J. Rodriguez, Eur. J. Org. Chem. 2004, 4957 (2004)
P.S. Watson, B. Jiang, B.A. Scott, Org. Lett. 2, 3679 (2000)
S. Petit, J.P. Nallet, M. Guillard, J. Dreux, R. Chermat, M. Poncelet, C. Bulach, P. Simon, C. Fontaine, M. Barthelmebs, J.L. Imbs, Eur. J. Med. Chem. 26, 19 (1991)
M. Misra, S.K. Pandey, V.P. Pandey, J. Pandey, R. Tripathi, R.P. Tripathi, Bioorg. Med. Chem. 17, 625 (2009)
I. Borza, G. Domany, Curr. Top. Med. Chem. 6, 687 (2006)
L. Mony, J.N. Kew, M.J. Gunthorpe, P. Paoletti, Br. J. Pharmacol. 157, 1301 (2009)
Y. Zhou, V.E. Gregor, B.K. Ayida, G.C. Winters, Z. Sun, D. Murphy, G. Haley, D. Bailey, J.M. Froelich, S. Fish, S.E. Webber, T. Hermann, D. Wall, Bioorg. Med. Chem. Lett. 17, 1206 (2007)
H. Bin, A.M. Crider, J.P. Stables, Eur. J. Med. Chem. 36, 265 (2001)
R. Gitto, L. De Luca, S. Ferro, F. Occhiuto, S. Samperi, G. De Sarro, E. Russo, L. Ciranna, L. Costa, A. Chimirri, Chem. Med. Chem. 3, 1539 (2008)
C.U. Kim, W. Lew, M.A. Williams, H. Liu, L. Zhang, S. Swaminathan, N. Bischofberger, M.S. Chen, D.B. Mendel, C.Y. Tai, W.G. Laver, R.C. Stevens, J. Am. Chem. Soc. 119, 681 (1997)
M. Von Itzstein, W.-Y. Wu, G.B. Kok, M.S. Pegg, J.C. Dyason, B. Jin, T.V. Phan, M.L. Smythe, H.F. White, S.W. Oliver, P.M. Colman, J.N. Varghese, D.M. Ryan, J.M. Woods, R.C. Bethell, V.J. Hothman, J.M. Camreon, C.R. Penn, Nature 363, 418 (1993)
P. Chand, P.L. Kotian, A. Dehghani, Y. El-Kattan, T.H. Lin, T.L. Hutchison, Y. Sudhakar Babu, S. Bantia, A.J. Elliott, J. Montgomery, J. Med. Chem. 44, 4379 (2001)
G.S. Jacob, Curr. Opin. Struct. Biol. 5, 605 (1995)
J.L. Treadway, P. Mendys, D.J. Hoover, Opin. Invest. Drugs 10, 439 (2001)
J.E. Groopman, Rev. Infect. Dis. 12, 931 (1990)
G.B. Karlsson, T.D. Butters, R.A. Dwek, F.M. Platt, J. Biol. Chem. 268, 570 (1993)
Y. Nishimura, T. Satoh, H. Adachi, S. Kondo, T. Takeuchi, M. Azetaka, H. Fukuyasu, Y. Iizuka, J. Med. Chem. 40, 2626 (1997)
N. Zitzmann, A.S. Mehta, S. Carrouee, T.D. Butters, F.M. Platt, J. Mc Cauley, B.S. Blumberg, R.A. Dwek, T.M. Block, Proc. Natl. Acad. Sci. USA 96, 11878 (1999)
G. Balme, E. Bossharth, N. Monteiro, Eur. J. Org. Chem. 2003, 4101 (2003)
P.A. Clarke, A.V. Zaytzev, A.C. Whitwood, Tetrahedron Lett. 48, 5209 (2007)
P.A. Clarke, A.V. Zaytzev, A.C. Whitwood, Synthesis. 2008, 3530 (2008)
A.T. Khan, M. Lal, M.M. Khan, Tetrahedron Lett. 51, 4419 (2010)
A.T. Khan, T. Parvin, L.H. Choudhury, J. Org. Chem. 73, 8398 (2008)
H.-J. Wang, L.-P. Mo, Z.-H. Zhang, ACS. Comb. Sci. 13, 181 (2011)
A.T. Khan, M.M. Khan, K.K.R. Bannuru, Tetrahedron 66, 7762 (2010)
S. Mishra, R. Ghosh, Tetrahedron Lett. 52, 2857 (2011)
S. Pal, L.H. Choudhury, T. Parvin, Mol. Divers. 16, 129 (2012)
G. Brahamachari, S. Das, Tetrahedron Lett. 53, 1479 (2012)
R. Ramachandran, S. Jayanthi, Y.T. Jeong, Tetrahedron 68, 363 (2012)
B. Umamahesh, V. Sathesh, G. Ramachandran, M. Sathishkumar, K. Sathiyanarayanan, Catal. Lett. 142, 895 (2012)
K. Zhang, M. Wang, D. Wang, C. Gao, J. Membr. Sci. 341, 246 (2009)
N. Marchitan, C. Cojocaru, A. Mereuta, G. Duca, I. Cretescu, M. Gonta, Sep. Purif. Technol. 75, 273 (2010)
A.C. Clark, P.D. Prenzler, G.R. Scollary, Food Chem. 102, 905 (2007)
H.A. Azab, I.T. Ahmed, M.R. Mahmoud, J. Chem. Eng. Data 42, 593 (1997)
S.S. Sajadikhah, M.T. Maghsoodlou, N. Hazeri, S.M. Habibi-Khorassani, S.J. Shams-Najafi, Monatsh. Chem. 143, 939 (2012)
S.S. Sajadikhah, M.T. Maghsoodlou, N. Hazeri, S.M. Habibi-Khorassani, A.C. Willis, Chin. Chem. Lett. 23, 569 (2012)
M.R. Mousavi, J. Aboonajmi, M.T. Maghsoodlou, N. Hazeri, S.M. Habibi-Khorassani, M. Safarzaei, Lett. Org. Chem. 10, 171 (2013)
J. Aboonajmi, M.R. Mousavi, M.T. Maghsoodlou T.N. Hazeri, A. Masoumnia, Res. Chem. Intermed. doi:10.1007/s11164-013-1320-z
N. Hazeri, M.T. Maghsoodlou, S.M. Habibi-Khorassani, J. Aboonajmi, S.S. Sajadikhah, J. Chin. Chem. Soc. 60, 355 (2013)
M. Lashkari, M.T. Maghsoodlou, N. Hazeri, S.M. Habibi-Khorassani, S.S. Sajadikhah, R. Doostmohamadi, Synth. Commun. 43, 635 (2013)
S.S. Sajadikhah, N. Hazeri, M.T. Maghsoodlou, S.M. Habibi-Khorassani, A. Beigbabaei, M. Lashkari, J. Chem. Res. 36, 463 (2012)
Acknowledgment
We gratefully acknowledge financial support from the Research Council of University of Sistan and Baluchestan, Iran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aboonajmi, J., Maghsoodlou, M.T., Hazeri, N. et al. Tartaric acid: a natural, green and highly efficient catalyst for the one-pot synthesis of functionalized piperidines. Res Chem Intermed 41, 8057–8065 (2015). https://doi.org/10.1007/s11164-014-1877-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-014-1877-1